Abstract
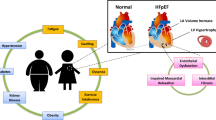
Cardiac left ventricular (LV) remodeling is the final common pathway of most primary cardiovascular diseases that manifest clinically as heart failure (HF). The more advanced the systolic HF and LV dysfunction, the worse the prognosis. The knowledge of the molecular, cellular, and neurohormonal mechanisms that lead to myocardial dysfunction and symptomatic HF has expanded rapidly and has allowed sophisticated approaches to understanding and management of the disease. New therapeutic targets for pharmacologic intervention in HF have also been identified through discovery of novel cellular and molecular components of membrane-bound receptor-mediated intracellular signal transduction cascades. Despite all advances, however, the prognosis of systolic HF has remained poor in general. This is, at least in part, related to the (1) relatively late institution of treatment due to reliance on gross functional and structural abnormalities that define the “heart failure phenotype” clinically; (2) remarkable genetic-based interindividual variations in the contribution of each of the many molecular components of cardiac remodeling; and (3) inability to monitor the activity of individual pathways to cardiac remodeling in order to estimate the potential benefits of pharmacologic agents, monitor the need for dose titration, and minimize side effects. Imaging of the recognized ultrastructural components of cardiac remodeling can allow redefinition of heart failure based on its “molecular phenotype,” and provide a guide to implementation of “personalized” and “evidence-based” evaluation, treatment, and longitudinal monitoring of the disease beyond what is currently available through randomized controlled clinical trials.




Similar content being viewed by others
Abbreviations
- CAD:
-
Coronary artery disease
- CMR:
-
Cardiac magnetic resonance
- CT:
-
Computed tomography
- FDG:
-
2-deoxy-2-(18F)fluoro-d-glucose
- HF:
-
Heart failure
- LV:
-
Left ventricle (ventricular)
- MRI:
-
Magnetic resonance imaging
- MRS:
-
Magnetic resonance spectroscopy
- PET:
-
Positron emission tomography
- SPECT:
-
Single-photon emission computed tomography
References
Kehat I, Molkentin JD. Molecular pathways underlying cardiac remodeling during pathophysiological stimulation. Circulation 2010;122:2727-35.
Konstam MA, Kramer DG, Patel AR, Maron MS, Udelson JE. Left ventricular remodeling in heart failure: Current concepts in clinical significance and assessment. JACC Cardiovasc Imaging 2011;4:98-108.
Saito T, Asai K, Sato S, Takano H, Mizuno K, Shimizu W. Ultrastructural features of cardiomyocytes in dilated cardiomyopathy with initially decompensated heart failure as a predictor of prognosis. Eur Heart J 2015;36:724-32.
Schmitter D, Cotter G, Voors AA. Clinical use of novel biomarkers in heart failure: Towards personalized medicine. Heart Fail Rev 2014;19:369-81.
Creemers EE, Wilde AA, Pinto YM. Heart failure: Advances through genomics. Nat Rev Genet 2011;12:357-62.
Parry HM, Doney ASF, Palmer CNA, Lang CC. State of play of pharmacogenetics and personalized medicine in heart failure. Cardiovasc Ther 2013;31:315-22.
Pleger ST, Brinks H, Ritterhoff J, Raake P, Koch WJ, Katus HA, Most P. Heart failure gene therapy: The path to clinical practice. Circ Res 2013;113:792-809.
Shirani J, Narula J, Eckelman WC, Narula N, Dilsizian V. Early imaging in heart failure: Exploring novel molecular targets. J Nucl Cardiol 2007;14:100-10.
Shirani J, Dilsizian V. Imaging left ventricular remodeling: Targeting the neurohumoral axis. Nat Clin Pract Cardiovasc Med 2008;5:S57-62.
Shirani J, Dilsizian V. Molecular imaging targets of cardiac remodeling. Curr Cardiol Rep 2009;11:148-54.
Harvey PA, Leinwand LA. The cell biology of disease: Cellular mechanisms of cardiomyopathy. J Cell Biol 2011;194:355-65.
Burchfield JS, Xie M, Hill JA. Pathological ventricular remodeling: Mechanisms: Part 1 of 2. Circulation 2013;128:388-400.
Xie M, Burchfield JS, Hill JA. Pathological ventricular remodeling: Mechanisms: Part 2 of 2. Circulation 2013;128:1021-30.
Konstantinidis K, Whelan RS, Kitsis RN. Mechanisms of cell death in heart disease. Arterioscler Thromb Vasc Biol 2012;32:1552-62.
Spinale FG, Zile MR. Integrating the myocardial matrix into heart failure recognition and management. Circ Res 2013;113:725-38.
Ma Y, de Castro Brás LE, Toba H, Iyer RP, Hall ME, Winniford MD, et al. Myofibroblasts and the extracellular matrix network in post-myocardial infarction cardiac remodeling. Pflugers Arch 2014;466:1113-27.
Zhang DY, Anderson AS. The sympathetic nervous system and heart failure. Cardiol Clin 2014;32:33-45.
Sayer G, Bhat G. The renin-angiotensin-aldosterone system and heart failure. Cardiol Clin 2014;32:21-32.
