Abstract
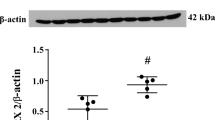
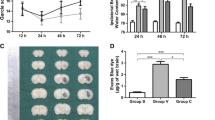
Carbonic anhydrase 1 (CA-1) is a metalloenzyme present at high concentrations in erythrocytes. Our previous studies showed that erythrocyte lysis contributes to brain edema formation after intracerebral hemorrhage (ICH), and a recent study indicates that CA-1 can cause blood–brain barrier disruption. The present study investigated the role of CA-1 in ICH-induced brain injury. There were three groups in the study. In the first, adult male Sprague Dawley rats received 100 μl autologous blood injection into the right caudate. Sham rats had a needle insertion. Rat brains were used for brain CA-1 level determination. In the second group, rats received an intracaudate injection of either 50 μl CA-1 (1 μg/μl) or saline. Brain water content, microglia activation, and neuronal death (Fluoro-Jade C staining) were examined 24 h later. In the third group, acetazolamide (AZA, 5 μl, 1 mM), an inhibitor of carbonic anhydrases, or vehicle was co-injected with 100 μl blood. Brain water content, neuronal death, and behavioral deficits were measured. We found that CA-I levels were elevated in the ipsilateral basal ganglia at 24 h after ICH. Intracaudate injection of CA-1 induced brain edema (79.0 ± 0.6 vs. 78.0 ± 0.2% in the saline group, p < 0.01), microglia activation, and neuronal death (p < 0.01) at 24 h. AZA, an inhibitor of CA, reduced ICH-induced brain water content (79.3 ± 0.7 vs. 81.0 ± 1.0% in the vehicle-treated group, p < 0.05), neuronal death, and improved functional outcome (p < 0.05). These results suggest that CA-1 from erythrocyte lysis contributes to brain injury after ICH.







Similar content being viewed by others
References
Xi G, Keep RF, Hoff JT. Erythrocytes and delayed brain edema formation following intracerebral hemorrhage in rats. J Neurosurg. 1998;89:991–6.
Wu J, Hua Y, Keep RF, Schallert T, Hoff JT, Xi G. Oxidative brain injury from extravasated erythrocytes after intracerebral hemorrhage. Brain Res. 2002;953(1–2):45–52.
Xi G, Keep RF, Hoff JT. Mechanisms of brain injury after intracerebral hemorrhage. Lancet Neurol. 2006;5(1):53–63.
Hua Y, Schallert T, Keep RF, Wu J, Hoff JT, Xi G. Behavioral tests after intracerebral hemorrhage in the rat. Stroke. 2002;33(10):2478–84.
Huang F, Xi G, Keep RF, Hua Y, Nemoianu A, Hoff JT. Brain edema after experimental intracerebral hemorrhage: role of hemoglobin degradation products. J Neurosurg. 2002;96(2):287–93.
Wu G, Xi G, Hua Y, Sagher O. T2* magnetic resonance imaging sequences reflect brain tissue iron deposition following intracerebral hemorrhage. Translat Stroke Res. 2010;1:31–4.
Gao BB, Clermont A, Rook S, Fonda SJ, Srinivasan VJ, Wojtkowski M, et al. Extracellular carbonic anhydrase mediates hemorrhagic retinal and cerebral vascular permeability through prekallikrein activation. Nat Med. 2007;13(2):181–8.
Xi G, Keep RF, Hua Y, Xiang JM, Hoff JT. Attenuation of thrombin-induced brain edema by cerebral thrombin preconditioning. Stroke. 1999;30(6):1247–55.
Okauchi M, Xi G, Keep RF, Hua Y. Tissue-type transglutaminase and the effects of cystamine on intracerebral hemorrhage-induced brain edema and neurological deficits. Brain Res. 2009;1249:229–36.
Schmued LC, Albertson C, Slikker Jr W. Fluoro-Jade: a novel fluorochrome for the sensitive and reliable histochemical localization of neuronal degeneration. Brain Res. 1997;751:37–46.
