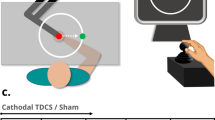
Abstract
When systematic movement errors occur, the brain responds with a systematic change in motor behavior. This type of adaptive motor learning can transfer intermanually; adaptation of movements of the right hand in response to training with a perturbed visual signal (visuomotor adaptation) may carry over to the left hand. While visuomotor adaptation has been studied extensively, it is unclear whether the cerebellum, a structure involved in adaptation, is important for intermanual transfer as well. We addressed this question with three experiments in which subjects reached with their right hands as a 30° visuomotor rotation was introduced. Subjects received anodal or sham transcranial direct current stimulation on the trained (experiment 1) or untrained (experiment 2) hemisphere of the cerebellum, or, for comparison, motor cortex (M1). After the training period, subjects reached with their left hand, without visual feedback, to assess intermanual transfer of learning aftereffects. Stimulation of the right cerebellum caused faster adaptation, but none of the stimulation sites affected transfer. To ascertain whether cerebellar stimulation would increase transfer if subjects learned faster as well as a larger amount, in experiment 3 anodal and sham cerebellar groups experienced a shortened training block such that the anodal group learned more than sham. Despite the difference in adaptation magnitude, transfer was similar across these groups, although smaller than in experiment 1. Our results suggest that intermanual transfer of visuomotor learning does not depend on cerebellar activity and that the number of movements performed at plateau is an important predictor of transfer.





Similar content being viewed by others
References
Lackner JR, DiZio P. Motor control and learning in altered dynamic environments. Curr Opin Neurobiol. 2005;15(6):653–9.
Duff SV, Sainburg RL. Lateralization of motor adaptation reveals independence in control of trajectory and steady-state position. Exp Brain Res. 2007;179(4):551–61.
Vasudevan EVL, Torres-Oviedo G, Morton SM, Yang JF, Bastian AJ. Younger is not always better: development of locomotor adaptation from childhood to adulthood. J Neurosci. 2011;31(8):3055–65.
Bizzi E, Mussa-Ivaldi FA. Neural basis of motor control and its cognitive implications. Trends Cogn Sci (Regul Ed). 1998;2(3):97–102. Mar 1.
Heuer H, Hegele M. Constraints on visuo-motor adaptation depend on the type of visual feedback during practice. Exp Brain Res. 2008;185(1):101–10.
Mistry S, Contreras-Vidal JL. Learning multiple visuomotor transformations: adaptation and context-dependent recall. Mot Control. 2004;8(4):534–46.
Wang J, Sainburg RL. The symmetry of interlimb transfer depends on workspace locations. Exp Brain Res. 2006;170(4):464–71.
Choe CS, Welch RB. Variables affecting the intermanual transfer and decay of prism adaptation. J Exp Psychol. 1974;102(6):1076–84.
Criscimagna-Hemminger SE, Donchin O, Gazzaniga MS, Shadmehr R. Learned dynamics of reaching movements generalize from dominant to nondominant arm. J Neurophysiol. 2003;89(1):168–76.
Wang J, Sainburg RL. Interlimb transfer of novel inertial dynamics is asymmetrical. J Neurophysiol. 2004;92(1):349–60.
Wang J. A dissociation between visual and motor workspace inhibits generalization of visuomotor adaptation across the limbs. Exp Brain Res. 2008;187(3):483–90.
Balitsky Thompson AK, Henriques DY. Visuomotor adaptation and intermanual transfer under different viewing conditions. Exp Brain Res. 2010;202(3):543–52.
Sainburg RL, Wang J. Interlimb transfer of visuomotor rotations: independence of direction and final position information. Exp Brain Res. 2002;145(4):437–47.
Chase C, Seidler R. Degree of handedness affects intermanual transfer of skill learning. Exp Brain Res. 2008;190(3):317–28.
Taylor JA, Wojaczynski GJ, Ivry RB. Trial-by-trial analysis of intermanual transfer during visuomotor adaptation. J.Neurophysiol. [Internet]. 2011;(0022–3077 (Linking)). Available from: PM:21917998
Ghilardi M, Ghez C, Dhawan V, Moeller J, Mentis M, Nakamura T, et al. Patterns of regional brain activation associated with different forms of motor learning. Brain Res. 2000;871(1):127–45.
Krakauer JW, Ghilardi MF, Mentis M, Barnes A, Veytsman M, Eidelberg D, et al. Differential cortical and subcortical activations in learning rotations and gains for reaching: a PET study. J Neurophysiol. 2004;91(2):924–33.
Weiner MJ, Hallett M, Funkenstein HH. Adaptation to lateral displacement of vision in patients with lesions of the central nervous system. Neurology. 1983;33(6):766–72.