Dilsizian V, Bacharach SL, Beanlands SR, Bergmann SR, Delbeke D, Dorbala S, Gropler RJ et al. ASNC/SNMMI imaging guidelines for nuclear cardiology procedures: PET myocardial perfusion and metabolism clinical imaging. J Nucl Cardiol 2016 (September issue, in press).
Dilsizian V. Transition from SPECT to PET myocardial perfusion imaging: A desirable change in nuclear cardiology to approach perfection. J Nucl Cardiol 2016;23:337-8.
Nekolla SG, Rischpler C, Batrice A, Schwaiger M. Cardiac PET/MRI. Curr Cardiovasc Imaging Rep 2013;6:158-68.
Smith MF, Dilsizian V. Display of 3D multimodality cardiac images with 2D polar maps: simplicity can be a virtue. JACC Cardiovasc Imaging 2016;9:712-4.
Shirani J, Lee J, Quigg RJ, Pick R, Bacharach SL, Dilsizian V. Relation of thallium uptake to morphologic features of chronic ischemic heart disease: Evidence for myocardial remodeling in non-infarct myocardium. J Am Coll Cardiol 2001;38:84-90.
Gould KL, Johnson N, Bateman T, Beanlands R, Bengel F, Bober R, et al. Anatomic versus physiologic assessment of coronary artery disease: Guiding management decisions using positron-emission tomography (PET) as a physiologic tool. J Am Coll Cardiol 2013;62:1639-53.
Gewirtz H, Dilsizian V. Integration of quantitative PET absolute myocardial blood flow in the clinical management of coronary artery disease. Circulation 2016;133:2180-96.
Schindler TH, Dilsizian V. PET-determined hyperemic myocardial blood flow: Further progress to clinical application. J Am Coll Cardiol 2014;64:1476-8.
Valenta I, Dilsizian V, Querciolo A, Ruddy TD, Schindler TH. Quantitative PET/CT measures of myocardial flow reserve and atherosclerosis for cardiac risk assessment and predicting adverse patient outcome. Curr Cardiol Rep 2013;15:344-54.
Schindler TH, Querciolo A, Valenta I, Ambrosio G, Wahl RL, Dilsizian V. quantitative assessment of myocardial blood flow: Clinical and research applications. Semin Nucl Med 2014;44:274-93.
Dilsizian V, Narula J. Capturing maximal coronary vasodilation for myocardial perfusion imaging: Is timing everything? JACC Cardiovasc Imaging 2015;8:499-500.
Ziadi MC, deKemp RA, Williams K, Guo A, Renaud JM, Chow BJW, et al. Does quantification of myocardial flow reserve using rubidium-82 positron emission tomography facilitate detection of multivessel coronary artery disease? J Nucl Cardiol 2012;19:670-80.
Schindler TH, Schelbert HR, Quercioli A, Dilsizian V. Cardiac PET imaging for the detection and monitoring of coronary artery disease and microvascular health. JACC Cardiovasc Imaging 2010;3:623-40.
Motwani M, Maredia N, Fairbairn TA, Kozerke S, Greenwood JP, Plein S. Assessment of ischaemic burden in angiographic three-vessel coronary artery disease with high-resolution myocardial perfusion cardiovascular magnetic resonance imaging. Eur Heart J: Cardiovasc Imaging 2014;15:701-8.
Taqueti VR, Everett BM, Murthy VL, Gaber M, Foster CR, Hainer J, et al. Interaction of impaired coronary flow reserve and cardiomyocyte injury on adverse cardiovascular outcomes in patients without overt coronary artery disease. Circulation 2015;131:528-35.
Herzog BA, Husmann L, Valenta I, Gaemperli O, Siegrist PT, Tay FM, et al. Long-term prognostic value of 13N-ammonia myocardial perfusion positron emission tomography added value of coronary flow reserve. J Am Coll Cardiol 2009;54:150-6.
Ziadi MC, deKemp RA, Williams KA, Guo A, Chow BJ, Renaud JM, et al. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol 2011;58:740-8.
Flachskampf FA, Dilsizian V. Leaning heavily on PET myocardial perfusion for prognosis. JACC Cardiovac Imaging 2014;7:288-91.
Bennett SK, Smith MF, Gottlieb SS, Fisher ML, Bacharach SL, Dilsizian V. Effect of metoprolol on absolute myocardial blood flow in patients with heart failure secondary to ischemic or nonischemic cardiomyopathy. Am J Cardiol 2002;89:1431-4.
Slart RH, Tio RA, van der Vleuten PA, Willems TP, Lubbers DD, Dierckx RA, van Veldhuisen DJ. Myocardial perfusion reserve and contractile pattern after beta-blocker therapy in patients with idiopathic dilated cardiomyopathy. J Nucl Cardiol 2010;17:479-85.
Dilsizian V. Metabolic adaptation to myocardial ischemia: The role of fatty acid imaging. J Nucl Cardiol 2007;14:S97-9.
Fink JC, Lodge MA, Smith MF, Hinduja A, Brown J, Dinits-Pensy MY, et al. Pre-clinical myocardial metabolic alterations in chronic kidney disease. Cardiology 2010;116:160-7.
Taegtmeyer H, Dilsizian V. Imaging myocardial metabolism and ischemic memory. Nat Clin Pract Cardiovasc Med. 2008;5:S42-8.
Zhang L, Lu Y, Jiang H, Zhang L, Sun A, Zou Y, et al. Additional use of trimetazidine in patients with chronic heart failure: A meta-analysis. J Am Coll Cardiol 2012;59:913-22.
