Abstract
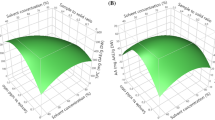
In this study, the optimal conditions for the extraction of antioxidants from hard winter wheat bran (WH711) were determined using response surface methodology (RSM). A central composite face centred design was used to investigate the effects of three independent variables, namely solvent composition (%v/v), extraction temperature (°C) and time (minutes) on the responses; free phenolic content (FPC), total flavonoid content (TFC), total antioxidant activity (TAA), ferric reducing power (FRP), hydrogen peroxide scavenging activity (HPSA). Regression analysis showed that more than 90% of the variation was explained by the models of different responses. The optimal conditions for the antioxidantss extraction were found to be methanol concentration of 85%v/v, extraction temperature of 75 °C, extraction time of 45 min, for wheat bran. The experimental values of FPC, TFC, TAA, FRP and HPSA were 0.921 mg GAE/g bran (B), 0.4588 mg CE/g B, 0.01408 mM AAE/g B, 2.532 mM AAE/g B and 3.193 mM TE/g B, respectively which agreed with those predicted, thus indicating suitability of the model employed and the suitability of RSM in optimizing the extraction conditions.






Similar content being viewed by others
References
Alam MA, Rahman MM, Subhan N, Majumder MM, Hasan SMR, Kter RM, Majumder EH, Faruque A (2009) Antioxidant potential of the ethanol extract of the leaves of Vitex Negundol L. Turk J Pharm Sci 6(1):11–20
Cacace JE, Mazza G (2002) Extraction of anthocyanin and other phenolics from black currants with sulfured water. J Agric Food Chem 50:5939–5946
Chan SW, Lee CY, Yap CF, Wan Aida WM, Ho CW (2009) Optimisation of extraction conditions for phenolic compounds from limau purut (Citrus hystrix) peels. Int Food Res J 16:203–213
Cheng Z, Su L, Moore J, Zhou K, Luther M, Yin JJ, Yu LL (2006) Effect of postharvest treatment and heat stress on availability of wheat antioxidants. J Agric Food Chem 54:5623–5629
Duh PD (1998) Antioxidant activity of Budrock (Arctium Laooa Linn): its scavenging effect on free radical and active oxygen. J Am Oil Chem 75:455–461
Duh PD, Tu YY, Yen GC (1999) Antioxidant activity of water extracts of Harng Jyur (Chrisanthemun morifolium Ramat). Lebensm Wiss Technol 32:269–277
Graf E (1992) Antioxidant potential of ferulic acid. Free Radic Biol Med 13:435–448
He GQ, Xiong HP, Chen QH, Ruan H, Wang ZY, Traore L (2005) Optimization of conditions for supercritical fluid extraction of flavonoids from hops (Humulus lupulus L.). J Zhejiang Univ Sci 6B(10):999–1004
Iqbal S, Bhanger MI, Anwar F (2005) Antioxidant properties and components of bran extracts from selected wheat varieties commercially available in Pakistan. Lebensm Wiss Technol 40:361–367
Jayaprakasha GK, Negi PS, Jena BS, Rao LJM (2007) Antioxidant and antimutagenic activities of Cinnamomum zeylanicum fruit extracts. J Food Compos Anal 20:330–336
Jayaprakasha GK, Jena BS, Negi PS, Sakariah KK (2002) Evaluation of antioxidant activities and antimutagenicity of turmeric oil: a byproduct from curcumin production. Z Naturforsch 57c:828–835
Kim K, Tsao R, Yang R, Cui SW (2006) Phenolic acid profiles and antioxidant activities of wheat bran extracts and the effects of hydrolysis conditions. Food Chem 95:466–473
Kumar S, Kumar D, Singh N, Vasisht BD (2007) In vitro, free radicals scavenging and antioxidant activity of Moringa Oleifera pods. J Herb Med Toxicol 1(2):17–22
Kumar ST, Baskar R, Shanmugam S, Rajsekaran P, Sadasivam S, Manikandan V (2008) Optimization of flavonoids extraction from the leaves of Tabernaemontana heyneana Wall. using L16 Orthogonal design. Nat Sci 6(3):14–25
Li W, Shan F, Sun S, Corke H, Beta T (2005) Free radical scavenging properties and phenolic content of Chinese black-grained wheat. J Agric Food Chem 53:8533–8536
Liyana-Pathirana CM, Shahidi F (2005) Optimization of extraction of phenolic compounds from wheat using response surface methodology. Food Chem 93:47–56
Luque De Castro MD, Tena MT (1996) Strategies for supercritical fluid extraction of polar and ionic compounds. Trends Anal Chem 15:32–37
