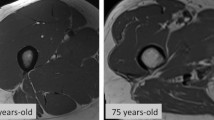
Abstract
Objectives
-
(i)
To investigate serum myostatin (absolute and normalized for total body lean mass (TBLM)) and IGF-1 as biomarkers of frailty and low relative appendicular skeletal muscle mass (RASM) in older adults, and;
-
(ii)
to examine gender differences in the association of serum myostatin and IGF-1 levels with frailty and low RASM.
Design
Cross-sectional study.
Setting
The “Longitudinal Assessment of Biomarkers for characterization of early Sarcopenia and predicting frailty and functional decline in community-dwelling Asian older adults Study” (GERI-LABS) study in Singapore.
Participants
200 subjects aged 50 years and older residing in the community.
Measurements
Frailty was assessed using the modified Fried criteria. Low RASM was defined using cutoffs for height-adjusted appendicular skeletal muscle mass measured by dual-energy X-ray absorptiometry as recommended by the Asian Working Group for Sarcopenia. Comorbidities, cognitive and functional performance, physical activity and nutritional status were assessed. Blood samples collected included serum myostatin, insulin-like growth factor 1 (IGF-1) and markers of inflammation (total white cell count, CRP, IL-6 and TNFaR1). Subjects were classified into 4 groups: Frail/Prefrail with low RASM (Frail/Low RASM), Frail/Prefrail with normal RASM (Frail/Normal RASM), Robust with low RASM (Robust/Low RASM) and Robust with normal RASM (Robust/Normal RASM).
Results
63 (32%) subjects were classified as Frail/Low RASM, 53 (27%) Frail/Normal RASM, 28 (14%) Robust/Low RASM and 56 (28%) Robust/Normal RASM respectively. Frail/Low RASM subjects were older and had lower BMI compared to Frail/Normal RASM and robust subjects. Mean (SE) normalized myostatin levels were higher in Frail/Low RASM compared to Frail/Normal RASM subjects (1.0 (0.04) versus 0.84 (0.05) ng/ml/kg, P=0.01). Median (IQR) IGF-1 level was lower amongst Frail/Low RASM subjects compared to Frail/Normal RASM subjects (102.3, (77.7, 102.5) vs 119.7 (82.7, 146.0) ng/ml, P=0.046). No differences in myostatin or IGF-1 were observed among robust individuals with or without low muscle mass. In adjusted multinomial logistic regression models with Robust/Normal RASM as the reference group, myostatin (P=0.05) and IGF-1 (P=0.043) were associated with Frail/Low RASM status in the whole cohort. When stratified by gender, myostatin was significantly associated with Frail/Low RASM status in men only (P=0.03). In women, serum IGF-1 was associated with Frail/Low RASM status (P=0.046), but not myostatin (P=0.53).
Conclusion
Serum myostatin, normalized for TBLM in men and IGF-1 in women are potential biomarkers for frail individuals with low RASM, and may identify a target group for intervention.
Similar content being viewed by others
References
Rosenberg, I.H., Sarcopenia: origins and clinical relevance. J Nutr, 1997;127(5 Suppl): p. 990S–991S.
Woo, J., J. Leung, A. Sham, et al., Defining sarcopenia in terms of risk of physical limitations: a 5-year follow-up study of 3,153 chinese men and women. J Am Geriatr Soc, 2009;57(12): p. 2224–31.
Arango-Lopera, V.E., P. Arroyo, L.M. Gutierrez-Robledo, et al., Mortality as an adverse outcome of sarcopenia. J Nutr Health Aging, 2013;17(3): p. 259–62.
Clegg, A., J. Young, S. Iliffe, et al., Frailty in elderly people. Lancet, 2013. 381(9868): p. 752–62.
Fried, L.P., C.M. Tangen, J. Walston, et al., Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci, 2001;56(3): p. M146–56.
Landi, F., R. Calvani, M. Cesari, et al., Sarcopenia as the Biological Substrate of Physical Frailty. Clin Geriatr Med, 2015;31(3): p. 367–74.
Yarasheski, K.E., S. Bhasin, I. Sinha-Hikim, et al., Serum myostatin-immunoreactive protein is increased in 60–92 year old women and men with muscle wasting. J Nutr Health Aging, 2002;6(5): p. 343–8.
Leger, B., W. Derave, K. De Bock, et al., Human sarcopenia reveals an increase in SOCS-3 and myostatin and a reduced efficiency of Akt phosphorylation. Rejuvenation Res, 2008;11(1): p. 163–175B.
Peng, L.N., W.J. Lee, L.K. Liu, et al., Healthy community-living older men differ from women in associations between myostatin levels and skeletal muscle mass. J Cachexia Sarcopenia Muscle, 2018.
Arrieta, H., G. Hervas, C. Rezola-Pardo, et al., Serum Myostatin Levels Are Higher in Fitter, More Active, and Non-Frail Long-Term Nursing Home Residents and Increase after a Physical Exercise Intervention. Gerontology, 2018;p. 1–11.
Fife, E., J. Kostka, L. Kroc, et al., Relationship of muscle function to circulating myostatin, follistatin and GDF11 in older women and men. BMC Geriatr, 2018;18(1): p. 200.
Ratkevicius, A., A. Joyson, I. Selmer, et al., Serum concentrations of myostatin and myostatin-interacting proteins do not differ between young and sarcopenic elderly men. J Gerontol A Biol Sci Med Sci, 2011;66(6): p. 620–6.
Roubenoff, R., H. Parise, H.A. Payette, et al., Cytokines, insulin-like growth factor 1, sarcopenia, and mortality in very old community-dwelling men and women: the Framingham Heart Study. Am J Med, 2003;115(6): p. 429–35.