Tillisch J, Brunken R, Marshall R, Schwaiger M, Mandelkern M, Phelps M, et al. Reversibility of cardiac wall-motion abnormalities predicted by positron tomography. N Engl J Med 1986;314:884-8.
Srinivasan G, Kitsiou AN, Bacharach SL, Bartlett ML, Miller-Davis C, Dilsizian V. 18F-fluorodeoxyglucose single photon emission computed tomography: Can it replace PET and thallium SPECT for the assessment of myocardial viability? Circulation 1998;97:843-50.
Kitsiou AN, Bacharach SL, Bartlett ML, Srinivasan G, Summers RM, Quyyumi AA, et al. 13N-Ammonia myocardial blood flow and uptake: Relation to functional outcome of asynergic regions after revascularization. J Am Coll Cardiol 1999;33:678-86.
Sarrazin JF, Philippon F, Tessier M, Guimond J, Molin F, Champagne J, et al. Usefulness of fluorine-18 positron emission tomography/computed tomography for identification of cardiovascular implantable electronic device infections. J Am Coll Cardiol 2012;59:1616-25.
Saby L, Laas O, Habib G, Cammilleri S, Mancini J, Tessonnier L, et al. Positron emission tomography/computed tomography for diagnosis of prosthetic valve endocarditis: Increased valvular 18F-fluorodeoxyglucose uptake as a novel major criterion. J Am Coll Cardiol 2013;61:2374-82.
Dilsizian V, Achenbach S, Narula J. On adding versus selecting imaging modalities for incremental diagnosis: A case-study of 18F-fluorodeoxyglucose PET/CT in prosthetic valve endocarditis. JACC Cardiovasc Imaging 2013;6:1020-1.
Chen W, Kim J, Molchanova-Cook OP, Dilsizian V. The potential of FDG PET/CT for early diagnosis of cardiac device and prosthetic valve infection before morphologic damages ensue. Curr Cardiol Rep 2014;16:459.
Kim J, Feller ED, Chen W, Dilsizian V. FDG PET-CT imaging for LVAD associated infections. JACC Cardiovasc Imaging 2014;7:839-42.
Dilsizian V. 2015 SNMMI highlights lecture: Cardiovascular nuclear and molecular imaging. J Nucl Med 2015;56:13N-9N.
Rosenbaum D, Millon A, Fayad ZA. Molecular imaging in atherosclerosis: FDG PET. Curr Atheroscler Rep 2012;14:429-37.
Herrero P, Sharp TL, Dence C, Haraden BM, Gropler RJ. Comparison of 1-(11)C-glucose and (18)F-FDG for quantifying myocardial glucose use with PET. J Nucl Med 2002;43:1530-41.
Herrero P, Kisrieva-Ware Z, Dence CS, Patterson B, Coggan AR, Han DH, et al. PET measurements of myocardial glucose metabolism with 1-11C-glucose and kinetic modeling. J Nucl Med 2007;48:955-64.
Dávila-Román VG, Vedala G, Vedala G, Herrero P, de las Fuentes L, Rogers JG, et al. Altered myocardial fatty acid and glucose metabolism in idiopathic dilated cardiomyopathy. J Am Coll Cardiol 2002;40:271-7.
Dilsizian V, Taillefer R. Journey in evolution of nuclear cardiology: Will there be another quantum leap with the f-18 labeled myocardial perfusion tracers? JACC Cardiovasc Imaging 2012;5:1269-84.
Taylor M, Wallhaus TR, Degrado TR, Russell DC, Stanko P, Nickles RJ, et al. An evaluation of myocardial fatty acid and glucose uptake using PET with [18F]fluoro-6-thia-heptadecanoic acid and [18F]FDG in patients with congestive heart failure. J Nucl Med 2001;42:55-62.
DeGrado TR, Kitapci MT, Wang S, Ying J, Lopaschuk GD. Validation of 18F-fluoro-4-thia-palmitate as a PET probe for myocardial fatty acid oxidation: effects of hypoxia and composition of exogenous fatty acids. J Nucl Med 2006;47:173-81.
Shoup TM, Elmaleh DR, Bonab AA, Fischman AJ. Evaluation of trans-9-18F-fluoro-3,4-methyleneheptadecanoic acid as a PET tracer for myocardial fatty acid imaging. J Nucl Med 2005;46:297-304.
DeGrado TR, Bhattacharyya F, Pandey MK, Belanger AP, Wang S. Synthesis and preliminary evaluation of 18-18F-fluoro-4-thia-oleate as a PET probe of fatty acid oxidation. J Nucl Med 2010;51:1310-7.
Tu Z, Li S, Sharp TL, Herrero P, Dence CS, Gropler RJ, et al. Synthesis and evaluation of 15-(4-(2-[18F]Fluoroethoxy)phenyl)pentadecanoic acid: A potential PET tracer for studying myocardial fatty acid metabolism. Bioconjug Chem 2010;21:2313-9.
Lee H, Chen DL, Rothfuss JM, Welch MJ, Gropler RJ, Mach RH. Synthesis and evaluation of 18F-labeled PPARγ antagonists. Nucl Med Biol 2012;39:77-87.
