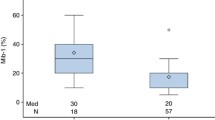
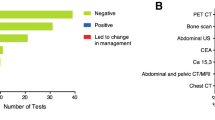
Abstract
Purpose of Review
The American Board of Internal Medicine Foundation’s Choosing Wisely (CW) campaign has promoted a dialogue about avoiding unnecessary medical services since 2012. We review the evidence base, trends in utilization, and implications of CW recommendations relevant to the routine workup of newly diagnosed early stage breast cancer patients.
Recent Findings
There are several CW recommendations addressing routine workup of newly diagnosed breast cancer:
• Do not perform PET, CT, and radionuclide bone scans in the staging of early breast cancer at low risk for metastasis
- These studies are obtained in 10–20% of patients, especially those with Stage IIB disease
• Do not routinely order breast MRI in new breast cancer patients with average risk
- Evidence of long-term benefit to preoperative MRI is lacking, but approximately 1 in 4 breast cancer patients receive one, often leading to additional interventions
• Do not routinely order specialized tumor gene testing in all new breast cancer patients
- Evolving guidelines are expanding eligible populations for genetic assay driven clinical decision-making, with mixed implications for adjuvant chemotherapy utilization
• Do not routinely use sentinel node biopsy in clinically node negative women ≥ 70 years of age with hormone receptor positive invasive breast cancer
- The vast majority of such patients—85–90%— undergo sentinel node biopsy despite questions about the benefit of axillary staging in an elderly population
Summary
CW recommendations offer a useful starting point for the discussion of optimal routine workup for breast cancer. Continued efforts are needed to define the marginal value of different tests and interventions and to develop mechanisms to incentivize and measure adoption of best practices.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Mariotto AB, Robin Yabroff K, Shao Y, Feuer EJ, Brown ML. Projections of the cost of cancer Care in the United States: 2010–2020. J Natl Cancer Inst. 2011;103(2):117–28.
Yabroff KR, Lund J, Kepka D, Mariotto A. Economic burden of cancer in the United States: estimates, projections, and future research. Cancer Epidemiol Biomark Prev. 2011;20(10):2006.
Soneji S, Yang J. New analysis reexamines the value of cancer care in the United States compared to Western Europe. Health Aff. 2015;34(3):390–7. https://doi.org/10.1377/hlthaff.2014.0174.
Shih Y-CT, Ganz PA, Aberle D, Abernethy A, Bekelman J, Brawley O, et al. Delivering high-quality and affordable care throughout the cancer care continuum. J Clin Oncol. 2013;31(32):4151–7. https://doi.org/10.1200/jco.2013.51.0651.
• Schnipper LE, Davidson NE, Wollins DS, Tyne C, Blayney DW, Blum D, et al. American Society of Clinical Oncology statement: a conceptual framework to assess the value of cancer treatment options. J Clin Oncol. 2015;33(23):2563–77. https://doi.org/10.1200/jco.2015.61.6706. A useful framework developed by the ASCO Value in Cancer Care Task Force is presented to facilitate defining value in cancer care and to promote patient-physician shared decision-making.
Kang R, Goodney PP, Wong SL. Importance of cost-effectiveness and value in cancer care and healthcare policy. J Surg Oncol. 2016;114(3):275–80. https://doi.org/10.1002/jso.24331.
Morgan DJ, Dhruva SS, Wright SM, Korenstein D. 2016 update on medical overuse: a systematic review. JAMA Intern Med. 2016;176(11):1687–92. https://doi.org/10.1001/jamainternmed.2016.5381.
Brownlee S, Chalkidou K, Doust J, Elshaug AG, Glasziou P, Heath I, et al. Evidence for overuse of medical services around the world. Lancet. 2017;390(10090):156–68. https://doi.org/10.1016/S0140-6736(16)32585-5.
Breast Cancer Facts & Figures 2015–2016. Atlanta: American Cancer Society, Inc.; 2015.
Schnipper LE, Smith TJ, Raghavan D, Blayney DW, Ganz PA, Mulvey TM, et al. American Society of Clinical Oncology identifies five key opportunities to improve care and reduce costs: the top five list for oncology. J Clin Oncol. 2012;30(14):1715–24. https://doi.org/10.1200/JCO.2012.42.8375.
Carlson RW, Allred DC, Anderson BO, Burstein HJ, Carter WB, Edge SB, et al. Invasive breast cancer. J Natl Compr Cancer Netw. 2011;9(2):136–222.
