Abstract
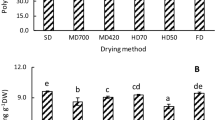
Purslane (Portulaca oleracea L.) has several health benefits, such as it reduces risk of CVD, obesity and diabetes. The objective of the study was to investigate the effect of different drying on retention of bioactive molecules, such as omega-3 fatty acids, total phenolic content and antiradical activity of purslane. Five different dehydration methods including microwave (100 MW, 5 min), tray, vacuum, low temperature low humidity, infrared were used at 55–60 °C for 5–7 h for dehydration of purslane. Three solvents, viz. water, ethanol and methanol were used for extraction of bioactive molecules from purslane. Total polyphenol content, antiradical activity and rehydration ratio of the bioactive molecules were determined. Results revealed that total PUFA, α-linolenic acid (ALA), total polyphenol content and antiradical activity were found to retain in the dried purslane in the range of (47.9–59.9 %), (42.5–50 %), (188–408GAE/100 g) and (33.0–88.8 mg/100 g) respectively. The highest values of ALA, total polyphenol content and antiradical activity were found to obtain in the vacuum dried sample. Rehydration ratio was found in the range of 3.2–4.3 and vacuum dried purslane showed maximum rehydration. It could be concluded that vacuum dehydration of purslane is an effective method for retention of bioactive molecules and good rehydration behaviour of dried purslane.


Similar content being viewed by others
References
Abbatemarco C, Ramaswamy HS (1995) End-over-end thermal processing of canned vegetables: Effect on texture and color. Food Res Int 27:327–334
Achanta S, Okos M. R (1995) Impact of drying on biological product quality. In: G. V. Barbosa-Canovas, Welti-Chanes (eds). Food preservation by moisture control: fundamentals and applications pp. 637–657
Ajayi OA, Osifo OA (1977) Home preservation of fruits and vegetables in Nigeria
AOAC (1998) Official Methods of Analysis, 16th edn. Association of Official Analytical Chemists, Washington, DC
Arshiya S, Khaleequr R (2013) Portulaca oleracea linn: a global panacea with ethnomedicinal and pharmacological potential. Int J Pharm Sci 5:33–39
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Chua KJ, Hawlader, MNA, Chou SK, Ho, JC (2002) On the study of time varying temperature drying-effect on drying kinetics and product quality. Drying Technol 20:1559–1577
Chuy, Labuza (1994) Caking and stickiness of dairy-based food powders as related to glass transition. J Food Sci 59:43–46
Cook NC, Samman S (1996) Flavonoids-chemistry, metabolism, cardioprotective effect and dietary sources. J Nutr Biochem 7:66–76
Davey MW, Van-Montagu M, Inze D, Sanmartin M, Kanellis A, Smirnoff N, Benzie IJJ, Strain JJ, Favell D, Fletcher J (2000) Plant l-ascorbic acid: Chemistry, function, metabolism, bioavailability and effects of processing. J Sci Food Agric 80:825–860
Dobiáš P, Pavlíková P, Adam M, Eisner A, Beòová B, Ventura K (2010) Comparison of pressurised fluid and ultrasonic extraction methods for analysis of plant antioxidants and their antioxidant capacity. Cent Eur J Chem 8:87–95
Dweck AC (2001). Purslane (Portulaca oleracea) - the global panacea. http://www.dweckdata.com/Published_papers/Portulaca_oleracea.pdf accessed.
Ferreres F, Gomes D, Valentão P, Gonçalves R, Pio R, Alves E, Seabra RM, Andrade PB (2009) Improved loquat (Eriobotrya japonica Lindl.) cultivars: variation of phenolics and antioxidative potential. Food Chem 114:1019–1027
Isong NC (1977) Nutrient losses in dehydrated foods. Crit Rev Food Technol 1:49–55
Karel M, Buera MP, Roos Y (1993) Effects of glass transitions on processing and storage. In J. M. V. Blanshard, & P. J. Lillford (eds), The glassy state in food. pp 12–34
Khanal RC, Howard LR, Prior RL (2010) Effect of heating on the stability of grape and blueberry pomace procyanidins and total anthocyanins. Food Res Int 43:1464–1469
Luh BS, Woodroof JG (1975) Commercial Vegetable processing. Avi. Pub. Co., Westport, Corn
Miliauskas G, Venskutonis PR, Van Beek TA (2004) Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem 2:231–237
Navarro JM, Flores P, Garrido C, Martinez V (2006) Changes in the contents of antioxidant compounds in pepper fruits at different ripening stages, as affected by salinity. Food Chem 96:66–73
O’Fallon JV, Busboom JR, Nelson ML, Gaskins CT (2007) A direct method for fatty acid methyl ester synthesis: Application to wet meat tissues, oils, and feedstuffs. J Anim Sci 85:1511–1521
Pande VK, Sonune AV, Philip SK (2000) Solar drying of coriander and methi leaves. J Food Sci Technol 23:639–641
Pervin S, Islam MS, Islam MN (2008) Study on Rehydration Characteristics of Dried Lablab Bean (Lablab Purpureus) Seeds. J Agric Rural Dev 6(1&2):157–163
Raghavan GSV, Orsat V (2007) Recent advances in drying of biomaterials for superior quality bioproducts. Asia-Pacific J Chem Eng 2:20–29
Ranganna S (1997) Handbook of Analysis and Quality Control of Fruit and Vegetable Products, 2nd edn. Tata McGraw Hill Publications. Co, New Delhi
Ross KA, Beta T, Arntfield SD (2009) A comparative study on the phenolic acids identified and quantified in dry beans using HPLC as affected by different extraction and hydrolysis methods. Food Chem 113:336–344
Samy J, Sugumaran M, Lee KLW (2004) Herbs of Malaysia: An Introduction to the medicinal, culinary, aromatic and cosmetic use of herbs. Times Edition, Kuala Lumpur
Schieber A, Keller P, Carle R (2001) Determination of phenolic acids and flavonoids of apple and pear by high-performance liquid chromatography. J Chromatogr A 910:265–273
Serrano M, Zapata PJ, Castillo S, Guillén F, Martínez-Romero D, Valero D (2010) Antioxidant and nutritive constituents during sweet pepper development and ripening are enhanced by nitrophenolate treatments. Food Chem 118:497–503
Sheetal G, Gowri BS, Lakshmi AJ, Prakash J (2013) Retention of nutrients in green leafy vegetables on dehydration. J Food Sci Technol. doi:10.1007/s13197-011-0407
Singleton VL, Rossi JA Jr (1965) Colorunetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Viticult 16:144–158
Whelan J, Rust C (2006) Innovative Dietary Sources of N-3 Fatty Acids. Annu Rev Nutr 26:75–103
Wijngaard HH, Rößle C, Brunton NA (2009) Survey of Irish fruit and vegetable waste and byproducts as a source of polyphenolic antioxidants. Food Chem 116:202–207
Yao L, Jiang Y, Datta N, Singanusong R, Liu X, Duan J (2004) HPLC analyses of flavonols and phenolic acids in the fresh young shoots of tea (Camellia sinensis) grown in Australia. Food Chem 84:253–263
Acknowledgement
The authors thank Prof. Ram Rajasekharan, Director, CSIR-CFTRI, Mysore for his constant encouragement and support. The first author gratefully acknowledges U.G.C for providing the financial assistance in the form of JRF to carry out the present investigation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shanker, N., Debnath, S. Impact of dehydration of purslane on retention of bioactive molecules and antioxidant activity. J Food Sci Technol 52, 6631–6638 (2015). https://doi.org/10.1007/s13197-015-1741-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-015-1741-3