Dilsizian V, Bateman TM, Bergmann SR, Des Prez R, Magram MY, Goodbody AE, et al. Metabolic imaging with beta-methyl-p-[(123)I]-iodophenyl-pentadecanoic acid identifies ischemic memory after demand ischemia. Circulation 2005;112:2169-74.
Kontos MC, Dilsizian V, Weiland F, DePuey G, Mahmarian JJ, Iskandrian AE, et al. Iodofiltic Acid I 123 (BMIPP) fatty acid imaging improves initial diagnosis in emergency department patients with suspected acute coronary syndromes: A multicenter trial. J Am Coll Cardiol 2010;56:290-9.
Dilsizian V. FDG uptake as a surrogate marker for antecedent ischemia. J Nucl Med 2008;49:1909-11.
Kaufmann B, Lewis C, Xie A, Mirza-Mohd A, Lindner JR. Detection of recent myocardial ischemia by molecular imaging of P-selectin with targeted contrast echocardiography. Eur Heart J 2007;28:2011-7.
Marwick TH, Dilsizian V, Narula J. Ischemic episode and hanging on to a painful memory. JACC Cardiovasc Imaging 2012;5:126-8.
Unnikrishnan S, Klibanov AL. Microbubbles as ultrasound contrast agents for molecular imaging: Preparation and application. Am J Roentgenol 2012;199:292-9.
Osterholt M, Sen S, Dilsizian V, Taegtmeyer H. Targeted metabolic imaging to improve the management of heart disease. JACC Cardiovasc Imaging 2012;5:214-26.
Smith CS, Bottomley PA, Schulman SP, Gerstenblith G, Weiss RG. Altered creatine kinase adenosine triphosphate kinetics in failing hypertrophied human myocardium. Circulation 2006;114:1151-8.
Beadle RM, Williams LK, Kuehl M, Bowater S, Abozguia K, Leyva F, et al. Improvement in cardiac energetics by perhexiline in heart failure due to dilated cardiomyopathy. J Am Coll Cardiol HF 2015;3:202-11.
Granér M, Pentikäinen MO, Nyman K, Siren R, Lundbom J, Hakkarainen A, et al. Cardiac steatosis in patients with dilated cardiomyopathy. Heart 2014;100:1107-12.
Corbett JR, Lewis M, Willerson JT, Nicod PH, Huxley RL, Simon T, et al. 99mTc-pyrophosphate imaging in patients with acute myocardial infarction: Comparison of planar imaging with single-photon tomography with and without blood pool overlay. Circulation 1984;69:1120-8.
Narula J, Southern JF, Dec GW, Palacios IF, Newell JB, Fallon JT, et al. Antimyosin uptake and myofibrillarlysis in dilated cardiomyopathy. J Nucl Cardiol 1995;2:2470-7.
Mariani G, Villa G, Rossettin PF, Spallarossa P, Bezante GP, Brunelli C, et al. Detection of acute myocardial infarction by 99mTc-labeled d-glucaric acid imaging in patients with acute chest pain. J Nucl Med 1999;40:1832-9.
Tahara N, Zandbergen HR, de Haas HJ, Petrov A, Pandurangi R, Yamaki T, et al. Noninvasive molecular imaging of cell death in myocardial infarction using 111In-GSAO. Sci Rep 2014;4:6826.
Wang Q, Yang S, Jiang C, Li J, Wang C, Chen L, et al. Discovery of radioiodinated monomeric anthraquinones as a novel class of necrosis avid agents for early imaging of necrotic myocardium. Sci Rep 2016;6:21341.
Narula J, Haider N, Virmani R, DiSalvo TG, Kolodgie FD, Hajjar RJ, et al. Apoptosis in myocytes in end-stage heart failure. N Engl J Med 1996;335:1182-9.
Olivetti G, Abbi R, Quaini F, Kajstura J, Cheng W, Nitahara JA, et al. Apoptosis in the failing human heart. N Engl J Med 1997;336:1131-41.
Hein S, Arnon E, Kostin S, Schonburg M, Elsasser A, Polyakova V, et al. Progression from compensated hypertrophy to failure in the pressure-overloaded human heart: Structural deterioration and compensatory mechanisms. Circulation 2003;107:984-91.
Wencker D, Chandra M, Nguyen K, Miao W, Garantziotis S, Factor SM, et al. A mechanistic role for cardiac myocyte apoptosis in heart failure. J Clin Invest 2003;111:1497-504.
Hayakawa Y, Chandra M, Miao W, Shirani J, Brown JH, Dorn GW, et al. Inhibition of cardiac myocyte apoptosis improves cardiac function and abolishes mortality in the peripartum cardiomyopathy of Galpha(q) transgenic mice. Circulation 2003;108:3036-41.
Hofstra L, Liem IH, Dumont EA, Boersma HH, van Heerde WL, Doevendans PA, et al. Visualisation of cell death in vivo in patients with acute myocardial infarction. Lancet 2000;15:209-12.
Thimister PW, Hofstra L, Liem IH, Boersma HH, Kemerink G, Reutelingsperger CP, et al. In vivo detection of cell death in the area at risk in acute myocardial infarction. J Nucl Med 2003;44:391-6.
Kietselaer BL, Reutelingsperger CP, Boersma HH, Heidendal GA, Liem IH, Crijns HJ, et al. Noninvasive detection of programmed cell loss with 99mTc-labeled annexin A5 in heart failure. J Nucl Med 2007;48:562-7.