(NCCN). NCCN. Breast cancer version 2.2017 NCCN clinical practice guidelines in Oncology 2017.
Rakovitch E, Nofech-Mozes S, Hanna W, Baehner FL, Saskin R, Butler SM, et al. A population-based validation study of the DCIS Score predicting recurrence risk in individuals treated by breast-conserving surgery alone. Breast Cancer Res Treat. 2015;152(2):389–98. https://doi.org/10.1007/s10549-015-3464-6.
Pilewskie M, King TA. Magnetic resonance imaging in patients with newly diagnosed breast cancer: a review of the literature. Cancer. 2014;120(14):2080–9. https://doi.org/10.1002/cncr.28700.
Houssami N, Ciatto S, Macaskill P, Lord SJ, Warren RM, Dixon JM, et al. Accuracy and surgical impact of magnetic resonance imaging in breast cancer staging: systematic review and meta-analysis in detection of multifocal and multicentric cancer. J Clin Oncol. 2008;26(19):3248–58. https://doi.org/10.1200/jco.2007.15.2108.
Houssami N, Turner R, Morrow M. Preoperative magnetic resonance imaging in breast cancer: meta-analysis of surgical outcomes. Ann Surg. 2013;257(2):249–55. https://doi.org/10.1097/SLA.0b013e31827a8d17.
Houssami N, Turner R, Macaskill P, Turnbull LW, McCready DR, Tuttle TM, et al. An individual person data meta-analysis of preoperative magnetic resonance imaging and breast cancer recurrence. J Clin Oncol. 2014;32(5):392–401. https://doi.org/10.1200/jco.2013.52.7515.
Turnbull L, Brown S, Harvey I, Olivier C, Drew P, Napp V, et al. Comparative effectiveness of MRI in breast cancer (COMICE) trial: a randomised controlled trial. Lancet. 2010;375(9714):563–71. https://doi.org/10.1016/S0140-6736(09)62070-5.
Peters NH, van Esser S, van den Bosch MA, Storm RK, Plaisier PW, van Dalen T, et al. Preoperative MRI and surgical management in patients with nonpalpable breast cancer: the MONET—randomised controlled trial. Eur J Cancer. 2011;47(6):879–86. https://doi.org/10.1016/j.ejca.2010.11.035.
MRI and mammography before surgery in patients with stage I-II breast cancer. https://ClinicalTrials.gov/show/NCT01805076.
MRI and gene expression in diagnosing patients with ductal breast cancer in situ. https://ClinicalTrials.gov/show/NCT02352883.
Carlson JJ, Roth JA. The impact of the Oncotype Dx breast cancer assay in clinical practice: a systematic review and meta-analysis. Breast Cancer Res Treat. 2013;141(1):13–22. https://doi.org/10.1007/s10549-013-2666-z.
Augustovski F, Soto N, Caporale J, Gonzalez L, Gibbons L, Ciapponi A. Decision-making impact on adjuvant chemotherapy allocation in early node-negative breast cancer with a 21-gene assay: systematic review and meta-analysis. Breast Cancer Res Treat. 2015;152(3):611–25. https://doi.org/10.1007/s10549-015-3483-3.
Paik S, Shak S, Tang G, Kim C, Baker J, Cronin M, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351(27):2817–26. https://doi.org/10.1056/NEJMoa041588.
Dowsett M, Cuzick J, Wale C, Forbes J, Mallon EA, Salter J, et al. Prediction of risk of distant recurrence using the 21-gene recurrence score in node-negative and node-positive postmenopausal patients with breast cancer treated with anastrozole or tamoxifen: a TransATAC study. J Clin Oncol. 2010;28(11):1829–34. https://doi.org/10.1200/jco.2009.24.4798.
• Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, et al. Prospective validation of a 21-gene expression assay in breast cancer. New Engl J Med. 2015;373(21):2005–14. https://doi.org/10.1056/NEJMoa1510764. Interim findings from the Trial Assigning Individualed Options for Treatment (TAILORx) using Oncotype Dx for patients with intermediate clinicopathologic risk. These interim findings related to patients with low-risk score (< 11) who completed endocrine therapy but no adjuvant chemotherapy; 5-year freedom from local-regional or distal recurrance was 98.7%.
