Abstract
Introduction
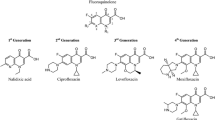
Beginning with second-generation ciprofloxacin 0.3% and ofloxacin 0.3%, fluoroquinolones have been widely used in the treatment and prophylaxis of ocular infections. However, their in-vitro potencies have been decreasing steadily since their introduction. Third-generation levofloxacin 0.5% produces higher ocular tissue penetration, thereby reducing the risk of selecting for decreased fluoroquinolone potency. Fourth-generation gatifloxacin 0.3% and moxifloxacin 0.5% have structural modifications that both reduce risk of resistance and improve potency against Gram-positive bacteria. A new third-generation formulation, levofloxacin 1.5%, was recently introduced, demonstrating increased ocular penetration compared with gatifloxacin 0.3% but clinical equivalence to its second-generation parent, ofloxacin 0.3%, in two randomized trials.
Methods
We investigated the therapeutic potential of levofloxacin 1.5% and compared it to that of existing fourth-generation fluoroquinolones. A MEDLINE search was conducted using the following search terms: moxifloxacin or gatifloxacin; levofloxacin; minimum inhibitory concentration or prevention or prophylaxis; keratitis or endophthalmitis.
Results
Nine eligible studies published between 2002 and 2008 were identified, eight of which are presented. The five in-vitro studies demonstrated that moxifloxacin and gatifloxacin are statistically more potent than levofloxacin against Gram-positive organisms and similar in potency in most cases of Gram-negative bacteria. In-vivo animal models testing moxifloxacin or gatifloxacin against levofloxacin 0.5% (no clinical trials testing the efficacy of levofloxacin 1.5% have yet been published) demonstrated that fourth-generation agents were superior to third-generation levofloxacin 0.5% for prophylaxis of Gram-positive bacteria-induced infections and were equal to, or better than, levofloxacin 0.5% for the treatment of Gram-negative infections.
Conclusion
Fourth-generation agents have increased potency against Gram-positive bacteria compared with levofloxacin, while maintaining similar potency against Gram-negative bacteria. Although levofloxacin 1.5% has demonstrated superior ocular penetration relative to gatifloxacin, the limited available data do not suggest this translates into superior clinical activity compared with moxifloxacin, which has significantly greater ocular penetration and better Gram-positive potency than gatifloxacin.
Similar content being viewed by others
References
Ciloxan® [package insert]. Fort Worth, TX: Alcon Laboratories, Inc.; 2006.
Ocuflox® [package insert]. Irvine, CA: Allergan, Inc.; 2002.
Olson RJ. Reducing the risk of postoperative endophthalmitis. Surv Ophthalmol. 2004;49(suppl. 2):55–61.
Blondeau JM. Fluoroquinolones: mechanism of action, classification, and development of resistance. Surv Ophthalmol. 2004;49(suppl. 2):73–78.
Garcia-Rodriguez JA, Gomez Garcia AC. The microbiology of moxifloxacin. Drugs Today (Barc). 2000;36:215–227.
Just PM. Overview of the fluoroquinolone antibiotics. Pharmacotherapy. 1993;13:4–17.
Chaudhry NA, Flynn HW Jr., Murray TG, Tabandeh H, Mello MO Jr., Miller D. Emerging ciprofloxacin-resistant Pseudomonas aeruginosa. Am J Ophthalmol. 1999;128:509–510.
Goldstein MH, Kowalski RP, Gordon J. Emerging fluoroquinolones resistance in bacterial keratitis: a 5-year review. Ophthalmology. 1999;106:1313–1318.
Alexandrakis G, Alfonso EC, Miller D. Shifting trends in bacterial keratitis in South Florida and emerging resistance to fluoroquinolones. Ophthalmology. 2000;107:1497–1502.
Watanabe K, Numata-Watanabe K, Hayasaka S. Methicillin-resistant staphylococci and ofloxacin-resistant bacteria from clinically healthy conjunctivas. Ophthalmic Res. 2001;33:136–139.
Quixin® [package insert]. Jacksonville, FL: Vistakon Pharmaceuticals, Inc.; 2006.
Healy DP, Holland EJ, Nordlund ML, et al. Concentrations of levofloxacin, ofloxacin, and ciprofloxacin in human corneal stromal tissue and aqueous humor after topical administration. Cornea. 2004;23:255–263.