Gong Y, Hua Y, Keep RF, Hoff JT, Xi G. Intracerebral hemorrhage: effects of aging on brain edema and neurological deficits. Stroke. 2004;35(11):2571–5.
Xi G, Hua Y, Bhasin RR, Ennis SR, Keep RF, Hoff JT. Mechanisms of edema formation after intracerebral hemorrhage: effects of extravasated red blood cells on blood flow and blood–brain barrier integrity. Stroke. 2001;32(12):2932–8.
Sly WS, Hu PY. Human carbonic anhydrases and carbonic anhydrase deficiencies. Annu Rev Biochem. 1995;64:375–401.
Ropper AH, King RB. Intracranial pressure monitoring in comatose patients with cerebral hemorrhage. Arch Neurol. 1984;41(7):725–8.
Ropper AH. Lateral displacement of the brain and level of consciousness in patients with an acute hemispheral mass. N Engl J Med. 1986;314(15):953–8.
Zazulia AR, Diringer MN, Derdeyn CP, Powers WJ. Progression of mass effect after intracerebral hemorrhage. Stroke. 1999;30:1167–73.
Wagner KR, Xi G, Hua Y, Kleinholz M, de Courten-Myers GM, Myers RE, et al. Lobar intracerebral hemorrhage model in pigs: rapid edema development in perihematomal white matter. Stroke. 1996;27(3):490–7.
Lee KR, Colon GP, Betz AL, Keep RF, Kim S, Hoff JT. Edema from intracerebral hemorrhage: the role of thrombin. J Neurosurg. 1996;84(1):91–6.
Xi G, Wagner KR, Keep RF, Hua Y, de Courten-Myers GM, Broderick JP, et al. The role of blood clot formation on early edema development following experimental intracerebral hemorrhage. Stroke. 1998;29:2580–6.
Hua Y, Xi G, Keep RF, Hoff JT. Complement activation in the brain after experimental intracerebral hemorrhage. J Neurosurg. 2000;92(6):1016–22.
Xi G, Hua Y, Keep RF, Younger JG, Hoff JT. Systemic complement depletion diminishes perihematomal brain edema. Stroke. 2001;32(1):162–7.
Mendelow AD. Mechanisms of ischemic brain damage with intracerebral hemorrhage. Stroke. 1993;24(12 Suppl):I115–7. discussion I8-9.
Maier-Hauff K, Baethmann AJ, Lange M, Schurer L, Unterberg A. The kallikrein-kinin system as mediator in vasogenic brain edema. Part 2: studies on kinin formation in focal and perifocal brain tissue. J Neurosurg. 1984;61(1):97–106.
Zweckberger K, Plesnila N. Anatibant, a selective non-peptide bradykinin B2 receptor antagonist, reduces intracranial hypertension and histopathological damage after experimental traumatic brain injury. Neurosci Lett. 2009;454(2):115–7.
Ding-Zhou L, Margaill I, Palmier B, Pruneau D, Plotkine M, Marchand-Verrecchia C. LF 16–0687 Ms, a bradykinin B2 receptor antagonist, reduces ischemic brain injury in a murine model of transient focal cerebral ischemia. Br J Pharmacol. 2003;139(8):1539–47.
Gardner TW, Antonetti DA. A prize catch for diabetic retinopathy. Nat Med. 2007;13(2):131–2.
Wagner KR, Kleinholz M, de Courten-Myers GM, Myers RE. Hyperglycemic versus normoglycemic stroke: topography of brain metabolites, intracellular pH, and infarct size. J Cereb Blood Flow Metab. 1992;12(2):213–22.
Qureshi AI, Mendelow AD, Hanley DF. Intracerebral haemorrhage. Lancet. 2009;373(9675):1632–44.
Mun-Bryce S, Kroh FO, White J, Rosenberg GA. Brain lactate and pH dissociation in edema: 1H- and 31P-NMR in collagenase-induced hemorrhage in rats. Am J Physiol. 1993;265(3 Pt 2):R697–702.
Wagner KR, Xi G, Hua Y, Kleinholz M, de Courten-Myers GM, Myers RE. Early metabolic alterations in edematous perihematomal brain regions following experimental intracerebral hemorrhage. J Neurosurg. 1998;88(6):1058–65.
