Abstract
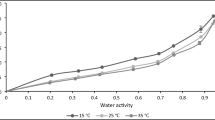
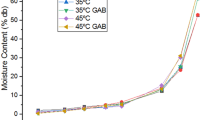
The equilibrium moisture content was determined for un-osmosed and osmosed (salt osmosed and sugar osmosed) aonla flakes using the static method at temperatures of 25, 40,50, 60 and 70 °C over a range of relative humidities from 20 to 90%. The sorption capacity of aonla decreased with an increase in temperature at constant water activity. The sorption isotherms exhibited hysteresis, in which the equilibrium moisture content was higher at a particular equilibrium relative humidity for desorption curve than for adsorption. The hysteresis effect was more pertinent for un-osmosed and salt osmosed samples in comparison to sugar osmosed samples. Five models namely the modified Chung Pfost, modified Halsey, modified Henderson, modified Exponential and Guggenheim-Anderson-de Boer (GAB) were evaluated to determine the best fit for the experimental data. For both adsorption and desorption process of aonla fruit, the equilibrium moisture content of un-osmosed and osmosed aonla samples can be predicted well by GAB model as well as modified Exponential model. Moreover, the modified Exponential model was found to be the best for describing the sorption behaviour of un-osmosed and salt osmosed samples while, GAB model for sugar osmosed aonla samples.


Similar content being viewed by others
Abbreviations
- Me:
-
: Equilibrium moisture content, decimal (dry basis)
- Me*:
-
: Equilibrium moisture content, (% d.b)
- rh:
-
: Equilibrium relative humidity, decimal
- T:
-
: Temperature (°K)
- A:
-
: Model equilibrium constant indicating sorption capacity
- B:
-
: Model equilibrium constant indicating temperature of environment
- C:
-
: Model equilibrium constant indicating relative humidity
- b, c,h1,h2 :
-
: Constants
- r2 :
-
: Coefficient of determination
- RMSE:
-
: Root mean square error
- P:
-
: Mean deviation modulus (%)
- UO:
-
: Un-Osmosed aonla sample
- Salt O:
-
: Salt osmosed aonla sample
- Sugar O:
-
: Sugar osmosed aonla sample
References
AOAC (2000) Official methods of analysis (984.25), 17th edn. Association of Official Analytical Chemists, Maryland
Basu S, Shivhare US, Majumdar AS (2006) Models for sorption isotherm for foods: a review. Drying Technol 24:917–930
Berg CV, Bruin S (1981) Water activity and its estimation in food systems: theoretical aspects. In: Rockland LB, Stewart GF (eds) Water activity: influences on food quality. Academic, London, pp 189–200
Chirife J, Iglesias HA (1978) Equations for fitting water sorption isotherms of foods. J Food Technol 13:159–174
Chung DS, Pfost HB (1967) Adsorption and desorption of water vapor by cereal grains and their products. Part III: A hypothesis for explaining the hysteresis effect. Trans ASAE 10:556–557
Dhingra D (2003) Optimization of the drying process of garlic (Allium sativum L.), Ph.D dessertation, Punjab Agricultural University, Ludhiana, India
Gomez KA, Gomez AA (1983) Statistical procedure for agricultural research. Wiley, New York, pp 356–422
Hossain MA, Bala BK (2000) Moisture isotherm characteristics for red chilli. Drying Technol 18:503–515
Hutchinson DH, Otten L (1984) Equilibrium moisture content of white beans. Cereal Chem 61(2):155–58
Iglesias HA, Chirife J (1976) Prediction of the effect of temperature on water sorption isotherms of food materials. J Food Technol 11:109–113
Jayaraj RK, Balasubramanyam BV, Ghosh BC, Kulkarni S (2007) Sorption isotherms of chhana podo. J Fd Sci Technol 44(2):198–200
Khalloufi S, Giasson J, Ratti C (2000) Water activity of freeze dried mushrooms and berries. Can Agric Eng 42:51–56
King CJ (1968) Rates of moisture sorption and desorption in porous, dried food stuffs. Food Technol 22:509–514
Labuza TP (1968) Sorption phenomena in foods. Food Technol 22(2):263–268
Lapinski M, Kostyrkoand K, Wlodarski W (1976) Modern methods for control and measurement of humidity and moisture. Translated from polish. Published by the Foreign Scientific, Technical and Economic Information, Warsaw, Poland
Lazarides HN, Nickolaidis A, Katsanidis E (1995) Sorption changes induced by osmotic pre-concentration of apple slices in different osmotic media. J Food Sci 60:348–350, 359
Mayor L, Moreira R, Chenlo F, Sereno AM (2005) Water sorption isotherms of fresh and partially osmotic dehydrated pumpkin parenchyma and seeds at several temperatures. Eur Food Res Technol 220:163–167
Menkov ND, Durakova AG (2007) Moisture sorption isotherms of sesame flour. Food Technol Biotechnol 45(1):96–100
Pacco HC, Vigneault C, Menegalli FC, Castro Rde, Cortez Luis AB (2008) Evaluation of sorption models for figs. Can Biosyst Eng 50:3.77–3.83
Patil KN, Grover PC (1986) Adsorption and desorption of green gram. J Agric Eng 24(1):40–46
Pawar VS, Dev DK, Pawar VD, Rodge AB, Surve VD, More DR (1992) Moisture adsorption isotherms of ground turmeric at different temperatures. J Fd Sci Technol 29:170–173
Prothon F, Ahrne LM (2004) Application of the Guggenheim-Anderson-de boer model to correlate water activity and moisture content during osmotic dehydration of apples. J Food Eng 61:467–470
Raquel PFG (2009) Sorption isotherms of pears using different models. Int J Fruit Sci 9:11–22
Shivhare US, Arora S, Ahmed J, Raghavan GSV (2004) Moisture adsorption isotherm for mushroom. Lebensmittel Technologie 37:133–137
Sitkiewicz I, Lenart A, Lewicki PP (1996) Mechanical properties of osmotic-convection dried apples. Polish J of Food Nut and Sci p 46,105
Soysal Y, Oztekin S (2001) Comparison of seven equilibrium moisture content equations for some medicinal and aromatic plants. J Agric Eng Res 78:73–79
Suresh KP, Sagar VR (2009) Influence of packaging materials and storage temperature on quality of osmo-vac dehydrated aonla segments. J Fd Sci Technol 46(3):259–262
Young JH (1976) Evaluation of models to describe sorption and desorption moisture content isotherms of Virginia type peanuts. Trans ASAE 19(1):146–150
Yu L, Mazza G, Jayas DS (1999) Moisture sorption characteristics of freeze-dried, osmo-freeze-dried cherries and blueberries. Trans ASAE 42:141–147
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alam, M.S., Singh, A. Sorption isotherm characteristics of aonla flakes. J Food Sci Technol 48, 335–343 (2011). https://doi.org/10.1007/s13197-011-0249-8
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-011-0249-8