Koch HR, Kulus SC, Roessler M, Ropo A, Geldsetzer K. Corneal penetration of fluoroquinolones: aqueous humor concentrations after topical application of levofloxacin 0.5% and ofloxacin 0.3% eyedrops. J Cataract Refract Surg. 2005;31:1377–1385.
Puustjärvi T, Teräsvirta M, Nurmenniemi P, Lokkila J, Uusitalo H. Penetration of topically applied levofloxacin 0.5% and ofloxacin 0.3% into the vitreous of the noninflamed human eye. Graefes Arch Clin Exp Ophthalmol. 2006;244:1633–1637.
Drugeon HB, Juvin ME, Bryskier A. Relative potential for selection of fluoroquinolone-resistant Streptococcus pneumoniae strains by levofloxacin: comparison with ciprofloxacin, sparfloxacin and ofloxacin. J Antimicrob Chemother. 1999;43(suppl. C):55–59.
Bauernfeind A. Pharmacodynamics of levofloxacin and ofloxacin against Streptococcus pneumoniae. J Antimicrob Chemother. 1999;43(suppl. C):77–82.
Miller D, Alfonso EC. Comparative in vitro activity of levofloxacin, ofloxacin, and ciprofloxacin against ocular streptococcal isolates. Cornea. 2004;23:289–293.
Fisher LM, Heaton VJ. Dual activity of fluoroquinolones against Streptococcus pneumoniae. J Antimicrob Chemother. 2003;51:463–464.
Sanders CC. Review of preclinical studies with ofloxacin. Clin Infect Dis. 1992;14: 526–538.
Wiseman LR, Balfour JA. Ciprofloxacin. A review of its pharmacological profile and therapeutic use in the elderly. Drugs Aging. 1994;4:145–173.
Khodursky AB, Zechiedrich EL, Cozzarelli NR. Topoisomerase IV is a target of quinolones in Escherichia coli. Proc Natl Acad Sci U S A. 1995;92:11801–11805.
Breines DM, Ouabdesselam S, Ng EY, et al. Quinolone resistance locus nfxD of Escherichia coli is a mutant allele of the parE gene encoding a subunit of topoisomerase IV. Antimicrob Agents Chemother. 1997;41:175–179.
Huelves L, Sevillano D, Martínez-Marín C, et al. Correlation between in vitro and in vivo activity of levofloxacin and moxifloxacin against pneumococcal strains with different susceptibilities to fluoroquinolones. Int J Antimicrob Agents. 2006;27:294–299.
Pestova E, Millichap JJ, Noskin GA, Peterson LR. Intracellular targets of moxifloxacin: a comparison with other fluoroquinolones. J Antimicrob Chemother. 2000;45:583–590.
Ogawa GS, Weinstein AJ, Melendez RF, et al. Perioperative regimen in a large series of cataract surgeries with no cases of endophthalmitis. Proceedings of the ASCRS Symposium on Cataract, IOL and Refractive Surgery; April 27–May 2, 2007; San Diego, CA, USA.
Iquix® [package insert]. Jacksonville, FL: Vistakon Pharmaceuticals, Inc.; 2007.
Wagner RS, Abelson MB, Shapiro A, Torkildsen G. Evaluation of moxifloxacin, ciprofloxacin, gatifloxacin, ofloxacin, and levofloxacin concentrations in human conjunctival tissue. Arch Ophthalmol. 2005;123:1282–1283.
Holland EJ, McCarthy M, Holland S. The ocular penetration of levofloxacin 1.5% and gatifloxacin 0.3% ophthalmic solutions in subjects undergoing corneal transplant surgery. Curr Med Res Opin. 2007;23:2955–2960.
Nicolau DP. Predicting antibacterial response from pharmacodynamic and pharmacokinetic profiles. Infection. 2001;29(suppl. 2):11–15.
Duggirala A, Joseph J, Sharma S, Nutheti R, Garg P, Das T. Activity of newer fluoroquinolones against gram-positive and gram-negative bacteria isolated from ocular infections: an in vitro comparison. Indian J Ophthalmol. 2007;55:15–19.
Mather R, Karenchak LM, Romanowski EG, Kowalski RP. Fourth generation fluoroquinolones: new weapons in the arsenal of ophthalmic antibiotics. Am J Ophthalmol. 2002;133:463–466.
Kowalski RP, Dhaliwal DK, Karenchak LM, et al. Gatifloxacin and moxifloxacin: an in vitro susceptibility comparison to levofloxacin, ciprofloxacin, and ofloxacin using bacterial keratitis isolates. Am J Ophthalmol. 2003;136:500–505.