Martin TA, Keating JG, Goodkin HP, Bastian AJ, Thach WT. Throwing while looking through prisms. I. Focal olivocerebellar lesions impair adaptation. Brain. 1996;119:1183–98. Pt 4)(0006–8950 (Print).
Diedrichsen J, Verstynen T, Lehman SL, Ivry RB. Cerebellar involvement in anticipating the consequences of self-produced actions during bimanual movements. J Neurophysiol. 2005;93(2):801–12.
Rabe K, Livne O, Gizewski ER, Aurich V, Beck A, Timmann D, et al. Adaptation to visuomotor rotation and force field perturbation is correlated to different brain areas in patients with cerebellar degeneration. J Neurophysiol. 2009;101(4):1961–71.
Chapman HL, Eramudugolla R, Gavrilescu M, Strudwick MW, Loftus A, Cunnington R, et al. Neural mechanisms underlying spatial realignment during adaptation to optical wedge prisms. Neuropsychologia. 2010;48(9):2595–601.
Galea JM, Vazquez A, Pasricha N, de Xivry J-JO, Celnik P. Dissociating the roles of the cerebellum and motor cortex during adaptive learning: the motor cortex retains what the cerebellum learns. Cereb Cortex. 2011;21(8):1761–70.
Parlow SE, Kinsbourne M. Asymmetrical transfer of training between hands: implications for interhemispheric communication in normal brain. Brain Cogn. 1989;11(1):98–113.
Nitsche MA, Paulus W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. 2000;527:633–9. Pt 3(0022–3751 (Linking)).
Galea JM, Jayaram G, Ajagbe L, Celnik P. Modulation of cerebellar excitability by polarity-specific noninvasive direct current stimulation. J Neurosci. 2009;29(28):9115–22.
Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia. 1971;9(1):97–113.
Hadipour-Niktarash A, Lee CK, Desmond JE, Shadmehr R. Impairment of retention but not acquisition of a visuomotor skill through time-dependent disruption of primary motor cortex. J Neurosci. 2007;27(49):13413–9.
Tseng YW, Diedrichsen J, Krakauer JW, Shadmehr R, Bastian AJ. Sensory prediction errors drive cerebellum-dependent adaptation of reaching. J Neurophysiol. 2007;98(1):54–62.
Iyer MB, Mattu U, Grafman J, Lomarev M, Sato S, Wassermann EM. Safety and cognitive effect of frontal DC brain polarization in healthy individuals. Neurology. 2005;64(5):872–5.
Ferrucci R, Marceglia S, Vergari M, Cogiamanian F, Mrakic-Sposta S, Mameli F, et al. Cerebellar transcranial direct current stimulation impairs the practice-dependent proficiency increase in working memory. J Cogn Neurosci. 2008;20(9):1687–97.
Wise SP, Moody SL, Blomstrom KJ, Mitz AR. Changes in motor cortical activity during visuomotor adaptation. Exp Brain Res. 1998;121(3):285–99.
Li CS, Padoa-Schioppa C, Bizzi E. Neuronal correlates of motor performance and motor learning in the primary motor cortex of monkeys adapting to an external force field. Neuron. 2001;30(2):593–607.
Paz R, Boraud T, Natan C, Bergman H, Vaadia E. Preparatory activity in motor cortex reflects learning of local visuomotor skills. Nat Neurosci. 2003;6(8):882–90.
Mandelblat-Cerf Y, Novick I, Vaadia E. Expressions of multiple neuronal dynamics during sensorimotor learning in the motor cortex of behaving monkeys. PLoS One. 2011;6(7):e21626.
Gandiga PC, Hummel FC, Cohen LG. Transcranial DC stimulation (tDCS): a tool for double-blind sham-controlled clinical studies in brain stimulation. Clin Neurophysiol. 2006;117(4):845–50.
Krakauer JW, Ghez C, Ghilardi MF. Adaptation to visuomotor transformations: consolidation, interference, and forgetting. J Neurosci. 2005;25(2):473–8.
Malone LA, Bastian AJ. Thinking about walking: effects of conscious correction versus distraction on locomotor adaptation. J Neurophysiol. 2010;103(4):1954–62.
Zar, J.H. Power and Sample Size in Analysis of Variance. Biostatistical Analysis. 4th ed. Upper Saddle River, NJ: Prentice Hall; 1999. p. 189–95.
Cunningham HA, Welch RB. Multiple concurrent visual-motor mappings: implications for models of adaptation. J Exp Psychol Hum Percept Perform. 1994;20(5):987–99.
