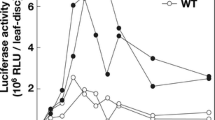
Abstract
Nuclear inclusion protein b (NIb) genes of three Potato virus Y isolates PVY-SD1 (O strain), PVY-SD4 (N strain), PVY-SD5 (NTN strain), and Tobacco etch virus isolate TEV-SD1 in Shandong Province were cloned and sequenced. Sequence analysis showed that the sequence homology of the entire NIb gene among the four viruses ranged from 65% to 95%. Hairpin RNA (hpRNA) constructs were designed based on five conserved regions derived from PVY-SD1 and introduced into tobacco plants. After asexual propagation, the transgenic plants were analyzed for resistance to PVY-SD1, PVY-SD4, PVY-SD5, and TEV-SD1. We obtained resistance ratios of 26.2%, 22.7%, 36.4%, 20.3%, and 21.7% to PVY-SD1. When inoculated with the PVY-SD5 virus, the transgenic plants had resistance ratios ranging from 2.4% to 15.9%, but no resistance at all to the other viruses, PVY-SD4 and TEV-SD1. No correlation was found between resistance of transgenic plants and the transgene copy numbers. Northern blot and small interfering RNA (siRNA) analysis demonstrated that the resistance was attributable to RNA silencing. Genetic analysis demonstrated that virus resistance was stably inherited in progeny T1 and T2. These results indicate that the siRNA molecules against conserved regions can confer virus resistance but are restricted to viruses with more than 90% sequence homology.





Similar content being viewed by others
References
Abel PP, Nelson RS, Hoffmann N, De B, Rogers SG, Fraley RT, Beachy RN (1986) Delay of disease development in transgenic plants that express the tobacco mosaic virus coat protein gene. Science 232:738–743
Amarzguioui M, Prydz H (2004) An algorithm for selection of functional siRNA sequences. Biochem Biophys Res Commun 316:1050–1058
Balmori-Melian E, MacDiarmid RM, Beck DL, Gardner RC, Forster RLS (2002) Sequence-, tissue-, and delivery-specific targeting of RNA during post-transcriptional gene silencing. Mol Plant-Microbe Interact 15:753–763
Bau HJ, Cheng YH, Yu TA, Yang JS, Yeh SD (2003) Broad-spectrum resistance to different geographic strains of Papaya ringspot virus in coat protein gene transgenic papaya. Phytopathology 93:112–120
Baulcombe DC (1996) Mechanisms of pathogen-derived resistance to viruses in transgenic plants. Plant Cell 8:1833–1844
Baulcombe DC (2002) RNA silencing. Curr Biol 12:R82–R84
Beachy RN (1997) Mechanisms and applications of pathogen-derived resistance in transgenic plants. Curr Opin Biotech 8:215–220
Bernstein E, Caudy AA, Hammond SM, Hannon GJ (2001) Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 409:363–366
Bucher E, Lohuis D, Pieter M, van Poppel JA, Geerts-Dimitriadou C, Goldbach R, Prins M (2006) Multiple virus resistance at a high frequency using a single transgene construct. J Gen Virol 87:3697–3701
Carr JP, Gal-On A, Palukaitis P, Zaitlin M (1994) Replicase-mediated resistance to cucumber mosaic virus in transgenic plants involves suppression of both virus replication in the inoculated leaves and long-distance movement. Virology 199:439–447
Dougherty WG, Parks TD (1995) Transgenes and gene suppression: telling us something new? Curr Op Cell Biol 7:399–405
Dougherty WG, Lindbo JA, Smith HA, Parks TD, Swaney S, Proebsting WM (1994) RNA-mediated resistance in transgenic plants: exploitation of a cellular pathway possibly involved in RNA degradation. Mol Plant-Microbe Interact 7:544–552
Elbashir SM, Lendeckel W, Tuschl T (2001) RNA interference is mediated by 21- and 22-nucleotide RNAs. Gene De 15:188–200