Fujishima M, Sugi T, Choki J, Yamaguchi T, Omae T. Cerebrospinal fluid and arterial lactate, pyruvate and acid-base balance in patients with intracranial hemorrhages. Stroke. 1975;6(6):707–14.
Sugi T, Fujishima M, Omae T. Lactate and pyruvate concentrations, and acid-base balance of cerebrospinal fluid in experimentally induced intracerebral and subarachnoid hemorrhage in dogs. Stroke. 1975;6(6):715–9.
Fedirko N, Avshalumov M, Rice ME, Chesler M. Regulation of postsynaptic Ca2+ influx in hippocampal CA1 pyramidal neurons via extracellular carbonic anhydrase. J Neurosci. 2007;27(5):1167–75.
Velisek L, Moshe SL, Stanton PK. Resistance of hippocampal synaptic transmission to hypoxia in carbonic anhydrase II-deficient mice. Brain Res. 1995;671(2):245–53.
Kniep EM, Roehlecke C, Ozkucur N, Steinberg A, Reber F, Knels L, et al. Inhibition of apoptosis and reduction of intracellular pH decrease in retinal neural cell cultures by a blocker of carbonic anhydrase. Invest Ophthalmol Vis Sci. 2006;47(3):1185–92.
Hackett PH, Roach RC. High-altitude illness. N Engl J Med. 2001;345(2):107–14.
Celebisoy N, Gokcay F, Sirin H, Akyurekli O. Treatment of idiopathic intracranial hypertension: topiramate vs acetazolamide, an open-label study. Acta Neurol Scand. 2007;116(5):322–7.
Lam DS, Lai JS, Tham CC, Chua JK, Poon AS. Argon laser peripheral iridoplasty versus conventional systemic medical therapy in treatment of acute primary angle-closure glaucoma: a prospective, randomized, controlled trial. Ophthalmology. 2002;109(9):1591–6.
Katayama F, Miura H, Takanashi S. Long-term effectiveness and side effects of acetazolamide as an adjunct to other anticonvulsants in the treatment of refractory epilepsies. Brain Dev. 2002;24:150–4.
Popa G, Amaireh M, Dinu M, Jipescu I, Alaicescu M, Stanescu A, et al. Acetazolamide therapy evaluation in haemorrhagic stroke. Rom J Neurol Psychiatry. 1995;33(2):145–55.
Kennedy CR, Ayers S, Campbell MJ, Elbourne D, Hope P, Johnson A. Randomized, controlled trial of acetazolamide and furosemide in posthemorrhagic ventricular dilation in infancy: follow-up at 1 year. Pediatrics. 2001;108(3):597–607.
Supuran CT. Carbonic anhydrases: novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov. 2008;7(2):168–81.
Pan P, Leppilampi M, Pastorekova S, Pastorek J, Waheed A, Sly WS, et al. Carbonic anhydrase gene expression in CA II-deficient (Car2−/−) and CA IX-deficient (Car9−/−) mice. J Physiol. 2006;571(Pt 2):319–27.
Svichar N, Esquenazi S, Waheed A, Sly WS, Chesler M. Functional demonstration of surface carbonic anhydrase IV activity on rat astrocytes. Glia. 2006;53(3):241–7.
Hua Y, Keep R, Hoff J, Xi G. Brain injury after intracerebral hemorrhage: the role of thrombin and iron. Stroke. 2007;38:759–62.
Siffert W, Fox G, Gros G. The effect of carbonic anhydrase inhibitin on the velocity of thrombin-stimulated platelet aggregation under physiological condition. Biochem Biophys Res Commun. 1984;121(1):266–70.
Acknowledgments
This study was supported by grants NS-017760, NS-039866, and NS-057539 from the National Institutes of Health (NIH) and 0840016N from the American Heart Association (AHA). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH and AHA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, F., Hua, Y., Wang, J. et al. Inhibition of Carbonic Anhydrase Reduces Brain Injury After Intracerebral Hemorrhage. Transl. Stroke Res. 3, 130–137 (2012). https://doi.org/10.1007/s12975-011-0106-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-011-0106-0