Abstract
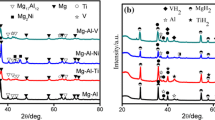
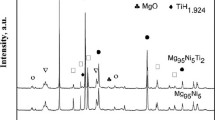
The Mg–Al hydrogen storage alloy was successfully prepared by combustion synthesis (CS) method. The formation of alloy phases during the CS process was studied using X-ray diffraction (XRD), scanning electron microscope (SEM), and differential scanning calorimetry (DSC). When the time increases from 0, 0.5, 1.0 to 2.0 h at 733 K, the products are Mg and Al; Mg2Al3, Mg and Al; Mg17Al12, Mg2Al3; and Mg; and eventually only Mg17Al12, respectively. Combined with three peaks in the DSC traces, it is concluded that the formation of Mg17Al12 during the CS includes three processes, namely, the formation of Mg2Al3 first; then the unsaturated solid solution, Mg17Al12; and finally the complete Mg17Al12 alloy. The formation of Mg2Al3 prior to Mg17Al12 in this work is different from those prepared by mechanical alloying. This is thought to be related to the instant high temperature during the thermal explosion of CS.




Similar content being viewed by others
References
Wang XL, Tu JP, Zhang PL, Zhang XB, Chen CP, Zhao XB. Hydrogenation properties of ball-milled MgH2-10 wt% Mg17Al12 composite. Int J Hydrogen Energy. 2007;32(15):3406.
Milanese C, Girella A, Bruni G, Berbenni V, Cofrancesco P, Marini A. Hydrogen storage in magnesium-metal mixtures: reversibility, kinetic aspects and phase analysis. J Alloy Compd. 2008;465(1–2):396.
Aguey-Zinsou KF, Ares-Femandez JR. Hydrogen in magnesium: new perspectives toward functional stores. Energy Environ Sci. 2010;3(5):526.
Jurczyk M, Nowak M, Szakek A, Jezierski A. Hydrogen storage by Mg-based nanocomposites. Int J Hydrogen Energy. 2012;37(4):3652.
Andreasen A. Hydrogenation properties of Mg–Al alloys. Int J Hydrogen Energy. 2008;33(24):7489.
Yabe H, Kuji T. Thermal stability and hydrogen absorption/desorption properties of Mg17Al12 produced by bulk mechanical alloying. J Alloy Compd. 2007;433(1–2):241.
Crivello JC, Nobuki T, Kato S, Abe M, Kuji T. Hydrogen absorption properties of the γ-Mg17Al12 phase and its Al-richer domain. J Alloy Compd. 2007;446:157.
Suwarno H, Adi WA, Insani A. The mechanism of Mg2Al3 formation by mechanism alloying. Atom Indonesia. 2009;35(1):11.
Suwarno H. A study on preparation and hydriding of β-Mg2Al3 and γ-Mg17Al12. J Tek Bhn Nukl. 2009;5(2):53.
Zanotti C, Giuliani P, Maglia F. Combustion synthesis of Co–Al and Ni–Al systems under reduced gravity. Intermetallics. 2006;14(2):213.
Godlewska E, Mars K, Mania R, Zimowski S. Combustion synthesis of Mg2Si. Intermetallics. 2011;19(12):1983.
Biswas A, Roy SK, Gurumurthy KR, Prabhu N, Banerjee S. A study of self-propagating high-temperature synthesis of NiAl in thermal explosion mode. Acta Mater. 2002;50:757.
Sato M, Kuji T. Thermodynamic consideration on multi-step hydrogenation of Mg17Al12 assisted by phase separation. Mater Trans. 2011;52(9):1773.
Li LQ, Saita I, Akiyama T. Intermediate products of hydriding combustion synthesis of Mg2NiH4 studied by optical microscopy and field-emission scanning electron microscopy. Intermetallics. 2005;13(6):6628.
Li LQ, Saita I, Saito K, Akiyama T. Effect of synthesis temperature on the purity of product in hydriding combustion synthesis of Mg2NiH4. J Alloy Compd. 2002;345(1–2):189.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 51071085 and 51171079), Specialized Research Fund for the Doctoral Program of High Education (No. 20093221110008) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, YL., Lu, Y., Zhu, YF. et al. Phase transformation and thermal analysis during combustion synthesis of Mg17Al12 alloy. Rare Met. 33, 37–40 (2014). https://doi.org/10.1007/s12598-013-0192-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-013-0192-0