Narula J, Acio ER, Narula N, Samuels LE, Fyfe B, Wood D, et al. Annexin-V imaging for noninvasive detection of cardiac allograft rejection. Nat Med 2001;7:1347-52.
Kown MH, Strauss HW, Blankenberg FG, Berry GJ, Stafford-Cecil S, Tait JF, et al. In vivo imaging of acute cardiac rejection in human patients using 99mTechnetium labeled annexin V. Am J Transplant 2001;1:270-7.
Kietselaer BL, Narula J, Hofstra L. The Annexin code: Revealing endocarditis. Eur Heart J 2007;28:948.
Hofstra L, Dumont EA, Thimister PW, Heidendal GA, DeBruine AP, Elenbaas TW, et al. In vivo detection of apoptosis in an intracardiac tumor. JAMA 2001;285:1841-2.
Kenis H, Zandbergen HR, Hofstra L, Petrov AD, Dumont EA, Blankenberg FD, et al. Annexin A5 uptake in ischemic myocardium: Demonstration of reversible phosphatidylserine externalization and feasibility of radionuclide imaging. J Nucl Med 2010;51:259-67.
Mann DL, Bristow MR. Mechanisms and models in heart failure: The biomechanical model and beyond. Circulation 2005;111:2837-49.
Schwartz PJ, De Ferrari GM. Sympathetic-parasympathetic interaction in health and disease: Abnormalities and relevance in heart failure. Heart Fail Rev 2011;16:101-7.
Floras JS. Sympathetic nervous system activation in human heart failure: Clinical implications of an updated model. J Am Coll Cardiol 2009;54:375-85.
Klein T, Dilsizian V, Cao Q, Chen W, Dickfeld TM. The potential role of iodine-123 metaiodobenzylguanidine imaging for identifying sustained ventricular tachycardia in patients with cardiomyopathy. Curr Cardiol Rep 2013;15:359.
Chen W, Dilsizian V. Cardiac Sympathetic Disturbance in the pathophysiology of takotsubo cardiomyopathy: primary etiology or a compensatory response to heart failure? JACC Cardiovasc Imaging 2016; July Issue. [Epub ahead of print].
Jacobson AF, Senior R, Cerqueira MD, Wong ND, Thomas GS, Lopez VA, et al. Myocardial iodine-123 meta-iodobenzylguanidine imaging and cardiac events in heart failure. Results of the prospective ADMIRE-HF (AdreView Myocardial Imaging for Risk Evaluation in Heart Failure) study. J Am Coll Cardiol 2010;55:2212-21.
Badarin FJ, Wimmer AP, Kennedy KF, Jacobson AF, Bateman TM. The utility of ADMIRE-HF risk score in predicting serious arrhythmic events in heart failure patients: Incremental prognostic benefit of cardiac 123I-mIBG scintigraphy. J Nucl Cardiol 2014;21:756-62.
Agostini D, Belin A, Amar MH, Darlas Y, Hamon M, Grollier G, et al. Improvement of cardiac neuronal function after carvedilol treatment in dilated cardiomyopathy: A 123I-MIBG scintigraphic study. J Nucl Med 2000;41:845-51.
Gerson MC, Craft LL, McGuire N, Suresh DP, Abraham WT, Wagoner LE. Carvedilol improves left ventricular function in heart failure patients with idiopathic dilated cardiomyopathy and a wide range of sympathetic nervous system function as measured by iodine 123 metaiodobenzylguanidine. J Nucl Cardiol 2002;9:608-15.
Kasama S, Toyama T, Kumakura H, Takayama Y, Ichikawa S, Suzuki T, et al. Addition of valsartan to an angiotensin-converting enzyme inhibitor improves cardiac sympathetic nerve activity and left ventricular function in patients with congestive heart failure. J Nucl Med 2003;44:884-90.
Kasama S, Toyama T, Kumakura H, Takayama Y, Ichikawa S, Suzuki T, et al. Spironolactone improves cardiac sympathetic nerve activity and symptoms in patients with congestive heart failure. J Nucl Med 2002;43:1279-85.
Cha YM, Oh J, Miyazaki C, Hayes DL, Rea RF, Shen WK, et al. Cardiac resynchronization therapy upregulates cardiac autonomic control. J Cardiovasc Electrophysiol 2008;19:1045-52.
Drakos SG, Athanasoulis T, Malliaras KG, Terrovitis JV, Diakos N, Koudoumas D, et al. Myocardial sympathetic innervation and long-term left ventricular mechanical unloading. JACC Cardiovasc Imaging 2010;3:64-70.
Nishioka SA, Martinelli Filho M, Brandão SC, Giorgi MC, Vieira ML, Costa R, et al. Cardiac sympathetic activity pre and post resynchronization therapy evaluated by 123I-MIBG myocardial scintigraphy. J Nucl Cardiol 2007;14:852-9.
Klein T, Huang R, Smith MF, Abdulghani M, Asoglu R, Remo BF, et al. Three-dimensional 123I-Meta-iodobenzylguanidine cardiac innervation maps to assess substrate and successful ablation sites for ventricular tachycardia: A feasibility study for a novel paradigm of innervation imaging. Circ Arrhythm Electrophysiol 2015;8:583-91.
