Abstract
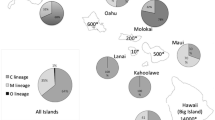
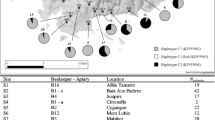
The black honeybee (Apis mellifera mellifera; M lineage) which is affected by widespread colony losses, colony introgression and population shifts within its natural range of distribution, needs proper protection and management planning. The DraI COI-COII test discriminates between and within honeybee lineages (M, C, A and O). With this test, a global assessment of the diversity found in the M lineage (i.e. description of haplotype frequencies, fragment length and size, nucleotide sequences) is made with colonies newly sampled in France and Belgium (n = 3,261 colonies) and previously described in the literature (n = 3,367). A total of 91 M haplotypes were described including 52 new ones. The haplotypes M4 and M4’ were the most frequent followed by M17, M7, M6, M19, M8, M17’ and by many rare ones. Finally, these data were used to infer hypotheses on the putative origin of the M haplotypic diversity and to determine a protocol for the establishment of protected areas of black honeybee colonies. This protocol is based on the DraI COI-COII test and on case-studies of protected areas of black honeybees from France.


Similar content being viewed by others
References
Arias MC, Sheppard WS (1996) Molecular phylogenetics of honeybee subspecies (Apis mellifera L.) inferred from mitochondrial DNA sequence. Mol Phylogenet Evol 5:557–566
Clarke KE, Olroyd BP, Javier J, Quezada-Euán G, Rinderer TE (2001) Origin of honeybees (Apis mellifera L.) from the Yucatan peninsula inferred from mitochondrial DNA analysis. Mol Ecol 10:1347–1355
Collet T, Ferreira KM, Arias MC, Soares AEE, Del Lama MA (2006) Genetic structure of Africanized honeybee populations (Apis mellifera L.) from Brazil and Uruguay viewed through mitochondrial DNA COI-COII patterns. Heredity 97:323–335
Cornuet JM, Garnery L (1991) Mitochondrial DNA variability in honeybees and its phylogeographic implications. Apidologie 22:627–642
Crozier RH, Crozier YC, Mackinlay AG (1989) The CO-I and CO-II region of the honeybee mitochondrial DNA: evidence for variation in insect mitochondrial evolutionary rates. Mol Biol Evol 6:399–695
De la Rúa P, Galián J, Serrano J (1998) Mitochondrial variability in the Canary Islands honeybees. Mol Ecol 7:1543–1547
De la Rúa P, Galián J, Serrano J, Moritz RFA (2001a) Molecular characterization and population structure of the honeybees from the Balearic Islands (Spain). Apidologie 32:417–427
De la Rúa P, Galián J, Serrano J, Moritz RFA (2001b) Genetic structure and distinctness of Apis mellifera L. populations from the Canary islands. Mol Ecol 10:1733–1742
De la Rúa P, Jiménez Y, Galián J, Serrano J (2004) Evaluation of the biodiversity of honey bee (Apis mellifera) populations from Eastern Spain. J Api Res 43:162–166
De la Rúa P, Hernández-García R, Jiménez Y, Galián J, Serrano J (2005) Biodiversity of Apis mellifera iberica (Hymenoptera: Apidae) from northeastern Spain assessed by mitochondrial analysis. Insect Syst. Evol 36:21–28
Dietemann V, Pirk CWW, Crewe R (2009) Is there a need for conservation of honeybees in Africa? Apidologie 40:285–295
EFSA European Food Safety Authority (2009) Bee mortality and bee surveillance in Europe. CFP/EFSA/AMU/2008/02. Scientific Report, Question number EFSA-Q-2009-00801. P 217
Franck P, Garnery L, Solignac M, Cornuet J-M (1998) The origin of West European subspecies of honeybees (Apis mellifera): new insights from microsatellite and mitochondrial data. Evolution 52:1119–1134
Franck P, Garnery L, Celebrano G, Solignac M, Cornuet J-M (2000a) Hybrid origin of honeybees from Italy (Apis mellifera ligustica) and Sicily (A. m. sicula). Mol Ecol 9:907–921
Franck P, Garnery L, Solignac M, Cornuet J-M (2000b) Molecular confirmation of a Middle East lineage in Apis mellifera. Apidologie 31:167–180
Franck P, Garnery L, Loiseau A, Oldroyd BP, Hepburn HR, Solignac M, Cornuet J-M (2001) Genetic diversity of the honeybee in Africa: microsatellite and mitochondrial data. Heredity 86:420–430
Garnery L, Cornuet J-M, Solignac M (1992) Evolutionary history of the honey bee Apis mellifera inferred from mitochondrial DNA analysis. Mol Ecol 1:145–154
Garnery L, Solignac M, Celebrano G, Cornuet J-M (1993) A simple test using restricted PCR-amplified mitochondrial DNA to study the genetic structure of Apis mellifera L. Experientia 49:1016–1021
Garnery L, Mosshine EH, Cornuet J-M (1995) Mitochondrial DNA variation in Moroccan and Spanish honey bee populations. Mol Ecol 4:465–471
Garnery L, Franck P, Baudry E, Vautrin D, Cornuet J-M, Solignac M (1998a) Genetic biodiversity of the West European honeybee (Apis mellifera mellifera and A. m. iberica). I. Mitochondrial DNA. Genet Sel Evol 30:31–47
Garnery L, Franck P, Baudry E, Vautrin D, Cornuet J-M, Solignac M (1998b) Genetic biodiversity of the West European honeybee (Apis mellifera mellifera and A. m. iberica). II. Microsatellite DNA. Genet Sel Evol 30:49–74
