Abstract
Background
Myocardial angiogenesis following reperfusion of an infarcted area may impact on patient prognosis and pro-angiogenic treatments are currently evaluated. The non-invasive imaging of angiogenesis would therefore be of potential clinical relevance in these settings. 99mTc-RAFT-RGD is a novel 99mTc-labeled tracer that targets the αvβ3 integrin. Our objective was to determine whether this tracer was suitable for myocardial angiogenesis imaging.
Methods and Results
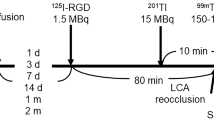
A rat model of reperfused myocardial infarction was employed. Fourteen days following reperfusion, the animals were injected with 99mTc-RAFT-RGD or with its negative control 99mTc-RAFT-RAD. Fourteen animals were dedicated to autoradiographic imaging, infarct staining, and gamma-well counting of myocardial activity. In vivo dual-isotope pinhole SPECT imaging of 201Tl and 99mTc-RAFT-RGD or 99mTc-RAFT-RAD was also performed in 11 additional animals. Neovessels were observed by immunostaining in the infarcted and peri-infarct areas. 99mTc-RAFT-RGD infarct-to-normal ratios by gamma-well counting and ex vivo imaging (2.5 ± 0.6 and 4.9 ± 0.9, respectively) were significantly higher than those of 99mTc-RAFT-RAD (1.7 ± 0.2 and 2.2 ± 0.4, respectively, P < .05). The infarcted area was readily visible in vivo by SPECT with 99mTc-RAFT-RGD but not with 99mTc-RAFT-RAD (infarct-to-normal zone activity ratio, 2.5 ± 0.6 and 1.7 ± 0.4, respectively, P < .05).
Conclusion
99mTc-RAFT-RGD allowed the experimental in vivo molecular imaging of myocardial angiogenesis.




Similar content being viewed by others
References
Cardiovascular Diseases. World Health Organization, fact sheet #317, 2007. Available at http://www.who.int/mediacentre/factsheets/fs317/en/index.html.
Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, et al. Heart disease and stroke statistics—2010 update. A report from the American Heart Association. Circulation. 2009. [Epub ahead of print].
Sutton MG, Sharpe N. Left ventricular remodeling after myocardial infarction—pathophysiology and therapy. Circulation 2000;101:2981-8.
Gheorghiade M, Sopko G, De Luca L, Velazquez EJ, Parker JD, Binkley PF, et al. Navigating the crossroads of coronary artery disease and heart failure. Circulation 2006;114:1202-13.
Frangogiannis NG, Smithb CW, Entman ML. The inflammatory response in myocardial infarction. Cardiovasc Res 2002;53:31-47.
Tucker GC. Integrins: Molecular targets in cancer therapy. Curr Oncol Rep 2006;8:96-103.
Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J, et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 2001;7:430-6.
Hao X, Mansson-Broberg A, Grinnemo KH, Siddiqui AJ, Dellgren G, Brodin LA, et al. Myocardial angiogenesis after plasmid or adenoviral VEGF-A(165) gene transfer in rat myocardial infarction model. Cardiovasc Res 2007;73:481-7.
Okazaki T, Ebihara S, Asada M, Yamanda S, Saijo Y, Shiraishi Y, et al. Macrophage colony-stimulating factor improves cardiac function after ischemic injury by inducing vascular endothelial growth factor production and survival of cardiomyocytes. Am J Pathol 2007;71:1093-103.
Chen LL, Yin H, Huang J. Inhibition of TGF-beta1 signaling by eNOS gene transfer improves ventricular remodeling after myocardial infarction through angiogenesis and reduction of apoptosis. Cardiovasc Pathol 2007;16:221-30.
Rafii S, Lyden D. Therapeutic stem and progenitor cell transplantation for organ vascularization and regeneration. Nat Med 2003;9:702-12.
Hughes GC, Biswas SS, Yin B, Coleman RE, DeGrado TR, Landolfo CK, et al. Therapeutic angiogenesis in chronically ischemic porcine myocardium: Comparative effects of bFGF and VEGF. Ann Thorac Surg 2004;77:812-8.
Simons M, Ware JA. Therapeutic angiogenesis in cardiovascular disease. Nat Rev Drug Discov 2003;2:863-71.
Koneru S, Penumathsa SV, Thirunavukkarasu M, Vidavalur R, Zhan L, Singal PK, et al. Sildenafil mediated neovascularization and protection against myocardial ischemia reperfusion injury in rats: Probable role of VEGF/angiopoietin-1. J Cell Mol Med 2008;12:2651-64.
Halkos ME, Zhao ZQ, Kerendi F, Wang NP, Jiang R, Schmarkey LS, et al. Intravenous infusion of mesenchymal stem cells enhances regional perfusion and improves ventricular function in a porcine model of myocardial infarction. Basic Res Cardiol 2008;103:525-36.
Yla-Herttuala S, Rissanen TT, Vajanto I, Hartikainen J. Vascular endothelial growth factors: Biology and current status of clinical applications in cardiovascular medicine. J Am Coll Cardiol 2007;49:1015-26.
Nordlie MA, Wold LE, Simkhovich BZ, Sesti C, Kloner RA. Molecular aspects of ischemic heart disease: Ischemia/reperfusion-induced genetic changes and potential applications of gene and RNA interference therapy. J Cardiovasc Pharmacol Ther 2006;11:17-30.