Abdulghani M, Duell J, Smith M, Chen W, Bentzen SM, Asoglu R, et al. Global and regional myocardial innervation before and after ablation of drug-refractory ventricular tachycardia assessed with 123I-MIBG. J Nucl Med 2015;56:52s-8s.
Levy WC, Lee KL, Hellkamp AS, Poole JE, Mozaffarian D, Linker DT, et al. Maximizing survival benefit with primary prevention implantable cardioverter-defibrillator therapy in a heart failure population. Circulation 2009;120:835-42.
Nakajima K, Nakata T, Yamada T, Yamashina S, Momose M, Kasama S, et al. A prediction model for 5-year cardiac mortality in patients with chronic heart failure using 123I-metaiodobenzylguanidine imaging. Eur J Nucl Med Mol Imaging 2014;41:1673-82.
Boogers MJ, Borleffs CJ, Henneman MM, van Bommel RJ, van Ramshorst J, Boersma E, et al. Cardiac sympathetic denervation assessed with 123-iodine metaiodobenzylguanidine imaging predicts ventricular arrhythmias in implantable cardioverter-defibrillator patients. J Am Coll Cardiol 2010;55:2769-77.
Eckelman WC, Dilsizian V. Chemistry and biology of radiotracers that target changes in sympathetic and parasympathetic nervous system in heart disease. J Nucl Med 2015;56:7S-10S.
Dilsizian V, Eckelman WC. Myocardial blood flow and innervation measures from a single scan: An appealing concept but a challenging paradigm. J Nucl Med 2015;56:1645-6.
Dilsizian V, Narula J. Have imagers aptly or inadvertently overlooked the neuronal myocardial compartment? J Nucl Med 2015;56:1S-2S.
Dilsizian V, Chandrashekhar Y, Narula J. Introduction of new tests: Low are the mountains, high the expectations. JACC Cardiovasc Imaging 2010;3:117-9.
Fallavollita JA, Heavey BM, Luisi AJ Jr, Michalek SM, Baldwa S, Mashtare TL Jr, et al. Regional myocardial sympathetic denervation predicts the risk of sudden cardiac arrest in ischemic cardiomyopathy. J Am Coll Cardiol 2014;63:141-9.
Matsunari I, Aoki H, Nomura Y, Takeda N, Chen WP, Taki J, et al. Iodine-123 metaiodobenzylguanidine imaging and carbon-11 hydroxyephedrine positron emission tomography compared in patients with left ventricular dysfunction. Circ Cardiovasc Imaging 2010;3:595-603.
Sinusas AJ, Lazewatsky J, Brunetti J, Heller G, Srivastava A, Liu YH, et al. Biodistribution and radiation dosimetry of LMI1195: First-in-human study of a novel 18F-labeled tracer for imaging myocardial innervation. J Nucl Med 2014;55:1445-51.
Elsinga PH, Van Waarde A, Visser GM, Vaalburg W. Synthesis and preliminary evaluation of (R, S)-1-[2-((carbamoyl-4-hydroxy)phenoxy)-ethylamino]-3-[4-(1-[11C]-met hyl-4-trifluoromethyl-2-imidazolyl)phenoxy]-2-propanol ([11C]CGP 20712A) as a selective beta 1-adrenoceptor ligand for PET. Nucl Med Biol 1994;21:211-7.
Naya M, Tsukamoto T, Morita K, Katoh C, Nishijima K, Komatsu H, et al. Myocardial beta-adrenergic receptor density assessed by 11C-CGP12177 PET predicts improvement of cardiac function after carvedilol treatment in patients with idiopathic dilated cardiomyopathy. J Nucl Med 2009;50:220-5.
Singh JP, Kandala J, Camm AJ. Non-pharmacological modulation of the autonomic tone to treat heart failure. Eur Heart J 2014;35:77-85.
Delforge J, Janier M, Syrota A, Crouzel C, Vallois JM, Cayla J, et al. Noninvasive quantification of muscarinic receptors in vivo with positron emission tomography in the dog heart. Circulation 1990;82:1494-504.
Bucerius J, Joe AY, Schmaljohann J, Gündisch D, Minnerop M, Biersack HJ, et al. Feasibility of 2-deoxy-2-[18F]fluoro-d-glucose-A85380-PET for imaging of human cardiac nicotinic acetylcholine receptors in vivo. Clin Res Cardiol 2006;95:105-9.
Syrota A, Comar D, Paillotin G, Davy JM, Aumont MC, Stulzaft O, et al. Muscarinic cholinergic receptor in the human heart evidenced under physiological conditions by positron emission tomography. Proc Natl Acad Sci USA 1985;82:584-8.
Le Guludec D, Cohen-Solal A, Delforge J, Delahaye N, Syrota A, Merlet P. Increased myocardial muscarinic receptor density in idiopathic dilated cardiomyopathy: An in vivo PET study. Circulation 1997;96:3416-22.
Mazzadi AN, Pineau J, Costes N, Le Bars D, Bonnefoi F, Croisille P, et al. Muscarinic receptor upregulation in patients with myocardial infarction: A new paradigm. Circ Cardiovasc Imaging 2009;2:365-72.
Bucerius J, Manka C, Schmaljohann J, Mani V, Gündisch D, Rudd JHF, et al. Feasibility of [18F]-2-Fluoro-A85380-PET imaging of human vascular nicotinic acetylcholine receptors in vivo. JACC Cardiovasc Imaging 2012;5:528-36.