• Duffy MJ, Harbeck N, Nap M, Molina R, Nicolini A, Senkus E, et al. Clinical use of biomarkers in breast cancer: updated guidelines from the European Group on Tumor Markers (EGTM). Eur J Cancer. 2017;75:284–98. https://doi.org/10.1016/j.ejca.2017.01.017. Excellent summary of current guidelines for most available biomarker assays across various clinical groups, such as node-negative/positive or HER2-negative/positive patients.
Harris LN, Ismaila N, McShane LM, Andre F, Collyar DE, Gonzalez-Angulo AM, et al. Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016;34(10):1134–50. https://doi.org/10.1200/jco.2015.65.2289.
Krop I, Ismaila N, Andre F, Bast RC, Barlow W, Collyar DE, et al. Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology clinical practice guideline focused update. J Clin Oncol. 2017;35(24):2838–47. https://doi.org/10.1200/jco.2017.74.0472.
Gluz O, Nitz UA, Christgen M, Kates RE, Shak S, Clemens M, et al. West German study group phase III PlanB trial: first prospective outcome data for the 21-gene recurrence score assay and concordance of prognostic markers by central and local pathology assessment. J Clin Oncol. 2016;34(20):2341–9. https://doi.org/10.1200/jco.2015.63.5383.
Cardoso F, van’t Veer LJ, Bogaerts J, Slaets L, Viale G, Delaloge S, et al. 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med. 2016;375(8):717–29. https://doi.org/10.1056/NEJMoa1602253.
Tamoxifen citrate, letrozole, anastrozole, or exemestane with or without chemotherapy in treating patients with invasive RxPONDER breast cancer.
Martelli G, Miceli R, Daidone MG, Vetrella G, Cerrotta AM, Piromalli D, et al. Axillary dissection versus no axillary dissection in elderly patients with breast cancer and no palpable axillary nodes: results after 15 years of follow-up. Ann Surg Oncol. 2011;18(1):125–33. https://doi.org/10.1245/s10434-010-1217-7.
Hughes KS, Schnaper LA, Bellon JR, Cirrincione CT, Berry DA, McCormick B, et al. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013;31(19):2382–7. https://doi.org/10.1200/JCO.2012.45.2615.
Brennan ME, Houssami N. Evaluation of the evidence on staging imaging for detection of asymptomatic distant metastases in newly diagnosed breast cancer. Breast. 21(2):112–23. https://doi.org/10.1016/j.breast.2011.10.005.
Smith-Bindman R, Lipson J, Marcus R, Kim KP, Mahesh M, Gould R, et al. Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med. 2009;169(22):2078–86. https://doi.org/10.1001/archinternmed.2009.427.
Puglisi F, Follador A, Minisini AM, Cardellino GG, Russo S, Andreetta C, et al. Baseline staging tests after a new diagnosis of breast cancer: further evidence of their limited indications. Ann Oncology. 2005;16(2):263–6. https://doi.org/10.1093/annonc/mdi063.
Barcenas CH, Niu J, Valero V, Smith B, Giordano SH. Abstract P3–06-02: the use of imaging and tumor markers in the staging of patients age< 65 years with early-stage breast cancer. Cancer Res 2014;73(24 Supplement):P3-06-2.
Makarov DV, Soulos PR, Gold HT, et al. Regional-level correlations in inappropriate imaging rates for prostate and breast cancers: potential implications for the choosing wisely campaign. JAMA Oncol. 2015;1(2):185–94. https://doi.org/10.1001/jamaoncol.2015.37.
Simos D, Hutton B, Clemons M. Are physicians choosing wisely when imaging for distant metastases in women with operable breast cancer? J Oncol Pract. 2015;11(1):62–8. https://doi.org/10.1200/jop.2014.000125.
Hahn EE, Tang T, Lee JS, Munoz-Plaza C, Adesina JO, Shen E, et al. Use of imaging for staging of early-stage breast cancer in two integrated health care systems: adherence with a choosing wisely recommendation. J Oncol Pract. 2015;11(3):e320–e8. https://doi.org/10.1200/jop.2014.002998.
• Ramsey SD, Fedorenko C, Chauhan R, McGee R, Lyman GH, Kreizenbeck K, et al. Baseline estimates of adherence to American Society of Clinical Oncology/American Board of Internal Medicine Choosing Wisely initiative among patients with cancer enrolled with a large regional commercial health insurer. J Oncol Pract. 2015;11(4):338–43. https://doi.org/10.1200/jop.2014.002717. Retrospective cohort examination of adherence to all five of ASCO’s 2012 Choosing Wisely guidelines linking SEER records to 22,359 patients in Western Washington State. Across all 5 ASCO guidelines, nonadherence for this population amounted to $29 million in additional expenditure.
