Abstract
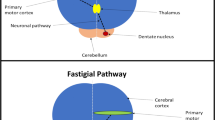
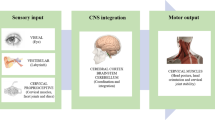
Phantom limb pain (PLP) is a disabling and intractable sensation arising in about 80% of patients after amputation. The aim of this study was to evaluate the possibility to modulate nociceptive processing and pain perception with cerebellar transcranial direct current stimulation (ctDCS) in patients suffering from painful and non-painful phantom limb sensations. Fourteen upper limb amputees underwent ctDCS (anodal or sham, 2.0 mA, 20 min per day, 5 days a week). Clinical scores and electrophysiological parameters were assessed before tDCS, at the end of the 5-day treatment, 2 and 4 weeks later. Laser-evoked potentials (LEPs) were obtained from the stump using a Nd:YAP laser by pulses with short duration (5 ms) and small diameter spots (5 mm). Changes in visual analogue scores (VAS) were evaluated (chronic pain, paroxysmal pain, stump pain, phantom movements, phantom sensations). Anodal polarization significantly dampened LEP amplitudes (N1, p = 0.021 and N2/P2, p = 0.0034), whereas sham intervention left them unchanged. Anodal ctDCS significantly reduced paroxysmal pain (p < 0.0001), non-painful phantom limb sensations (p < 0.0001) and phantom limb movements (p = 0.0003), whereas phantom limb and stump pain did not change compared to the sham condition. Anodal ctDCS significantly improves both paroxysmal pain and non-painful phantom limb sensations, which are likely induced by maladaptive changes in the sensorimotor network and posterior parietal cortex respectively.



Similar content being viewed by others
References
Alviar MJ, Hale T, Dungca M. Pharmacologic interventions for treating phantom limb pain. Cochrane Database Syst Rev. 2016;10:CD006380.
Flor H. Phantom-limb pain: characteristics, causes, and treatment. Lancet Neurol. 2002;1(3):182–9.
Kooijman CM, Dijkstra PU, Geertzen JH, Elzinga A, van der Schans CP. Phantom pain and phantom sensations in upper limb amputees: an epidemiological study. Pain. 2000;87(1):33–41.
Keil G. So-called initial description of phantom pain by Ambroise pare. “chose digne d’admiration et quasi incredible”: the “douleur es parties mortes et amputees”. Fortschr Med. 1990;108(4):62–6.
Diers M, Kamping S, Kirsch P, Rance M, Bekrater-Bodmann R, Foell J, et al. Illusion-related brain activations: a new virtual reality mirror box system for use during functional magnetic resonance imaging. Brain Res. 2015;1594:173–82.
Diers M, Christmann C, Koeppe C, Ruf M, Flor H. Mirrored, imagined and executed movements differentially activate sensorimotor cortex in amputees with and without phantom limb pain. Pain. 2010;149(2):296–304.
Flor H, Nikolajsen L, Staehelin JT. Phantom limb pain: a case of maladaptive CNS plasticity? Nat Rev Neurosci. 2006;7(11):873–81.
Lotze M, Flor H, Grodd W, Larbig W, Birbaumer N. Phantom movements and pain. An fMRI study in upper limb amputees. Brain. 2001;124(Pt 11):2268–77.
Suzuki R, Dickenson A. Spinal and supraspinal contributions to central sensitization in peripheral neuropathy. Neurosignals. 2005;14(4):175–81.
Karl A, Birbaumer N, Lutzenberger W, Cohen LG, Flor H. Reorganization of motor and somatosensory cortex in upper extremity amputees with phantom limb pain. J Neurosci. 2001;21(10):3609–18.
Wrigley PJ, Press SR, Gustin SM, Macefield VG, Gandevia SC, Cousins MJ, et al. Neuropathic pain and primary somatosensory cortex reorganization following spinal cord injury. Pain. 2009;141(1–2):52–9.
Raffin E, Richard N, Giraux P, Reilly KT. Primary motor cortex changes after amputation correlate with phantom limb pain and the ability to move the phantom limb. Neuroimage. 2016;130:134–44.