Marama M, Amarowicz R, Weidner S, Abe S, Shahidi F (2004) Antioxidant activity of Triticale caryopses and embryos extracts. Food Sci Biotechnol 13:421–424
Mpofu A, Sapirstein HD, Beta T (2006) Genotype and environmental variation in phenolic content, phenolic acid composition, and antioxidant activity of hard spring wheat. J Agric Food Chem 54:1265–1270
Negi PS, Chauhan AS, Sadia GA, Rohinishree YS, Ramteke RS (2005) Antioxidant and antibacterial activities of various seabuckthorn (Hippophae rhamnoides L.) seed extracts. Food Chem 92:119–124
Nieva Moreno MI, Isla MI, Sampietro AR, Vattuone MA (2000) Comparison of the free radicalscavenging activity of propolis from several regions of Argentina. J Ethnopharmacol 71:109–114
Onyeneho SN, Hettiarachchy NS (1992) Antioxidant activity of durum wheat bran. J Agric Food Chem 40(9):1496–1500
Oyaizu M (1986) Studies on products of browning reaction: antioxidative activities of products of browning reaction prepared from glucosamine. Jpn J Nutr 44:307–315
Ozddemir M, Ozen BF, Dock LL, Floros JD (2008) Optimization of osmostic dehydration of diced green peppers by response surface methodology. Lebensm Wiss Technol 41:2044–2050
Pinzino C (1999) Aging, free radicals, and antioxidants in wheat seeds. J Agric Food Chem 47:1333–1339
Qu H, Madl RL, Takemoto DJ, Baybutt RC, Wang W (2005) Lignans are involved in the antitumor activity of wheat bran in colon cancer SW480 cells. J Nutr 135:598–602
Ramamoorthy PK, Bongo A (2007) Antioxidant activity total phenolic and flavonoid content of Morinda Citrifolia fruit extracts from various extraction processes. J Eng Sci Technol 2(1):70–80
Ruch RJ, Cheng SJ, Klaunig JE (1989) Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 10:1003–1008
Satoh E, Tohyama N, Nishimura M (2005) Comparison of the antioxidant activity of roasted tea with green, oolong, and black teas. Int J Food Sci Nutr 56:551–559
Shahidi F (2000) Antioxidants in food and food antioxidants. Nahrung 44:158–163
Silva EM, Rogez H, Larondelle Y (2007) Optimization of extraction of phenolic from Inga edulis leaves using response surface methodology. Sep Purif Technol 55:381–387
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Turkmen N, Velioglu Y, Sedat SF, Polat G (2007) Effect of extraction conditions on measured total polyphenol contents and antioxidant and antibacterial activities of black tea. Molecules 12:484–496
Wang S, Chen F, Wu J, Wang Z, Liao X, Hu X (2007) Optimization of pectin extraction assisted by microwave from apple pomace using response surface methodology. J Food Eng 78:693–700
Wettasinghe M, Shahidi F (1999) Evening primrose meal: a source of natural antioxidants and scavenger of hydrogen peroxide and oxygen-derived free radicals. J Agric Food Chem 47:1801–1812
Yen WJ, Chen BH (1995) Isolation of xanthophylls from Taiwanese orange peels and their effects on the stability of soybean oil. Food Chem 53:417–426
Yilmaz Y, Toledo RT (2006) Oxygen radical absorbance capacities of grape/wine industry byproducts and effect of solvent type on extraction of grape seed polyphenols. J Food Compos Anal 19:41–48
Yu L (2007) Wheat antioxidants. Wiley, New Jersey, pp 102–104
Yu L, Haley S, Perret J, Harris M (2002a) Antioxidant properties of hard winter wheat extracts. Food Chem 78:457–461
Yu L, Haley S, Perret J, Harris M, Wilson J, Qian M (2002b) Free radical scavenging properties of wheat extracts. J Agric Food Chem 50:1619–1624
Zhou K, Su L, Yu L (2004) Phytochemical and antioxidant properties in wheat bran. J Agric Food Chem 52:6108–6114
Zielinski H, Kozlowska H (2000) Antioxidant activity and total phenolics in selected cereal grains and their different morphological fractions. J Agric Food Chem 48:2008–2016
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, B., Sharma, H.K. & Sarkar, B.C. Optimization of extraction of antioxidants from wheat bran (Triticum spp.) using response surface methodology. J Food Sci Technol 49, 294–308 (2012). https://doi.org/10.1007/s13197-011-0276-5
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-011-0276-5