Abstract
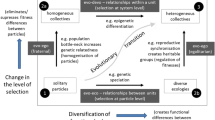
In a macroevolutionary timescale, evolvability itself evolves. Lineages are sorted based on their ability to generate adaptive novelties, which leads to the optimization of their genotype-phenotype map. The system of translation of genetic or epigenetic changes to the phenotype may reach significant horizontal and vertical complexity, and may even exhibit certain aspects of learning behaviour. This continuously evolving semiotic system probably enables the origin of complex yet functional and internally compatible adaptations. However, it also has a second, “darker”, side. As was pointed out by several authors, the same process gradually reduces the probability of the origination of significant evolutionary novelties. In a similar way to the evolution of societies, teachings, or languages, in which the growing number of internal linkages gradually solidifies their overall structure and the structure or interpretation of their constitutive elements, the evolutionary potential of lineages decreases during biological evolution. Possible adaptations become limited to small “peripheral” modifications. According to the Frozen Evolution theory, some of the proximate causes of this “macroevolutionary freezing” are more pronounced or present exclusively in sexual lineages. Sorting based on the highest (remaining) evolvability probably leads to the establishment of certain structural features of complex organisms, e.g. the modular character of their development and morphology. However, modules also “macroevolutionary freeze” whereas the hypothetical “thawing” of modules or their novel adaptive combinations becomes rarer and rarer. Some possible ways out of this dead end include the rearrangement of individual development, e.g. neoteny, radical simplification, i.e. sacculinization, and transition to a higher level of organization, e.g. symbiosis or symbiogenesis. The evolution of evolvability is essentially a biosemiotic process situated at the intersection of the genocentric modern synthesis and the evo-devo-centric extended synthesis. Therefore, evolvability may eventually connect these three not necessarily contradictory approaches.
Similar content being viewed by others
Notes
This is not to say evolutionary lineages or the whole biosphere live or learn in the same way individual organisms do. However, in order to avoid any anthropomorphic tone, it would be necessary to discuss these aspects of evolution of evolvability purely in terms of mutually responsive interactions. This would make the exposition less clear and possible analogies with individual learning less obvious. Therefore, we (as numerous authors before us) stuck with the term “learning”, which, however, should not be thought as identical to individual learning but only analogous with it some degree (see, e.g. Davies 2014; Watson et al. 2014, 2016; Lindholm 2015; or Watson and Szathmary 2016 for more detail).
It remains an open question whether all forms of learning and similar processes in living nature are based on the same principle – “domesticated” natural selection. Broad array of processes, e.g. the affinity maturation of antibodies in our immune system (Manser 1990) or “testing” of hypotheses about outside world that define Dennett’s (1995) Popperian organisms, seems to support this possibility.
FPT is, in some regards, close to older punctuational theories of evolution (for systematic review, see Flegr 2013). However, it is based on the existence of alleles with frequency dependent effects on fitness rather than on the existence of epistasis and offers the most complex scenario of related events.
References
Arthur, W. (1982). A developmental approach to the problem of variation in evolutionary rates. Biological Journal of the Linnean Society, 18(3), 243–261. https://doi.org/10.1111/j.1095-8312.1982.tb02038.x.
Arthur, W. (1984). Mechanisms of morphological evolution: A combined genetic, developmental, and ecological approach. Chichester: Wiley.
Azevedo, R., Lohaus, R., Srinivasan, S., Dang, K., & Burch, C. (2006). Sexual reproduction selects for robustness and negative epistasis in artificial gene networks. Nature, 440(7080), 87–90. https://doi.org/10.1038/nature04488.
Bonner, J. (1988). The evolution of complexity by means of natural selection. Princeton, USA: Princeton University Press.
Brigandt, I. (2015). From developmental constraint to evolvability: How concepts figure in explanation and disciplinary identity. In A. Love (Ed.), Conceptual change in biology (pp. 305–352). Dordrecht: Springer Science+Business Media.
Budd, G. (2006). On the origin and evolution of major morphological characters. Biological Reviews, 81(4), 609–628. https://doi.org/10.1017/S1464793106007135.
Budd, G., & Jackson, I. (2016). Ecological innovations in the Cambrian and the origins of the crown group phyla (article). Philosophical Transactions of the Royal Society B-Biological Sciences, 371(1685). https://doi.org/10.1098/rstb.2015.0287,20150287.
Calcott, B., & Sterelny, K. (2001). The major transitions in evolution revisited. Cambridge, USA: MIT Press.
Callebaut, W., & Rasskin-Gutman, D. (2005). Modularity: Understanding the development and evolution of natural complex systems. Cambridge, USA: MIT Press.
Canning, E., Okamura, B., Baker, J., Muller, R., & Rollinson, D. (2004). Biodiversity and evolution of the myxozoa. Advances in Parasitology, 56(56), 43–131. https://doi.org/10.1016/S0065-308X(03)56002-X.