Andoh J, Milde C, Tsao JW, Flor H. Cortical plasticity as a basis of phantom limb pain: fact or fiction? Neuroscience. 2018;387:85–91.
Kikkert S, Johansen-Berg H, Tracey I, Makin TR. Reaffirming the link between chronic phantom limb pain and maintained missing hand representation. Cortex. 2018;106:174–84.
Makin TR, Scholz J, Filippini N, Henderson Slater D, Tracey I, Johansen-Berg H. Phantom pain is associated with preserved structure and function in the former hand area. Nat Commun. 2013;4:1570.
Ferraro F, Jacopetti M, Spallone V, Padua L, Traballesi M, Brunelli S, et al. Diagnosis and treatment of pain in plexopathy, radiculopathy, peripheral neuropathy and phantom limb pain. Evidence and recommendations from the Italian consensus conference on pain on Neurorehabilitation. Eur J Phys Rehabil Med. 2016;52(6):855–66.
McAuley J, van Groningen R, Green C. Spinal cord stimulation for intractable pain following limb amputation. Neuromodulation. 2013;16(6):530–6 discussion 6.
Aiyer R, Barkin RL, Bhatia A, Gungor S. A systematic review on the treatment of phantom limb pain with spinal cord stimulation. Pain Manag. 2017;7(1):59–69.
Borsook D, Moulton EA, Tully S, Schmahmann JD, Becerra L. Human cerebellar responses to brush and heat stimuli in healthy and neuropathic pain subjects. Cerebellum. 2008;7(3):252–72.
Duerden EG, Albanese MC. Localization of pain-related brain activation: a meta-analysis of neuroimaging data. Hum Brain Mapp. 2013;34(1):109–49.
Bocci T, Santarcangelo E, Vannini B, Torzini A, Carli G, Ferrucci R, et al. Cerebellar direct current stimulation modulates pain perception in humans. Restor Neurol Neurosci. 2015;33(5):597–609.
Bocci T, Barloscio D, Parenti L, Sartucci F, Carli G, Santarcangelo EL. High hypnotizability impairs the cerebellar control of pain. Cerebellum. 2017;16(1):55–61.
Baarbe JK, Yielder P, Haavik H, Holmes MWR, Murphy BA. Subclinical recurrent neck pain and its treatment impacts motor training-induced plasticity of the cerebellum and motor cortex. PLoS One. 2018;13(2):e0193413.
Pereira M, Rafiq B, Chowdhury E, Babayev J, Boo H, Metwaly R, et al. Anodal cerebellar tDCS modulates lower extremity pain perception. NeuroRehabilitation. 2017;40(2):195–200.
Valeriani M, Le Pera D, Restuccia D, de Armas L, Miliucci R, Betti V, et al. Parallel spinal pathways generate the middle-latency N1 and the late P2 components of the laser evoked potentials. Clin Neurophysiol. 2007;118(5):1097–104.
Valeriani M, Pazzaglia C, Cruccu G, Truini A. Clinical usefulness of laser evoked potentials. Neurophysiol Clin. 2012;42(5):345–53.
Garcia-Larrea L, Frot M, Valeriani M. Brain generators of laser-evoked potentials: from dipoles to functional significance. Neurophysiol Clin. 2003;33(6):279–92.
Vogt BA, Derbyshire S, Jones AK. Pain processing in four regions of human cingulate cortex localized with co-registered PET and MR imaging. Eur J Neurosci. 1996;8(7):1461–73.
Bolognini N, Olgiati E, Maravita A, Ferraro F, Fregni F. Motor and parietal cortex stimulation for phantom limb pain and sensations. Pain. 2013;154(8):1274–80.
Bolognini N, Spandri V, Ferraro F, Salmaggi A, Molinari AC, Fregni F, et al. Immediate and sustained effects of 5-day transcranial direct current stimulation of the motor cortex in phantom limb pain. J Pain. 2015;16(7):657–65.
Truini A, Galeotti F, Romaniello A, Virtuoso M, Iannetti GD, Cruccu G. Laser-evoked potentials: normative values. Clin Neurophysiol. 2005;116(4):821–6.