Sinusas AJ. Imaging of angiogenesis. J Nucl Cardiol 2004;11:617-33.
Brooks PC, Clark RA, Cheresh DA. Requirement of vascular integrin αVβ3 for angiogenesis. Science 1994;264:569-71.
Ruoslahti E. The RGD story: A personal account. Matrix Biol 2003;22:459-65.
Haubner R, Weber WA, Beer AJ, Vabuliene E, Reim D, Sarbia M, et al. Non-invasive visualization of the activated αVβ3 integrin in cancer patients by positron emission tomography and [18F]-galacto-RGD. PLoS Med 2005;2:244-52.
Fani M, Psimadas D, Zikos C, Xanthopoulos S, Loudos GK, Bouziotis P, et al. Comparative evaluation of linear and cyclic 99mTc-RGD peptides for targeting of integrins in tumor angiogenesis. Anticancer Res 2006;26:431-4.
Boturyn D, Coll JL, Garanger E, Favrot MC, Dumy P. Template assembled cyclopeptides as multimeric system for integrin targeting and endocytosis. J Am Chem Soc 2004;126:5730-9.
Sancey L, Ardisson V, Riou LM, Ahmadi M, Marti-Batlle D, Boturyn D, et al. In vivo imaging of tumour angiogenesis in mice with the alpha(v)beta (3) integrin-targeted tracer 99mTc-RAFT-RGD. Eur J Nucl Med Mol Imaging 2007;34:2037-47.
Sancey L, Garanger E, Foillard S, Schoehn G, Hurbin A, Albiges-Rizo C, et al. Clustering and internalization of integrin alphavbeta3 with a tetrameric RGD-synthetic peptide. Mol Ther 2009;17:837-43.
Makowski MR, Ebersberger U, Nekolla S, Schwaiger M. In vivo molecular imaging of angiogenesis, targeting alphavbeta3 integrin expression, in a patient after acute myocardial infarction. Eur Heart J 2008;29:2201.
Meoli DF, Sadeghi MM, Krassilnikova S, Bourke BN, Giordano FJ, Dione DP, et al. Noninvasive imaging of myocardial angiogenesis following experimental myocardial infarction. J Clin Invest 2004;113:1684-91.
Savill J, Dransfield I, Hogg N, Haslett C. Vitronectin receptor-mediated phagocytosis of cells undergoing apoptosis. Nature 1990;343:170-3.
Sun Y, Zhang JQ, Zhang J, Lamparter S. Cardiac remodeling by fibrous tissue after infarction in rats. J Lab Clin Med 2000;135:316-23.
Higuchi T, Bengel FM, Seidl S, Watzlowik P, Kessler H, Hegenloh R, et al. Assessment of avb3 integrin expression after myocardial infarction by positron emission tomography. Cardiovasc Res 2008;78:395-403.
Dijkgraaf I, Kruijtzer JA, Liu S, Soede AC, Oyen WJ, Corstens FH, et al. Improved targeting of the alpha(v)beta(3) integrin by multimerisation of RGD peptides. Eur J Nucl Med Mol Imaging 2007;34:267-73.
Garanger E, Boturyn D, Coll JL, Favrot MC, Dumy P. Multivalent RGD synthetic peptides as potent alphaVbeta3 integrin ligands. Org Biomol Chem 2006;4:1958-65.
Cheng YF, Kramer RH. Human microvascular endothelial cells express integrin-related complexes that mediate adhesion to the extracellular matrix. J Cell Physiol 1989;139:275-86.
Whalen DA, Hamilton DG, Ganote CE, Jennings RB. Effect of a transient period of ischemia on myocardial cells. I. Effects on cell volume regulation. Am J Pathol 1974;74:381-97.
Riou LM, Broisat A, Lartizien C, Toufektsian MC, Maitrejean S, Janier M, et al. Assessment of non-reperfused and reperfused myocardial infarction using diffusible or deposited radiolabelled perfusion imaging agents. Eur J Nucl Med Mol Imaging 2007;34:330-7.
Mulder WJ, Strijkers GJ, Habets JW, Bleeker EJ, van der Schaft DW, Storm G, et al. MR molecular imaging and fluorescence microscopy for identification of activated tumor endothelium using a bimodal lipidic nanoparticle. FASEB J 2005;19:2008-10.
Lindsey ML, Escobar PG, Dobrucki LW, Goshorn DK, Bouges S, Mingoia JT, et al. Matrix metalloproteinase-9 gene deletion facilitates angiogenesis after myocardial infarction. Am J Physiol Heart Circ Physiol 2006;290:H232-9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding
Financial support was provided by the National Institute for Health and Medical Research (INSERM), the National Agency for Research and technology (ANRT), and ERAS Labo. The authors have no conflict of interest to disclose.
Rights and permissions
About this article
Cite this article
Dimastromatteo, J., Riou, L.M., Ahmadi, M. et al. In vivo molecular imaging of myocardial angiogenesis using the αvβ3 integrin-targeted tracer 99mTc-RAFT-RGD. J. Nucl. Cardiol. 17, 435–443 (2010). https://doi.org/10.1007/s12350-010-9191-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-010-9191-9