•• Henry NL, Braun TM, Breslin TM, Gorski DH, Silver SM, Griggs JJ. Variation in the use of advanced imaging at the time of breast cancer diagnosis in a statewide registry. Cancer. 2017;123(15):2975–83. https://doi.org/10.1002/cncr.30674. This retrospective cohort study of 34,078 patients in the Michigan Breast Oncology Quality Initiative offers an assessment of variation in use of advanced imaging studies for newly diagnosed Stage 0-II breast cancer patients. 20.1% of patients in their cohort received a CT, PET, or bone scan in the 90 days following diagnosis. This decreased over the study period except for Stage IIb patients, amounting to a 33% reduction in total imaging costs.
Landercasper J, Bailey L, Berry TS, Buras RR, Degnim AC, Fayanju OM, et al. Measures of appropriateness and value for breast surgeons and their patients: the American Society of Breast Surgeons Choosing Wisely ((R)) initiative. Ann Surg Oncol. 2016;23(10):3112–8. https://doi.org/10.1245/s10434-016-5327-8.
Society of Surgical Oncology. Five things physicians and patients should question. 2016. http://www.surgonc.org/docs/default-source/default-document-library/sso-five-things-physicians-and-patients-should-question-7-11-2016.pdf?sfvrsn=2. Accessed July 22 2017.
Houssami N, Turner RM, Morrow M. Meta-analysis of pre-operative magnetic resonance imaging (MRI) and surgical treatment for breast cancer. Breast Cancer Res Treat. 2017;165(2):273–83. https://doi.org/10.1007/s10549-017-4324-3.
•• Vapiwala N, Hwang WT, Kushner CJ, Schnall MD, Freedman GM, Solin LJ. No impact of breast magnetic resonance imaging on 15-year outcomes in patients with ductal carcinoma in situ or early-stage invasive breast cancer managed with breast conservation therapy. Cancer. 2017;123(8):1324–32. https://doi.org/10.1002/cncr.30479. This retrospective cohort study from the University of Pennsylvania followed 755 women with early stage breast cancer who underwent breast-conserving therapy and radiotherapy between 1992 and 2001. They compared 15-year outcomes between the 28% of patients who had preoperative MRI and those who did not, finding no significant difference in overall survival, freedom from distant metastasis, and contralateral breast cancer.
Killelea BK, Long JB, Chagpar AB, Ma X, Soulos PR, Ross JS, et al. Trends and clinical implications of preoperative breast MRI in Medicare beneficiaries with breast cancer. Breast Cancer Res Treat. 2013;141(1):155–63. https://doi.org/10.1007/s10549-013-2656-1.
Arnaout A, Catley C, Booth CM, et al. Use of preoperative magnetic resonance imaging for breast cancer: a Canadian population-based study. JAMA Oncol. 2015;1(9):1238–50. https://doi.org/10.1001/jamaoncol.2015.3018.
Wernli KJ, DeMartini WB, Ichikawa L, Lehman CD, Onega T, Kerlikowske K, et al. Patterns of breast magnetic resonance imaging use in community practice. JAMA Intern Med. 2014;174(1):125–32. https://doi.org/10.1001/jamainternmed.2013.11963.
Stout NK, Nekhlyudov L, Li L, Malin ES, Ross-Degnan D, Buist DS, et al. Rapid increase in breast magnetic resonance imaging use: trends from 2000 to 2011. JAMA Intern Med. 2014;174(1):114–21. https://doi.org/10.1001/jamainternmed.2013.11958.
• Morrow M, Hawley ST, McLeod MC, Hamilton AS, Ward KC, Katz SJ, et al. Surgeon attitudes and use of MRI in patients newly diagnosed with breast cancer. Ann Surg Oncol. 2017;24(7):1889–96. https://doi.org/10.1245/s10434-017-5840-4. Survey of 377 breast surgeons which identified high and low MRI users via SEER registry and linked this to perceptions about MRI use. A quarter said they would obtain MRI for Stage I disease. High utilizers had significantly higher surgical volume and significantly more misconceptions about MRI benefits.