Truini A, Panuccio G, Galeotti F, Maluccio MR, Sartucci F, Avoli M, et al. Laser-evoked potentials as a tool for assessing the efficacy of antinociceptive drugs. Eur J Pain. 2010;14(2):222–5.
Cruccu G, Aminoff MJ, Curio G, Guerit JM, Kakigi R, Mauguiere F, et al. Recommendations for the clinical use of somatosensory-evoked potentials. Clin Neurophysiol. 2008;119(8):1705–19.
Ferrucci R, Marceglia S, Vergari M, Cogiamanian F, Mrakic-Sposta S, Mameli F, et al. Cerebellar transcranial direct current stimulation impairs the practice-dependent proficiency increase in working memory. J Cogn Neurosci. 2008;20(9):1687–97.
Ferrucci R, Brunoni AR, Parazzini M, Vergari M, Rossi E, Fumagalli M, et al. Modulating human procedural learning by cerebellar transcranial direct current stimulation. Cerebellum. 2013;12(4):485–92.
Iyer MB, Mattu U, Grafman J, Lomarev M, Sato S, Wassermann EM. Safety and cognitive effect of frontal DC brain polarization in healthy individuals. Neurology. 2005;64(5):872–5.
Nitsche MA, Liebetanz D, Lang N, Antal A, Tergau F, Paulus W. Safety criteria for transcranial direct current stimulation (tDCS) in humans. Clin Neurophysiol. 2003;114(11):2220–2 author reply 2-3.
Gandiga PC, Hummel FC, Cohen LG. Transcranial DC stimulation (tDCS): a tool for double-blind sham-controlled clinical studies in brain stimulation. Clin Neurophysiol. 2006;117(4):845–50.
Galea JM, Jayaram G, Ajagbe L, Celnik P. Modulation of cerebellar excitability by polarity-specific noninvasive direct current stimulation. J Neurosci. 2009;29(28):9115–22.
Bocci T, Ferrucci R, Barloscio D, Parenti L, Cortese F, Priori A, et al. Cerebellar direct current stimulation modulates hand blink reflex: implications for defensive behavior in humans. Physiol Rep. 2018;6(10):e13471.
Parazzini M, Rossi E, Ferrucci R, Liorni I, Priori A, Ravazzani P. Modelling the electric field and the current density generated by cerebellar transcranial DC stimulation in humans. Clin Neurophysiol. 2014;125(3):577–84.
Parazzini M, Rossi E, Ferrucci R, Fiocchi S, Liorni I, Priori A, et al. Computational model of cerebellar transcranial direct current stimulation. Conf Proc IEEE Eng Med Biol Soc. 2013;2013:237–40.
Brunoni AR, Amadera J, Berbel B, Volz MS, Rizzerio BG, Fregni F. A systematic review on reporting and assessment of adverse effects associated with transcranial direct current stimulation. Int J Neuropsychopharmacol. 2011;14(8):1133–45.
Pinto CB, Saleh Velez FG, Bolognini N, Crandell D, Merabet LB, Fregni F. Optimizing rehabilitation for phantom limb pain using mirror therapy and transcranial direct current stimulation: a randomized, double-blind clinical trial study protocol. JMIR Res Protoc. 2016;5(3):e138.
Moseley GL, Flor H. Targeting cortical representations in the treatment of chronic pain: a review. Neurorehabil Neural Repair. 2012;26(6):646–52.
MacIver K, Lloyd DM, Kelly S, Roberts N, Nurmikko T. Phantom limb pain, cortical reorganization and the therapeutic effect of mental imagery. Brain. 2008;131(Pt 8):2181–91.
Malavera A, Silva FA, Fregni F, Carrillo S, Garcia RG. Repetitive transcranial magnetic stimulation for phantom limb pain in land mine victims: a double-blinded, randomized, sham-controlled trial. J Pain. 2016;17(8):911–8.
Topper R, Foltys H, Meister IG, Sparing R, Boroojerdi B. Repetitive transcranial magnetic stimulation of the parietal cortex transiently ameliorates phantom limb pain-like syndrome. Clin Neurophysiol. 2003;114(8):1521–30.