Imamizu H, Shimojo S. The locus of visual-motor learning at the task or manipulator level: implications from intermanual transfer. J Exp Psychol Hum Percept Perform. 1995;21(4):719–33.
Smith MA, Shadmehr R. Intact ability to learn internal models of arm dynamics in Huntington’s disease but not cerebellar degeneration. J Neurophysiol. 2005;93(5):2809–21.
Werner S, Bock O, Timmann D. The effect of cerebellar cortical degeneration on adaptive plasticity and movement control. Exp Brain Res. 2009;193(2):189–96.
Taylor HG, Heilman KM. Left-hemisphere motor dominance in righthanders. Cortex. 1980;16(4):587–603.
Anguera JA, Russell CA, Noll DC, Seidler RD. Neural correlates associated with intermanual transfer of sensorimotor adaptation. Brain Res. 2007 Dec 14;1185(0006–8993 (Linking)):136–51.
Camus M, Ragert P, Vandermeeren Y, Cohen LG. Mechanisms controlling motor output to a transfer hand after learning a sequential pinch force skill with the opposite hand. Clin Neurophysiol. 2009;120(10):1859–65.
Morton SM, Lang CE, Bastian AJ. Inter- and intra-limb generalization of adaptation during catching. Exp Brain Res. 2001;141(4):438–45.
Laszlo JI, Baguley RA, Bairstow PJ. Bilateral transfer in tapping skill in the absence of peripheral information. J Mot Behav. 1970;2(4):261–71.
Grafton ST, Hazeltine E, Ivry RB. Abstract and effector-specific representations of motor sequences identified with PET. J Neurosci. 1998;18(22):9420–8.
Nitsche MA, Paulus W. Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology. 2001;57(10):1899–901.
Fregni F, Boggio PS, Mansur CG, Wagner T, Ferreira MJ, Lima MC, et al. Transcranial direct current stimulation of the unaffected hemisphere in stroke patients. NeuroReport. 2005;16(14):1551–5.
Jayaram G, Tang B, Pallegadda R, Vasudevan EVL, Celnik P, Bastian A. Modulating locomotor adaptation with cerebellar stimulation. J Neurophysiol. 2012;107(11):2950–7.
Séverac Cauquil A, Martinez P, Ouaknine M, Tardy-Gervet MF. Orientation of the body response to galvanic stimulation as a function of the inter-vestibular imbalance. Exp Brain Res. 2000;133(4):501–5.
Deshpande N, Patla AE. Postural responses and spatial orientation to neck proprioceptive and vestibular inputs during locomotion in young and older adults. Exp Brain Res. 2005;167(3):468–74.
Malfait N, Ostry DJ. Is interlimb transfer of force-field adaptation a cognitive response to the sudden introduction of load? J Neurosci. 2004;24(37):8084–9.
Wang J, Joshi M, Lei Y. The extent of interlimb transfer following adaptation to a novel visuomotor condition does not depend on awareness of the condition. J Neurophysiol. 2011;106(1):259–64.
Lee JH, van Donkelaar P. The human dorsal premotor cortex generates on-line error corrections during sensorimotor adaptation. J Neurosci. 2006;26(12):3330–4.
Toni I, Passingham RE. Prefrontal-basal ganglia pathways are involved in the learning of arbitrary visuomotor associations: a PET study. Exp Brain Res. 1999;127(1):19–32.
Verschueren SM, Swinnen SP, Dom R, De Weerdt W. Interlimb coordination in patients with Parkinson’s disease: motor learning deficits and the importance of augmented information feedback. Exp Brain Res. 1997;113(3):497–508.
Latash ML. Mirror writing: learning, transfer, and implications for internal inverse models. J Mot Behav. 1999;31(2):107–11.
Smith MA, Ghazizadeh A, Shadmehr R. Interacting adaptive processes with different timescales underlie short-term motor learning. PLoS Biol. 2006;4(6):1035–43.
Shadmehr R, Smith MA, Krakauer JW. Error correction, sensory prediction, and adaptation in motor control. Annu Rev Neurosci. 2010;33:89–108.
de Xivry JJ O, Criscimagna-Hemminger SE, Shadmehr R. Contributions of the motor cortex to adaptive control of reaching depend on the perturbation schedule. Cerebral Cortex. 2011;21:1475–84.
Acknowledgments
This work was supported by NIH grants R21 HD 060169, R01HD053793 and T32 HD007414-16
Conflict of Interest
The authors have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Block, H., Celnik, P. Stimulating the Cerebellum Affects Visuomotor Adaptation but not Intermanual Transfer of Learning. Cerebellum 12, 781–793 (2013). https://doi.org/10.1007/s12311-013-0486-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-013-0486-7