Miller D, Flynn PM, Scott IU, Alfonso EC, Flynn HW Jr. In vitro fluoroquinolone resistance in staphylococcal endophthalmitis isolates. Arch Ophthalmol. 2006;124: 479–483.
Rhee MK, Kowalski RP, Romanowski EG, Mah FS, Ritterband DC, Gordon YJ. A laboratory evaluation of antibiotic therapy for ciprofloxacin-resistant Pseudomonas aeruginosa. Am J Ophthalmol. 2004;138:226–230.
Miller JJ, Scott IU, Flynn HW Jr., Smiddy WE, Corey RP, Miller D. Endophthalmitis caused by Streptococcus pneumoniae. Am J Ophthalmol. 2004;138:231–236.
Kowalski RP, Romanowski EG, Mah FS, Sasaki H, Fukuda M, Gordon YJ. A comparison of moxifloxacin and levofloxacin topical prophylaxis in a fluoroquinolone-resistant Staphylococcus aureus rabbit model. Jpn J Ophthalmol. 2008;52:211–216.
Thibodeaux BA, Dajcs JJ, Caballero AR, Marquart ME, Girgis DO, O’Callaghan RJ. Quantitative comparison of fluoroquinolone therapies of experimental gram-negative bacterial keratitis. Curr Eye Res. 2004;28:337–342.
Tungsiripat T, Sarayba MA, Kaufman MB, et al. Fluoroquinolone therapy in multipledrug resistant staphylococcal keratitis after lamellar keratectomy in a rabbit model. Am J Ophthalmol. 2003;136:76–81.
Han DP, Wisniewski SR, Wilson LA, et al. Spectrum and susceptibilities of microbiologic isolates in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol. 1996;122:1–17. Erratum in: Am J Ophthalmol. 1996;122:920.
Bourcier T, Thomas F, Borderie V, Chaumeil C, Laroche L. Bacterial keratitis: predisposing factors, clinical and microbiological review of 300 cases. Br J Ophthalmol. 2003;87:834–838.
Wagoner MD, Al-Swailem SA, Sutphin JE, Zimmerman MB. Bacterial keratitis after penetrating keratoplasty: incidence, microbiological profile, graft survival, and visual outcome. Ophthalmology. 2007;114:1073–1079.
Vajpayee RB, Sharma N, Sinha R, Agarwal T, Singhvi A. Infectious keratitis following keratoplasty. Surv Ophthalmol. 2007;52:1–12.
Kim DH, Stark WJ, O’Brien TP, Dick JD. Aqueous penetration and biological activity of moxifloxacin 0.5% ophthalmic solution and gatifloxacin 0.3% solution in cataract surgery patients. Ophthalmology. 2005;112:1992–1996.
Holland EJ, Lane SS, Kim T, Raizman M, Dunn S. Ocular penetration and pharmacokinetics of topical gatifloxacin 0.3% and moxifloxacin 0.5% ophthalmic solutions after keratoplasty. Cornea. 2008;27:314–319.
Chang D. Cataract clinical committee survey on antibiotic usage: TASS and endophthalmitis symposium. Proceedings of the ASCRS Symposium on Cataract, IOL and Refractive Surgery; April 27–May 2, 2007; San Diego, CA, USA.
Aliprandis E, Ciralsky J, Lai H, Herling I, Katz HR. Comparative efficacy of topical moxifloxacin versus ciprofloxacin and vancomycin in the treatment of P. aeruginosa and ciprofloxacin-resistant MRSA keratitis in rabbits. Cornea. 2005;24:201–205.
Caballero AR, Marquart ME, O’Callaghan RJ, Thibodeaux BA, Johnston KH, Dajcs JJ. Effectiveness of fluoroquinolones against Mycobacterium abscesses in vivo. Curr Eye Res. 2006;31:23–29.
Babayan Sosa A, Epstein SP, Asbell PA. Evaluation of toxicity of commercial ophthalmic fluoroquinolone antibiotics as assessed on immortalized corneal and conjunctival epithelial cells. Cornea. 2008;27:930–934.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scoper, S.V. Review of third-and fourth-generation fluoroquinolones in ophthalmology: in-vitro and in-vivo efficacy. Adv Therapy 25, 979–994 (2008). https://doi.org/10.1007/s12325-008-0107-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-008-0107-x