Jaffé R, Dietmann V, Alssopp M, Costa C, Crew R, Dall’Olio R, De la Rúa P, El-Niweiri Mogbel AA, Fries I, Kezic N, Meusel M, Paxton RJ, Shaibi T, Stolle E, Moritz RFA (2009) Estimating the density of honeybee colonies across their natural range to fill the gap in pollinator decline censuses. Cons Bio 24(2):583–591
Jensen AB, Palmer KA, Boomsma JJ, BoV Pedersen (2005a) Varying degrees of Apis mellifera ligustica introgression in protected populations of the black honeybee, Apis mellifera mellifera, in northwest Europe. Mol Ecol 14:93–106
Jensen AB, Palmer KA, Chaline N, Raine NE, Tofilski NE, Martin SJ, BoV Pedersen, Boomsma JJ, Ratnieks FLW (2005b) Quantifying honey bee mating range and isolation in semi-isolated valleys by DNA microsatellite paternity analysis. Conserv Biol 6:527–537
Kandemir I, Kence M, Sheppard WS, Kence A (2006a) Mitochondrial DNA variation in honey bee (Apis mellifera L.) populations from Turkey. J Api Res 45:33–38
Kandemir I, Meixner MD, Ozkan A, Sheppard WS (2006b) Genetic characterization of honey bee (Apis mellifera cypria) populations in northern Cyprus. Apidologie 37:547–555
Kauhausen-Keller D, Keller R (1994) Morphometrical control of pure race breeding of honeybee (Apis mellifera L.). Apidologie 25:133–143
Maul V, Hähnle A (1994) Morphometric studies with purebred stock of Apis mellifera carnica Pollmann from Hessen. Apidologie 25:19–132
Miguel I, Iriondo M, Garnery L, Sheppard WS, Estonba A (2007) Gene flow within the M evolutionary lineage of Apis mellifera: role of the Pyrenees, isolation by distance and post-glacial recolonization routes in the Western Europe. Apidologie 38:141–155
Palmer MR, Smith DR, Kaftanoglu O (2000) Turkish honeybees: genetic variation and evidence of a fourth lineage of Apis mellifera mtDNA. J Hered 91:42–46
Perrier C, Strange J, Langella O, Shepppard WS, Garnery L (2003) Diversité génétique, introgressions mitochondriales et nucléaires dans une population d’abeilles des Landes de Gascogne. Actes du BRG 4:79–100
Rhymer JM, Simberloff D (1996) Extension by hybridization and introgression. Annu Rev Ecol Syst 27:83–109
Rortais A, Arnold G, Garnery L (2007) Introgressions and structure of the genetic diversity of the black honeybee in France. Proceedings of the second European conference of Apidology Eurbee. Vesely V, Vořechovská M, Titěra Ed. Prague, Czech Republic, 10–16 September 2006. P 59
Rortais A, Arnold G, Garnery L (2009) Utilisation des marqueurs moléculaires pour la mise en place de conservatoires d’abeilles noires (Apis mellifera mellifera). Mémoires de la SEF 8:115–120
Rortais A, Baylac M, Arnold G, Garnery L (2010) A Geometric morphometric tool for the conservation of the black honeybee in Europe—In: Settele J, Penev L, Georgiev T, Grabaum R, Grobelnik V, Hammen V, Klotz S, Kotarac M, Kühn I (Eds), Atlas of biodiversity risk. Pensoft Publishers, Sofia-Moscow. p 180
Ruttner F (1988) Biogeography and taxonomy of honeybees. Spinger-Verlag, Berlin
Ruttner F (1992) Naturgeschichte der Honigbienen. Ehrenwisth Verlag, Münich
Sheppard WS, Meixner D (2003) Apis mellifera pomonella, a new honey bee subspecies from Central Asia. Apidologie 34:367–375
Sheppard WS, Smith DR (2000) Identification of African-derived bees in the Americas: a survey of methods. Ann Entomol Soc Am 93:159–176
Sheppard WS, Arias MC, Grech A, Meixner MD (1997) Apis mellifera ruttneri, a new honey bee subspecies from Malta. Apidologie 28:287–293
Sheppard WS, Rinderer TE, Garnery L, Shimanuki H (1999) Analysis of Africanized honey bee mitochondrial DNA reveals further diversity of origin. Genet Mol Biol 22:73–75
Smith DR (1991) Mitochondrial DNA and honeybee biogeography. In: Smith DR (ed) Diversity in the genus apis. West-view, Boulder, CO
Strange J-P, Garnery L, Sheppard WS (2007) Morphological and molecular characterization of the Landes honeybee (Apis mellifera L.) ecotype for genetic conservation. J Insect Conserv 12:527–537
Susnik S, Kozmus P, Poklukar J, Meglic V (2004) Molecular characterisation of indigenous Apis mellifera carnica in Slovenia. Apidologie 35:623–636
Walsh PS, Metzqer DA, Higuchi R (1991) Chelex 100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. Biotechniques 10:506–513
Acknowledgments
This study was financed by the European Commission (CE no. 1,221/97). We are grateful to the national and regional beekeeping associations which collaborated to the sampling of the honeybees in France and Belgium.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rortais, A., Arnold, G., Alburaki, M. et al. Review of the DraI COI-COII test for the conservation of the black honeybee (Apis mellifera mellifera). Conservation Genet Resour 3, 383–391 (2011). https://doi.org/10.1007/s12686-010-9351-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12686-010-9351-x