Abstract
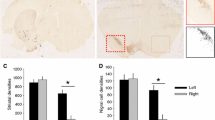
Non-invasive stimulation of the human cerebellum, such as by transcranial magnetic stimulation (TMS), is increasingly used to investigate cerebellar function and identify potential treatment for cerebellar dysfunction. However, the effects of TMS on cerebellar neurons remain poorly defined. We applied low-intensity repetitive TMS (LI-rTMS) to the mouse cerebellum in vivo and in vitro and examined the cellular and molecular sequelae. In normal C57/Bl6 mice, 4 weeks of LI-rTMS using a complex biomimetic high-frequency stimulation (BHFS) alters Purkinje cell (PC) dendritic and spine morphology; the effects persist 4 weeks after the end of stimulation. We then evaluated whether LI-rTMS could induce climbing fibre (CF) reinnervation to denervated PCs. After unilateral pedunculotomy in adult mice and 2 weeks sham or BHFS stimulation, VGLUT2 immunohistochemistry was used to quantify CF reinnervation. In contrast to sham, LI-rTMS induced CF reinnervation to the denervated hemicerebellum. To examine potential mechanisms underlying the LI-rTMS effect, we verified that BHFS could induce CF reinnervation using our in vitro olivocerebellar explants in which denervated cerebellar tissue is co-cultured adjacent to intact cerebella and treated with brain-derived neurotrophic factor (BDNF) (as a positive control), sham or LI-rTMS for 2 weeks. Compared with sham, BDNF and BHFS LI-rTMS significantly increased CF reinnervation, without additive effect. To identify potential underlying mechanisms, we examined intracellular calcium flux during the 10-min stimulation. Complex high-frequency stimulation increased intracellular calcium by release from intracellular stores. Thus, even at low intensity, rTMS modifies PC structure and induces CF reinnervation.

Similar content being viewed by others
References
Pell GS, Roth Y, Zangen A. Modulation of cortical excitability induced by repetitive transcranial magnetic stimulation: Influence of timing and geometrical parameters and underlying mechanisms. Prog Neurobiol. 2011;93:59–98.
Grimaldi G, Argyropoulos GP, Boehringer A, Celnik P, Edwards MJ, Ferrucci R, et al. Non-invasive cerebellar stimulation—a consensus paper. Cerebellum. 2014;13:121–38.
Welsh JP, Lang EJ, Sugihara I, Llinas R. Dynamic organization of motor control within the olivocerebellar system. Nature. 1995;374:453–7.
Lang EJ, Sugihara I, Llinas R. Olivocerebellar modulation of motor cortex ability to generate vibrissal movements in rat. J Physiol. 2006;571:101–20.
Fournier B, Lohof AM, Bower AJ, Mariani J, Sherrard RM. Developmental modifications of olivocerebellar topography: the granuloprival cerebellum reveals multiple routes from the inferior olive. J Comp Neurol. 2005;490:85–97.
Sugihara I. Organization and remodeling of the olivocerebellar climbing fiber projection. Cerebellum. 2006;5:15–22.
Siebner HR, Lang N, Rizzo V, Nitsche MA, Paulus W, Lemon RN et al. Preconditioning of low frequency repetitive transcranial magnetic stimulation with transcranial direct current stimulation: evidence for homeostatic plasticity in the human motor cortex. J Neurosci. 2004;24:3379–3385.
Dixon KJ, Sherrard RM. Brain-derived neurotrophic factor induces post-lesion transcommissural growth of olivary axons that develop normal climbing fibers on mature Purkinje cells. Exp Neurol. 2006;202:44–56.
Willson ML, McElnea C, Mariani J, Lohof AM, Sherrard RM. BDNF increases homotypic olivocerebellar reinnervation and associated fine motor and cognitive skill. Brain. 2008;131:1099–112.
Wang HY, Crupi D, Liu J, Stucky A, Cruciata G, Di Rocco A, et al. Repetitive transcranial magnetic stimulation enhances BDNF–TrkB signaling in both brain and lymphocyte. J Neurosci. 2011;31:11044–54.
Rodger J, Mo C, Wilks T, Dunlop SA, Sherrard RM. Transcranial pulsed magnetic field stimulation facilitates reorganization of abnormal neural circuits and corrects behavioral deficits without disrupting normal connectivity. FASEB J. 2012;26:1593–606.
Chen X, Heck N, Lohof AM, Rochefort C, Morel MP, Wehrlé R, et al. Mature Purkinje cells require the retinoic acid-related orphan receptor alpha (RORα) to maintain climbing fiber monoinnervation and other adult characteristics. J Neurosci. 2013;33:9546–62.
Chédotal A, Bloch-Gallego E, Sotelo C. The embryonic cerebellum contains topographic cues that guide developing inferior olivary axons. Development. 1997;124:861–70.
Letellier M, Wehrlé R, Mariani J, Lohof AM. Synapse elimination in olivo-cerebellar explants occurs during a critical period and leaves an indelible trace in Purkinje cells. Proc Natl Acad Sci U S A. 2009;106:14102–7.
Grehl S, Viola H, Fuller-Carter PI, Carter KW, Dunlop SA, Hool L, Sherrard RM, Rodger J Cellular and molecular changes to cortical neurons following low intensity repetitive magnetic stimulation at different frequencies. Brain Stim. 2014.
Acknowledgments
We wish to thank Tom Dufor, Anaëlle Dumas, Jennifer Fransson and Alix Noel de la Font Reaulx for technical assistance. The study was supported in part by the Institut de la Recherche sur la Moelle épinière et l’Encéphale in France and the Neurotrauma Research Program Western Australia.
Conflict of Interest
There are no current or potential conflicts of interest for any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
N Morellini and S Grehl contributed equally to this work.
AM Lohof and RM Sherrard are co-senior authors.
Rights and permissions
About this article
Cite this article
Morellini, N., Grehl, S., Tang, A. et al. What Does Low-Intensity rTMS Do to the Cerebellum?. Cerebellum 14, 23–26 (2015). https://doi.org/10.1007/s12311-014-0617-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-014-0617-9