Caravan P, Das B, Dumas S, Epstein FH, Helm PA, Jacques V, et al. Collagen-targeted MRI contrast agent for molecular imaging of fibrosis. Angew Chem Int Ed Engl2007;46:8171-3.
Helm PA, Caravan P, French BA, Jacques V, Shen L, Xu Y, et al. Postinfarction myocardial scarring in mice: Molecular MR imaging with use of a collagen-targeting contrast agent. Radiology 2008;247:788-96.
Spuentrup E, Ruhl KM, Botnar RM, Wiethoff AJ, Buhl A, Jacques V, et al. Molecular magnetic resonance imaging of myocardial perfusion with EP-3600, a collagen-specific contrast agent: Initial feasibility study in a swine model. Circulation 2009;119:1768-75.
Muzard J, Sarda-Mantel L, Loyau S, Meulemans A, Louedec L, Bantsimba-Malanda C, et al. Non-invasive molecular imaging of fibrosis using a collagen-targeted peptidomimetic of the platelet collagen receptor glycoprotein VI. PLoS One 2009;4:e5585.
Velikyan I, Rosenström U, Bulenga TN, Eriksson O, Antoni G. Feasibility of multiple examinations using 68Ga-labelled collagelin analogues: Organ distribution in rat for extrapolation to human organ and whole-body radiation dosimetry. Pharmaceuticals 2016;9:31. doi:10.3390/ph9020031.
Phatharajaree W, Phrommintikul A, Chattipakorn N. Matrix metalloproteinases and myocardial infarction. Can J Cardiol 2007;23:727-33.
Chen J, Tung C-H, Allport JR, Chen S, Weissleder R, Huang PL. Near-infrared fluorescent imaging of matrix metalloproteinase activity after myocardial infarction. Circulation 2005;111:1800-5.
Nahrendorf M, Hu K, Frantz S, Jaffer FA, Tung CH, Hiller KH, et al. Factor XIII deficiency causes cardiac rupture, impairs wound healing, and aggravates cardiac remodeling in mice with myocardial infarction. Circulation 2006;113:1196-202.
Su H, Spinale FG, Dobrucki LW, Song J, Hua J, Sweterlitsch S, et al. Noninvasive targeted imaging of matrix metalloproteinase activation in a murine model of postinfarction remodeling. Circulation 2005;112:3157-67.
Sahul ZH, Mukherjee R, Song J, McAteer J, Stroud RE, Dione DP, et al. Targeted imaging of the spatial and temporal variation of matrix metalloproteinase activity in a porcine model of postinfarct remodeling: Relationship to myocardial dysfunction. Circ Cardiovasc Imaging 2011;4:381-91.
de Haas HJ, van den Borne SW, Boersma HH, Slart RH, Fuster V, Narula J. Evolving role of molecular imaging for new understanding: Targeting myofibroblasts to predict remodeling. Ann N Y Acad Sci 2012;1254:33-41.
Gillmore JD, Maurer MS, Falk RH, Merlini G, Damy T, Dispenzieri A, et al. Non-biopsy diagnosis of cardiac transthyretin amyloidosis. Circulation 2016;133:2404-12.
Aras O, Messina SA, Shirani J, Eckelman WC, Dilsizian V. The role and regulation of cardiac angiotensin-converting enzyme for noninvasive molecular imaging in heart failure. Curr Cardiol Rep 2007;9:150-8.
Shirani J, Dilsizian V. Novel molecular angiotensin converting enzyme and angiotensin receptor imaging techniques. Curr Cardiol Rep 2014;16:466.
Gregori M, Tocci G, Marra A, et al. Inadequate RAAS suppression is associated with excessive left ventricular mass and systo-diastolic dysfunction. Clin Res Cardiol 2013;102:725-33.
Oemrawsingh RM, Akkerhuis KM, Van Vark LC, Redekop WK, Rudez G, Remme WJ, et al. Individualized angiotensin-converting enzyme (ACE)-inhibitor therapy in stable coronary artery disease based on clinical and pharmacogenetic determinants: The PERindopril GENEtic (PERGENE) risk model. J Am Heart Assoc 2016;4:e002688.
Kuenzli A, Bucher HC, Anand I, Arutiunov G, Kum LC, McKelvie R, et al. Meta-analysis of combined therapy with angiotensin receptor antagonists versus ACE inhibitors alone in patients with heart failure. PLoS One 2010;5:e9946.
McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014;371:993-1004.
Mathews WB, Burns HD, Dannals RF, Ravert HT, Naylor MN. Carbon-11 labeling of the potent nonpeptide angiotensin-II antagonist MK-996. J Label Compd Radiopharm 1995;36:729-37.
Hamill TG, Burns HD, Dannals RF, Mathews WB, Musachio JL, Ravert HT, et al. Development of [11C]L-159,884: A radiolabelled, nonpeptide angiotensin II antagonist that is useful for angiotensin II, AT1 receptor imaging. Appl Radiat Isot 1996;47:211-8.
Mathews WB, Yoo SE, Lee SH, Scheffel U, Rauseo PA, Zober TG, et al. A novel radioligand for imaging the AT1 angiotensin receptor with PET. Nucl Med Biol 2004;31:571-4.
