Abstract
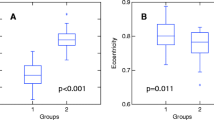
Although Friedreich’s ataxia is characterized by spinal cord atrophy, it remains to be investigated the possible correlation of such atrophy with clinical disability and genetic parameters. Thirty-three patients with Friedreich’s ataxia and 30 healthy controls underwent MRI on a 3 T scanner. We used T1-weighted 3D images to estimate spinal cord area and eccentricity at C2/C3 level based on a semi-automatic image segmentation protocol. We quantified severity of ataxia with the Friedreich ataxia rating scale (FARS). Mean cord area in Friedreich’s ataxia was smaller than in controls (38 vs 67.9 mm2, p < 0.001). In contrast, mean cord eccentricity was significantly higher in Friedreich’s ataxia when compared to the controls (0.82 vs 0.76, p < 0.001). There was a significant correlation between cord areas and the FARS scores (r = −0.53, p = 0.002). Cord damage in Friedreich’s ataxia results in atrophy combined with flattening. Cord area is associated to clinical disability and might be useful as a biomarker in the disease.


Similar content being viewed by others
References
Campuzano V, Montermini L, Moltò MD, Pianese L, Cossée M, Cavalcanti F, et al. Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science. 1996;271:1423–7.
Pandolfo M. Friedreich Ataxia. Arch Neurol. 2008;65:1296–303.
Koeppen AH. Friedreich’s ataxia: pathology, pathogenesis, and molecular genetics. J Neurol Sci. 2011;303:1–12.
Morral JA, Davis AN, Qian J, Gelman BB, Koeppen AH. Pathology and pathogenesis of sensory neuropathy in Friedreich’s ataxia. Acta Neuropathol. 2010;120:97–108.
De Michele G, Di Salle F, Filla A, D’Alessio G, Ambrosio G, Viscardi L, et al. Magnetic resonance imaging in “typical” and “late onset” Friedreich’s disease and early onset cerebellar ataxia with retained tendon reflexes. Ital J Neurol Sci. 1995;16:303–8.
Wessel K, Schroth G, Diener HC, Müller-Forell W, Dichgans J. Significance of MRI-confirmed atrophy of the cranial spinal cord in Friedreich’s ataxia. Eur Arch Psychiatry Neurol Sci. 1989;238:225–30.
Klockgether T, Petersen D, Grodd W, Dichgans J. Early onset cerebellar ataxia with retained tendon reflexes. Clinical, electrophysiological and MRI observations in comparison with Friedreich’s ataxia. Brain. 1991;114:1559–73.
Mascalchi M, Salvi F, Piacentini S, Bartolozzi C. Friedreich’s ataxia: MR findings involving the cervical portion of the spinal cord. AJR Am J Roentgenol. 1994;163:187–91.
Bonati U, Fisniku LK, Altmann DR, Yiannakas MC, Furby J, Thompson AJ, et al. Cervical cord and brain grey matter atrophy independently associate with long-term MS disability. J Neurol Neurosurg Psychiatry. 2011;82:471–2.
Bakshi R, Dandamudi VS, Neema M, De C, Bermel RA. Measurement of brain and spinal cord atrophy by magnetic resonance imaging as a tool to monitor multiple sclerosis. J Neuroimaging. 2005;15(4 Suppl):30S–45S.
Lukas C, Hahn HK, Bellenberg B, Hellwig K, Globas C, Schimrigk SK, et al. Spinal cord atrophy in spinocerebellar ataxia type 3 and 6: impact on clinical disability. J Neurol. 2008;255:1244–9.
França Jr MC, D’Abreu A, Zanardi VA, Faria AV, Nucci A, Lopes-Cendes I, et al. MRI shows dorsal lesions and spinal cord atrophy in chronic sensory neuronopathies. J Neuroimaging. 2008;18:168–72.
Filla A, De Michele G, Cavalcanti F, Pianese L, Monticelli A, Campanella G, Cocozza S. The relationship between trinucleotide (GAA) repeat length and clinical features in Friedreich ataxia. Am J Hum Genet. 1996;59:554–60.
Subramony SH, May W, Lynch D, Gomez C, Fischbeck K, Hallett M, et al. Measuring Friedreich ataxia: interrater reliability of a neurologic rating scale. Neurology. 2005;64:1261–2.
Bergo FPG (2011) SpineSeg—Semi-Automatic Spinal Cord Segmentation—Usage Guide. http://www.liv.ic.unicamp.br/∼bergo/spineseg/; access date: 12 February 2012.
Bergo FPG, Falcão AX, Miranda PAV, et al. Automatic image segmentation by tree pruning. J Math Imaging Vis. 2007;29:141–62.
Fitzgibbon A, Pilu M, Fisher RB. Direct least square fitting of ellipses. IEEE Trans Patt Anal Mach Intel. 1999;21:476–80.
Lamarche JB, Lemieux B, Lieu HB. The neuropathology of “typical” Friedreich’s ataxia in Quebec. Can J Neurol Sci. 1984;11(4 Suppl):592–600.
Chevis CF, França Jr MC, Bergo FPG, D’Abreu A, Guimarães RP, Lopes-Cendes I, et al. Distinct patterns of spinal cord atrophy in Machado-Joseph Disease and Friedreich’s ataxia. Neurology. 2011;76:A413.
Rüb U, Brunt ER, Deller T. New insights into the pathoanatomy of spinocerebellar ataxia type 3 (Machado–Joseph disease). Curr Opin Neurol. 2008;21:111–6.
Rizzo G, Tonon C, Valentino ML, Manners D, Fortuna F, Gellera C, et al. Brain diffusion-weighted imaging in Friedreich’s ataxia. Mov Disord. 2011;26:705–12.
Akhlaghi H, Corben L, Georgiou-Karistianis N, Bradshaw J, Storey E, Delatycki MB, et al. Superior cerebellar peduncle atrophy in Friedreich’s ataxia correlates with disease symptoms. Cerebellum. 2011;10:81–7.
Pagani E, Ginestroni A, Della Nave R, Agosta F, Salvi F, De Michele G, et al. Assessment of brain white matter fiber bundle atrophy in patients with Friedreich ataxia. Radiology. 2010;255:882–9.
Della Nave R, Ginestroni A, Tessa C, Salvatore E, Bartolomei I, Salvi F, et al. Brain white matter tracts degeneration in Friedreich ataxia. An in vivo MRI study using tract-based spatial statistics and voxel-based morphometry. Neuroimage. 2008;40:19–25.
Fahey MC, Corben L, Collins V, Churchyard AJ, Delatycki MB. How is disease progress in Friedreich’s ataxia best measured? A study of four rating scales. J Neurol Neurosurg Psychiatry. 2007;78:411–3.
Acknowledgments
This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP).
Conflict of Interest
The authors report no conflict of interests regarding this research. This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chevis, C.F., da Silva, C.B., D’Abreu, A. et al. Spinal Cord Atrophy Correlates with Disability in Friedreich’s Ataxia. Cerebellum 12, 43–47 (2013). https://doi.org/10.1007/s12311-012-0390-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-012-0390-6