Fuchs M, McFerson JR, Tricoli DM, McMaster JR, Deng RZ, Boeshore ML, Reynolds JF, Russell PF, Quemada HD, Gonsalves D (1997) Cantaloupe line CZW-30 containing coat protein genes of cucumber mosaic virus, zucchini yellow mosaic virus, and watermelon mosaic virus-2 is resistance to these three virus in the field. Mol Breed 3:279–290
Glais L, Tribodet M, Kerlan C (2002) Genomic variability in Potato potyvirus Y (PVY): evidence that PVYNW and PVYNTN variants are single to multiple recombinants between PVYO and PVYN isolates. Arch Viroy 147:363–378
Golemboski D, Lomonossoff GP, Zaitlin M (1990) Plants transformed with a tobacco mosaic virus nonstructural gene sequence are resistant to the virus. Proc Natl Acad Sci 87:6311–6315
Gredell JA, Berger AK, Walton SP (2008) Impact of target mRNA structure on siRNA silencing efficiency: a large-scale study. Biotechnol Bioeng 100:744–755
Guo XQ, Liu SE, Zhu CX, Song YZ, Meng XB, Zheng CC, Wen FJ (2001) RNA mediated viral resistance against Potato virus Y (PVY) in transgenic tobacco plants. Acta Phytopathol Sin 31:349–356
Hamilton AJ, Baulcombe DC (1999) A species of small antisense RNA in posttranscriptional gene silencing in plants. Science 286:950–952
Hammond SM, Bernstein E, Beach D, Hannon GJ (2000) An RNA-directed nuclease mediates post-transcriptional gene silencing in Drosophila cells. Nature 404:293–296
Helliwell C, Waterhouse P (2003) Constructs and methods for high-throughput gene silencing in plants. Methods 30:289–295
Hohjoh H (2004) Enhancement of RNAi activity by improved siRNA duplexes. FEBS Lett 557:193–198
Holen T, Amarzguioui M, Wiiger MT, Babaie E, Prydz H (2002) Positional effects of short interfering RNAs targeting the human coagulation trigger tissue factor. Nucleic Acids Res 30:1757–1766
Jan FJ, Fagoaga C, Pang SZ, Gonsalves D (2000) A single chimeric transgene derived from two distinct viruses confers multi-virus resistance in transgenic plants through homology-dependent gene silencing. J Gen Virol 81:2103–2109
Jones AL, Johansen IE, Bean SJ, Bach I, Maule AJ (1998) Specificity of resistance to pea seed-borne mosaic potyvirus in transgenic peas expressing the viral replicase(Nlb) gene. J Gen Virol 79:3129–3137
Kamer G, Argos P (1984) Primary structural comparison of RNA-dependent polymerases from plant, animal and bacterial viruses. Nucleic Acid Res 12:7269–7282
Kaniewsli W, Lawson C, Sammoms B, Haley L, Hart J, Delannay X, Tumer NE (1990) Field resistance of transgenic Russet Burbank potato to effects of infection by potato virus X and potato virus Y. Bio-Technology 8:750–754
Kouassi NK, Chen L, Sire C, Bangratz-Reyser M, Beachy RN, Fauquet CM, Brugidou C (2006) Expression of rice yellow mottle virus coat protein enhances virus infection in transgenic plants. Arch Virol 151:2111–2122
Krubphachaya P, Jurícek M, Kertbundit S (2007) Induction of RNA-mediated resistance to papaya ringspot virus type W. J Biochem Mol Biol 40:404–411
Li P, Song YZ, Liu XL, Zhu CX, Wen FJ (2007) Study of virus resistance mediated by inverted repeats derive from 5′and 3′ends of coat protein gene of Potato virus Y. Acta Phytopathol Sin 37:69–72
Liao JY, Yin JQ, Chen F, Liu TG, Yue JC (2008) A study on the fundamental factors determining the efficacy of siRNAs with high C/G contents. Cell Mol Biol Lett 13:283–302
Lindbo JA, Dougherty WG (1992) Untranslatable transcripts of the tobacco etch virus coat protein gene sequence can interfere with tobacco etch virus replication in transgenic plants and protoplasts. Virology 189:725–733
Liu M, Chen W, Ni Z, Yan W, Fei L, Jiao Y, Zhang J, Du Q, Wei X, Chen J, Liu Y, Zheng Z (2005) Cross-inhibition to heterologous foot-and-mouth disease virus infection induced by RNA interference targeting the conserved regions of viral genome. Virology 336:51–59