•• Tan S, David J, Lalonde L, El Khoury M, Labelle M, Younan R, et al. Breast magnetic resonance imaging: are those who need it getting it? Curr Oncol. 2017;24(3):e205–e13. https://doi.org/10.3747/co.24.3441. Excellent example from Quebec of operationalizing current evidence into best practice guidelines for MRI use in breast cancer patients, particularly for high-risk screening and preoperative staging. This involved a consensus meeting of the multisciplinary breast team followed by implementation of a new ordering form based on the consensus guidelines. In the year following implementation, use of MRI for preoperative staging dropped from 18 to 9% of studies even as total MRIs performed increased.
Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S, et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol. 2007;25(33):5287–312. https://doi.org/10.1200/jco.2007.14.2364.
Buyse M, Loi S, van't Veer L, Viale G, Delorenzi M, Glas AM, et al. Validation and clinical utility of a 70-gene prognostic signature for women with node-negative breast cancer. J Natl Cancer Inst. 2006;98(17):1183–92. https://doi.org/10.1093/jnci/djj329.
Oncotype DX for breast cancer. http://www.genomichealth.com/en-US/oncotype_iq_products/oncotype_dx.aspx. Accessed 11 Dec 2017.
Paik S, Tang G, Shak S, Kim C, Baker J, Kim W, et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol. 2006;24(23):3726–34. https://doi.org/10.1200/jco.2005.04.7985.
Albain KS, Barlow WE, Shak S, Hortobagyi GN, Livingston RB, Yeh IT, et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol. 2010;11(1):55–65. https://doi.org/10.1016/s1470-2045(09)70314-6.
Mammaprint 70-gene breast cancer recurrence assay http://www.agendia.com/healthcare-professionals/breast-cancer/mammaprint/. Accessed December 11 2017.
Trosman JR, Van Bebber SL, Phillips KA. Coverage policy development for personalized medicine: private payer perspectives on developing policy for the 21-gene assay. J Oncol Pract. 2010;6(5):238–42. https://doi.org/10.1200/JOP.000075.
Dinan MA, Mi X, Reed SD, Hirsch BR, Lyman GH, Curtis LH. Initial trends in the use of the 21-gene recurrence score assay for patients with breast cancer in the medicare population, 2005-2009. JAMA Oncol. 2015;1(2):158–66. https://doi.org/10.1001/jamaoncol.2015.43.
Hassett MJ, Silver SM, Hughes ME, Blayney DW, Edge SB, Herman JG, et al. Adoption of gene expression profile testing and association with use of chemotherapy among women with breast cancer. J Clin Oncol. 2012;30(18):2218–26. https://doi.org/10.1200/jco.2011.38.5740.
•• Orucevic A, Heidel RE, Bell JL. Utilization and impact of 21-gene recurrence score assay for breast cancer in clinical practice across the United States: lessons learned from the 2010 to 2012 National Cancer Data Base analysis. Breast Cancer Res Treat. 2016;157:427–35. https://doi.org/10.1007/s10549-016-3833-9. Retrospective examination of genetic expression profile use from 2010–2012 in the National Cancer Data Base, which captures ~ 70% of new diagnoses in the USA annually. Oncotype Dx accounted for 94% of ordered tests, with about 1 in 5 ER-positive patients receiving testing.
Ray GT, Mandelblatt J, Habel LA, Ramsey S, Kushi LH, Li Y, et al. Breast cancer multigene testing trends and impact on chemotherapy use. Am J Manag Care. 2016;22(5):e153–e60.
Lucci A, McCall LM, Beitsch PD, Whitworth PW, Reintgen DS, Blumencranz PW, et al. Surgical complications associated with sentinel lymph node dissection (SLND) plus axillary lymph node dissection compared with SLND alone in the American College of Surgeons Oncology Group Trial Z0011. J Clin Oncol. 2007;25(24):3657–63. https://doi.org/10.1200/jco.2006.07.4062.
Rescigno J, Zampell JC, Axelrod D. Patterns of axillary surgical care for breast cancer in the era of sentinel lymph node biopsy. Ann Surg Oncol. 2009;16(3):687–96. https://doi.org/10.1245/s10434-008-0195-5.
Giuliano AE, Ballman KV, McCall L, Beitsch PD, Brennan MB, Kelemen PR, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: the ACOSOG Z0011 (alliance) randomized clinical trial. JAMA. 2017;318(10):918–26. https://doi.org/10.1001/jama.2017.11470.
Chung A, Gangi A, Amersi F, Zhang X, Giuliano A. Not performing a sentinel node biopsy for older patients with early-stage invasive breast cancer. JAMA Surg. 2015;150(7):683–4. https://doi.org/10.1001/jamasurg.2015.0647.