Higuchi T, Fukushima K, Xia J, Mathews WB, Lautamäki R, Bravo PE, et al. Radionuclide imaging of angiotensin II type 1 receptor upregulation after myocardial ischemia-reperfusion injury. J Nucl Med 2010;51:1956-61.
Zober TG, Mathews WB, Seckin E, Yoo SE, Hilton J, Xia J, et al. PET imaging of the AT1 receptor with [11C]KR31173. Nucl Med Biol 2006;33:5-13.
Fukushima K, Bravo PE, Higuchi T, Schuleri KH, Lin X, Abraham MR, Xia J, et al. Molecular hybrid positron emission tomography/computed tomography imaging of cardiac angiotensin II type 1 receptors. J Am Coll Cardiol 2012;60:2527-34.
Schindler TH, Dilsizian V. Cardiac positron emission tomography/computed tomography imaging of the renin-angiotensin system in humans holds promise for image-guided approach to heart failure therapy. J Am Coll Cardiol 2012;5:1269-84.
Ibrahim IT, Sanad MH. Radiolabeling and biological evaluation of losartan as a possible cardiac imaging agent. Radiochemistry 2013;55:336-40.
Hadizad T, Collins J, Antoun RE, Beanlands RS, DaSilva JN. [11C]Methyl-losartan as a potential ligand for PET imaging angiotensin II AT1 receptors. J Label Compd Radiopharm 2011;54:754-7.
Arksey N, Hadizad T, Ismail B, Hachem M, Valdivia AC, Beanlands RS, deKemp RA, DaSilva JN. Synthesis and evaluation of the novel 2-[18F]fluoro-3-propoxy-triazole-pyridine-substituted losartan for imaging AT1 receptors. Bioorg Med Chem 2014;22:3931-7.
Verjans JW, Lovhaug D, Narula N, Petrov AD, Indrevoll B, Bjurgert E, et al. Noninvasive imaging of angiotensin receptors after myocardial infarction. JACC Cardiovasc Imaging 2008;1:354-62.
Hadizad T, Kirkpatrick SA, Mason S, Burns K, Beanlands RS, Dasilva JN. Novel O-[(11)C]methylated derivatives of candesartan as angiotensin II AT(1) receptor imaging ligands: Radiosynthesis and ex vivo evaluation in rats. Bioorg Med Chem 2009;17:7971-7.
Lortie M, DaSilva JN, Kirkpatrick SA, Hadizad T, Ismail BA, Beanlands RS, deKemp RA. Analysis of [11C]methyl-candesartan kinetics in the rat kidney for the assessment of angiotensin II type 1 receptor density in vivo with PET. Nucl Med Biol 2013;40:252-61.
Hwang DR, Eckelman WC, Mathias CJ, Petrillo EW Jr, Lloyd J, Welch MJ. Positron-labeled angiotensin-converting enzyme (ACE) inhibitor: Fluorine-18-fluorocaptopril. Probing the ACE activity in vivo by positron emission tomography. J Nucl Med 1991;32:1730-7.
Lee YHC, Kiesewetter DO, Lang L, Jagoda EM, Shirani J, Dilsizian V, Eckelman WC. Synthesis of 4-[18F]fluorobenzoyllisinopril: A radioligand for angiotensin converting enzyme (ACE) imaging with positron emission tomography. J Label Compd Radiopharm 2001;44:S268-70.
Qing F, McCarthy TJ, Markham J, Schuster DP. Pulmonary angiotensin-converting enzyme (ACE) binding and inhibition in humans. A positron emission tomography study. Am J Respir Crit Care Med 2000;161:2019-25.
Matarrese M, Salimbeni A, Turolla EA, Turozzi D, Moresco RM, Poma D, et al. 11C-Radiosynthesis and preliminary human evaluation of the disposition of the ACE inhibitor [11C]zofenoprilat. Bioorg Med Chem 2004;12:603-11.
Jackson B, Cubela R, Johnston C. Angiotensin converting enzyme (ACE), characterization by 125I-MK351A binding studies of plasma and tissue ACE during variation of salt status in the rat. J Hypertens 1986;4:759-65.
Femia FJ, Maresca KP, Hillier SM, Zimmerman CN, Joyal JL, Barrett JA, et al. Synthesis and evaluation of a series of 99mTc(CO)3+ lisinopril complexes for in vivo imaging of angiotensin-converting enzyme expression. J Nucl Med 2008;49:970-7.
Dilsizian V, Eckelman WC, Loredo ML, Jagoda EM, Shirani J. Evidence for tissue angiotensin-converting enzyme in explanted hearts of ischemic cardiomyopathy using targeted radiotracer technique. J Nucl Med 2007;48:182-7.
Dilsizian V, Zynda TK, Petrov A, Ohshima S, Tahara N, Haider N, et al. Molecular imaging of human ACE-1 expression in transgenic rats. JACC Cardiovasc Imaging 2012;5:409-18.
Chen W, Dilsizian V. PET Assessment of vascular inflammation and atherosclerotic plaques: SUV or TBR? J Nucl Med 2015;56:503-4.
Disclosure
JS, AS, SA, and VD have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shirani, J., Singh, A., Agrawal, S. et al. Cardiac molecular imaging to track left ventricular remodeling in heart failure. J. Nucl. Cardiol. 24, 574–590 (2017). https://doi.org/10.1007/s12350-016-0620-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-016-0620-2