Doi, T., H. Makizako, K. Tsutsumimoto, et al., Association between Insulin-Like Growth Factor-1 and Frailty among Older Adults. J Nutr Health Aging, 2018;22(1): p. 68–72.
Doi, T., H. Shimada, H. Makizako, et al., Insulin-Like Growth Factor-1 Related to Disability Among Older Adults. J Gerontol A Biol Sci Med Sci, 2016;71(6): p. 797–802.
Harris, T.B., D. Kiel, R. Roubenoff, et al., Association of insulin-like growth factor-I with body composition, weight history, and past health behaviors in the very old: the Framingham Heart Study. J Am Geriatr Soc, 1997;45(2): p. 133–9.
Bergen, H.R., 3rd, J.N. Farr, P.M. Vanderboom, et al., Myostatin as a mediator of sarcopenia versus homeostatic regulator of muscle mass: insights using a new mass spectrometry-based assay. Skelet Muscle, 2015;5: p. 21.
Tay, L., Y.Y. Ding, B.P. Leung, et al., Sex-specific differences in risk factors for sarcopenia amongst community-dwelling older adults. Age (Dordr), 2015;37(6): p. 121.
Taekema, D.G., C.H. Ling, G.J. Blauw, et al., Circulating levels of IGF1 are associated with muscle strength in middle-aged- and oldest-old women. Eur J Endocrinol, 2011;164(2): p. 189–96.
Calvani, R., F. Marini, M. Cesari, et al., Biomarkers for physical frailty and sarcopenia: state of the science and future developments. J Cachexia Sarcopenia Muscle, 2015;6(4): p. 278–86.
Sahadevan, S., P.P. Lim, N.J. Tan, et al., Diagnostic performance of two mental status tests in the older chinese: influence of education and age on cut-off values. Int J Geriatr Psychiatry, 2000;15(3): p. 234–41.
Chan, M., Y.P. Lim, A. Ernest, et al., Nutritional assessment in an Asian nursing home and its association with mortality. J Nutr Health Aging, 2010;14(1): p. 23–8.
Mahoney, F.I. and D.W. Barthel, Functional Evaluation: The Barthel Index. Md State Med J, 1965. 14: p. 61–5.
Lawton, M.P. and E.M. Brody, Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist, 1969;9(3): p. 179–86.
Schafer, M.J., E.J. Atkinson, P.M. Vanderboom, et al., Quantification of GDF11 and Myostatin in Human Aging and Cardiovascular Disease. Cell Metab, 2016;23(6): p. 1207–1215.
Chen, L.K., L.K. Liu, J. Woo, et al., Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc, 2014;15(2): p. 95–101.
Schuling, J., R. de Haan, M. Limburg, et al., The Frenchay Activities Index. Assessment of functional status in stroke patients. Stroke, 1993;24(8): p. 1173–7.
Anderson, S.B., A.L. Goldberg, and M. Whitman, Identification of a novel pool of extracellular pro-myostatin in skeletal muscle. J Biol Chem, 2008;283(11): p. 7027–35.
Pirruccello-Straub, M., J. Jackson, S. Wawersik, et al., Blocking extracellular activation of myostatin as a strategy for treating muscle wasting. Sci Rep, 2018;8(1): p. 2292.
White, T.A. and N.K. LeBrasseur, Myostatin and sarcopenia: opportunities and challenges - a mini-review. Gerontology, 2014;60(4): p. 289–93.
Kim, J.K., S.R. Choi, M.J. Choi, et al., Prevalence of and factors associated with sarcopenia in elderly patients with end-stage renal disease. Clin Nutr, 2014;33(1): p. 64–8.
Garcia-Fontana, B., R. Reyes-Garcia, S. Morales-Santana, et al., Relationship between myostatin and irisin in type 2 diabetes mellitus: a compensatory mechanism to an unfavourable metabolic state? Endocrine, 2016;52(1): p. 54–62.
Zugel, M., S. Qiu, R. Laszlo, et al., The role of sex, adiposity, and gonadectomy in the regulation of irisin secretion. Endocrine, 2016;54(1): p. 101–110.
Stitt, T.N., D. Drujan, B.A. Clarke, et al., The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol Cell, 2004;14(3): p. 395–403.
Cruz-Jentoft, A.J., G. Bahat, J. Bauer, et al., Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing, 2019;48(1): p. 16–31.
Woodhouse, L., R. Gandhi, S.J. Warden, et al., A Phase 2 Randomized Study Investigating the Efficacy and Safety of Myostatin Antibody LY2495655 versus Placebo in Patients Undergoing Elective Total Hip Arthroplasty. J Frailty Aging, 2016;5(1): p. 62–70.
Cawthon, P.M., E.S. Orwoll, K.E. Peters, et al., Strong Relation between Muscle Mass Determined by D3-creatine Dilution, Physical Performance and Incidence of Falls and Mobility Limitations in a Prospective Cohort of Older Men. J Gerontol A Biol Sci Med Sci, 2018.
Dent, E., J.E. Morley, A.J. Cruz-Jentoft, et al., International Clinical Practice Guidelines for Sarcopenia (ICFSR): Screening, Diagnosis and Management. J Nutr Health Aging, 2018;22(10): p. 1148–1161.
Acknowledgements
We would like to thank all participants who contributed to this study. This study was supported by the Lee Foundation Grant 2013.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards: Ethics approval for the study was obtained from by the Domain Specific Review Board of the National Healthcare Group and informed written consent was obtained from the participants.
Conflict of interest: The authors have no conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Chew, J., Tay, L., Lim, J.P. et al. Serum Myostatin and IGF-1 as Gender-Specific Biomarkers of Frailty and Low Muscle Mass in Community-Dwelling Older Adults. J Nutr Health Aging 23, 979–986 (2019). https://doi.org/10.1007/s12603-019-1255-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-019-1255-1