Chagpar AB, McMasters KM, Edwards MJ. Can sentinel node biopsy be avoided in some elderly breast cancer patients? Ann Surg. 2009;249(3):455–60. https://doi.org/10.1097/SLA.0b013e318194d16b.
•• Boughey JC, Haffty BG, Habermann EB, Hoskin TL, Goetz MP. Has the time come to stop surgical staging of the axilla for all women age 70 years or older with hormone receptor-positive breast cancer? Ann Surg Oncol. 2017;24(3):614–7. https://doi.org/10.1245/s10434-016-5740-z. A discussion of the role of surgical staging of the axilla in patients 70 years or older accompanies contemporary estimates of axillary surgery in this population using the National Cancer Data Base (2004–2013 with 86.8% having axillary staging) and Mayo Clinic Rochester (2008–2016 with 91.% having axillary staging). Authors make note of the clinical need to distinguish age and life expectancy.
Chagpar AB, Horowitz N, Sanft T, Wilson LD, Silber A, Killelea B, et al. Does lymph node status influence adjuvant therapy decision-making in women 70 years of age or older with clinically node negative hormone receptor positive breast cancer? Am J Surg. 2017;214(6):1082–8. https://doi.org/10.1016/j.amjsurg.2017.07.036.
Greer LT, Rosman M, Charles Mylander W, Liang W, Buras RR, Chagpar AB. A prediction model for the presence of axillary lymph node involvement in women with invasive breast cancer: a focus on older women. Breast J. 2014;20(2):147–53. https://doi.org/10.1111/tbj.12233.
• Welsh JL, Hoskin TL, Day CN, Habermann EB, Goetz MP, Boughey JC. Predicting nodal positivity in women 70 years of age and older with hormone receptor-positive breast cancer to aid incorporation of a society of surgical oncology choosing wisely guideline into clinical practice. Ann Surg Oncol. 2017;24(10):2881–8. https://doi.org/10.1245/s10434-017-5932-1. A clinical and multivariate predictive model was developed from the National Cancer Data Base for hormone-receptor positive, node-negative women 70 years or older. Those who met a clinical rule for avoiding axillary surgery—grade 1, cT1mi-T1c, or grade 2, cT1mi-T1b—were almost three times less likely to have nodal positivity (7.8%) compared to those not meeting the criteria (22.3%).
Onega T, Tosteson ANA, Weiss J, Alford-Teaster J, Hubbard RA, Henderson LM, et al. Costs of diagnostic and preoperative workup with and without breast MRI in older women with a breast cancer diagnosis. BMC Health Serv Res. 2016;16:76. https://doi.org/10.1186/s12913-016-1317-6.
Roberts MC, Weinberger M, Dusetzina SB, Dinan MA, Reeder-Hayes KE, Carey LA, et al. Racial variation in the uptake of oncotype DX testing for early-stage breast cancer. J Clin Oncol. 2016;34(2):130–8. https://doi.org/10.1200/jco.2015.63.2489.
Simos D, Hutton B, Graham ID, Arnaout A, Caudrelier J-M, Mazzarello S, et al. Patient perceptions and expectations regarding imaging for metastatic disease in early stage breast cancer. SpringerPlus. 2014;3(1):176. https://doi.org/10.1186/2193-1801-3-176.
Pezzin LE, Laud P, Neuner J, Yen TW, Nattinger AB. A statewide controlled trial intervention to reduce use of unproven or ineffective breast cancer care. Contemp Clin Trials. 2016;50:150–6. https://doi.org/10.1016/j.cct.2016.08.005.
Bedford C, Arnaout A, Anas R, Catley C, Clemons M, Earle C, et al. Driving quality improvement with public reporting: use of imaging tests outside guidelines for early-stage breast cancer in Ontario. J Clin Oncol. 2017;35(8_suppl):193. https://doi.org/10.1200/JCO.2017.35.8_suppl.193.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Local-Regional Evaluation and Therapy
Rights and permissions
About this article
Cite this article
Trooboff, S.W., Kang, R., Margenthaler, J. et al. Choosing Wisely: Optimizing Routine Workup for the Newly Diagnosed Breast Cancer Patient. Curr Breast Cancer Rep 10, 62–73 (2018). https://doi.org/10.1007/s12609-018-0268-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12609-018-0268-9