Lomonossoff GP (1995) Pathogen-derived resistance to plant viruses. Annu Rev of Phytopathol 33:323–343
McManus MT, Sharp PA (2002) Gene silencing in mammals by small interfering RNAs. Nature Rev Genet 3:737–747
Molnár A, Csorba T, Lakatos L, Várallyay E, Lacomme C, Burgyán J (2005) Plant virus-derived small interfering RNAs originate predominantly from highly structured single-stranded viral RNAs. J Virol 79:7812–7818
Moreno M, Bernal JJ, Jimenez I, Rodriguez-Cerezo E (1998) Resistance in plants transformed with the P1 or P3 gene of tobacco vein mottling potyvirus. J Gen Virol 79:2819–2827
Mueller E, Gilbert J, Davenport G, Brigneti G, Baulcombe DC (1995) Homology-dependent resistance: Transgenic virus resistance in plants related to homology-dependent gene silencing. Plant J 7:1001–1013
Nomura K, Ohshima K, Anai T, Uekusa H, Kita N (2004) RNA silencing of the introduced coat protein gene of turnip mosaic virus confers broad-spectrum resistance in transgenic arabidopsis. Phytopathology 94:730–736
Nykänen A, Haley B, Zamore PD (2001) ATP requirements and small interfering RNA structure in the RNA interference pathway. Cell 107:309–321
O’Brien L (2007) Inhibition of multiple strains of Venezuelan equine encephalitis virus by a pool of four short interfering RNAs. Antivir Res 75:20–29
Poch O, Sauvagey L, Delarue M, Tordo N (1989) Identification of four conserved motifs among the RNA-dependent polymerase encoding elements. EMBO J 8:3867–3874
Prins M, de Haan P, Luyten R, Van Veller M, Van Grinsven MQ, Goldbach R (1995) Broad resistance to tospoviruses in transgenic tobacco plants expressing three tospoviral nucleoprotein sequence. Mol Plant-Microbe Interact 8:85–91
Pusch O, Boden D, Silbermann R, Lee F, Tucker L, Ramratnam B (2003) Nucleotide sequence homology requirements of HIV-1-specific short hairpin RNA. Nucleic Acids Res 31:6444–6449
Sambrook J, Fristsch EF, Maniatis T (1992) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Sanford JC, Johnson SA (1985) The concept of parasite-derived resistance: deriving resistance genes from the parasite own genome. J Theor Biol 113:395–405
Sonoda S, Mori M, Nishiguchi M (1999) Homology-dependent virus resistance in transgenic plants with the coat protein gene of sweet potato feathery mottle potyvirus: target specificity and transgene methylation. Phytopathology 89:385–391
Westerhout EM, Berkhout B (2007) A systematic analysis of the effect of target RNA structure on RNA interference. Nucleic Acids Res 35:4322–4330
Zamore PD, Tuschl T, Sharp PA, Bartel DP (2000) RNAi: Double-stranded RNA directs the ATP-dependent cleavage of mRNA at 21 to 23 nucleotide intervals. Cell 101:25–33
Zhou XP, Li DB (2000) Genetically engineering resistance to viruses and environmental risk assessment of release of transgenic plants. Chinese Bull Sci 12:4–6
Zhu JH, Zhu CX, Wen FJ, Song YZ (2004) Comparison of resistance to Potato virus Y mediated by direct and inverted repeats of the Coat protein gene segments in transgenic tobacco plants. Acta Phytopathol Sin 34:133–140
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (Nos. 30771408 and 30500327).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Li Xu and YunZhi Song have equal contribution.
Rights and permissions
About this article
Cite this article
Xu, L., Song, Y., Zhu, J. et al. Conserved Sequences of Replicase Gene-Mediated Resistance to Potyvirus through RNA Silencing. J. Plant Biol. 52, 550–559 (2009). https://doi.org/10.1007/s12374-009-9071-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-009-9071-5