Ahmed MA, Mohamed SA, Sayed D. Long-term antalgic effects of repetitive transcranial magnetic stimulation of motor cortex and serum beta-endorphin in patients with phantom pain. Neurol Res. 2011;33(9):953–8.
Lee JH, Byun JH, Choe YR, Lim SK, Lee KY, Choi IS. Successful treatment of phantom limb pain by 1 Hz repetitive transcranial magnetic stimulation over affected supplementary motor complex: a case report. Ann Rehabil Med. 2015;39(4):630–3.
Garcia-Larrea L, Peyron R, Laurent B, Mauguiere F. Association and dissociation between laser-evoked potentials and pain perception. Neuroreport. 1997;8(17):3785–9.
Rosen G, Hugdahl K, Ersland L, Lundervold A, Smievoll AI, Barndon R, et al. Different brain areas activated during imagery of painful and non-painful ‘finger movements’ in a subject with an amputated arm. Neurocase. 2001;7(3):255–60.
Anderson WS, Lenz FA. Review of motor and phantom-related imagery. Neuroreport. 2011;22(17):939–42.
Raffin E, Mattout J, Reilly KT, Giraux P. Disentangling motor execution from motor imagery with the phantom limb. Brain. 2012;135(Pt 2):582–95.
Kikkert S, Mezue M, O'Shea J, Henderson Slater D, Johansen-Berg H, Tracey I, et al. Neural basis of induced phantom limb pain relief. Ann Neurol. 2019;85(1):59–73.
Cohen LG, Bandinelli S, Findley TW, Hallett M. Motor reorganization after upper limb amputation in man. A study with focal magnetic stimulation. Brain. 1991;114(Pt 1B):615–27.
Dettmers C, Adler T, Rzanny R, van Schayck R, Gaser C, Weiss T, et al. Increased excitability in the primary motor cortex and supplementary motor area in patients with phantom limb pain after upper limb amputation. Neurosci Lett. 2001;307(2):109–12.
Truini A, Galeotti F, Haanpaa M, Zucchi R, Albanesi A, Biasiotta A, et al. Pathophysiology of pain in postherpetic neuralgia: a clinical and neurophysiological study. Pain. 2008;140(3):405–10.
Truini A, Padua L, Biasiotta A, Caliandro P, Pazzaglia C, Galeotti F, et al. Differential involvement of A-delta and A-beta fibres in neuropathic pain related to carpal tunnel syndrome. Pain. 2009;145(1–2):105–9.
Singer T, Seymour B, O'Doherty J, Kaube H, Dolan RJ, Frith CD. Empathy for pain involves the affective but not sensory components of pain. Science. 2004;303(5661):1157–62.
Ehde DM, Czerniecki JM, Smith DG, Campbell KM, Edwards WT, Jensen MP, et al. Chronic phantom sensations, phantom pain, residual limb pain, and other regional pain after lower limb amputation. Arch Phys Med Rehabil. 2000;81(8):1039–44.
Hunter JP, Katz J, Davis KD. Dissociation of phantom limb phenomena from stump tactile spatial acuity and sensory thresholds. Brain. 2005;128(Pt 2):308–20.
Becotte A, de Medicis E, Lapie V, Martin R, Gagnon V. Preoperative continuous sciatic nerve block for perioperative analgesia and for phantom limb prevention. J Clin Anesth. 2012;24(3):256–7.
Acknowledgments
We gratefully acknowledge the participation of all subjects, as well as Mr. C. Orsini and Mr. D. Barloscio for their excellent technical assistance. The paper was supported in part by the Italian operating and development MIUR PRIN grant year 2006, n.2006062332_002.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Table 1
(DOC 34 kb)
Rights and permissions
About this article
Cite this article
Bocci, T., De Carolis, G., Ferrucci, R. et al. Cerebellar Transcranial Direct Current Stimulation (ctDCS) Ameliorates Phantom Limb Pain and Non-painful Phantom Limb Sensations. Cerebellum 18, 527–535 (2019). https://doi.org/10.1007/s12311-019-01020-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-019-01020-w