Carroll, S. (2001). Chance and necessity: The evolution of morphological complexity and diversity. Nature, 409(6823), 1102–1109. https://doi.org/10.1038/35059227.
Carroll, S. (2005). Evolution at two levels: On genes and form. PLoS Biology, 3(7), 1159–1166. https://doi.org/10.1371/journal.pbio.0030245.
Clune, J., Mouret, J., & Lipson, H. (2013). The evolutionary origins of modularity. Proceedings of the Royal Society B-Biological Sciences, 280(1755), 20122863. https://doi.org/10.1098/rspb.2012.2863.
Darwin, C. (1859). On the origin of species by means of natural selection or the preservation of favoured races in the struggle for life. London: John Murray.
Davidson, E., & Erwin, D. (2006). Gene regulatory networks and the evolution of animal body plans. Science, 311(5762), 796–800. https://doi.org/10.1126/science.1113832.
Davies, A. (2014) On the interaction of function, constraint and complexity in evolutionary systems (Doctoral disertation). University of Southampton.
Dawkins, R. (1976). Selfish gene. Oxford: Oxford University Press.
Dawkins, R. (1989). The evolution of evolvability. In Langton (Ed.), Artificial life (Santa Fe institute studies in the sciences of complexity, Vol. VI) (pp. 201–220). Redwood City, California: Addison-Wesley.
de Beer, G. (1954). Archaeopteryx lithographica: A study based upon the British museum specimen. London: Trustees of the British Museum.
Dececchi, T., & Larsson, H. (2013). Body and limb size dissociation at the origin of birds: Uncoupling allometric constraints across a macroevolutionary transition. Evolution, 67(9), 2741–2752. https://doi.org/10.1111/evo.12150.
Dennett, D. (1995). Darwin's dangerous idea: Evolution and the meanings of life. New York, USA: Simon & Schuster.
DiMichele, W., & Bateman, R. (1996). Plant paleoecology and evolutionary inference: Two examples from the Paleozoic. Review of Palaeobotany and Palynology, 90(3–4), 223–247. https://doi.org/10.1016/0034-6667(95)00085-2.
Domes, K., Norton, R., Maraun, M., & Scheu, S. (2007). Reevolution of sexuality breaks Dollo's law. Proceedings of the National Academy of Sciences of the United States of America, 104(17), 7139–7144. https://doi.org/10.1073/pnas.0700034104.
Eble, G. (1998). The role of development in evolutionary radiations. In M. McKinney & J. Drake (Eds.), Biodiversity dynamics: Turnover of populations, taxa, and communities (pp. 132–161). New York: Columbia University Press.
Eble, G. (1999). Originations: Land and sea compared. Geobios, 32(2), 223–234. https://doi.org/10.1016/S0016-6995(99)80036-9.
Eble, G. (2005). Morphological modularity and macroevolution: Conceptual and empirical aspects. In W. Callebaut, & R.-G. D (Eds.), Modularity: Understanding the development and evolution of natural complex systems (pp. 221–238). Cambridge, London: MIT Press.
Erwin, D. (2007). Disparity: Morphological pattern and developmental context. Palaeontology, 50, 57–73. https://doi.org/10.1111/j.1475-4983.2006.00614.x.
Erwin, D., Valentine, J., & Sepkoski, J. (1987). A comparative study of diversification events: The early Paleozoic versus the Mesozoic. Evolution, 41(6), 1177–1186. https://doi.org/10.2307/2409086.
Espinosa-Soto, C. (2014). Evolution of modularity. In M. Benítez, O. Miramontes, & A. Valiente-Banuet (Eds.), Frontiers in ecology, evolution and complexity. CopIt-arXives: Mexico City, Mexico.
Flegr, J. (1998). On the "origin" of natural selection by means of speciation. Rivista Di Biologia-Biology Forum, 91(2), 291–304.
Flegr, J. (2010). Elastic, not plastic species: Frozen plasticity theory and the origin of adaptive evolution in sexually reproducing organisms. Biology Direct, 5, −. https://doi.org/10.1186/1745-6150-5-2.
Flegr, J. (2013). Microevolutionary, macroevolutionary, ecological and taxonomical implications of punctuational theories of adaptive evolution. Biology Direct, 8. https://doi.org/10.1186/1745-6150-8-1.
Flegr, J. (2015). Evoluční tání aneb O původu rodů. (On the Origin of Genera). Prague: Academia.
Foote, M. (1997). The evolution of morphological diversity. Annual Review of Ecology and Systematics, 28, 129–152. https://doi.org/10.1146/annurev.ecolsys.28.1.129.
Gerhart, J., & Kirschner, M. (2007). The theory of facilitated variation. Proceedings of the National Academy of Sciences of the United States of America, 104, 8582-8589. https://doi.org/10.1073/pnas.0701035104.
Glenner, H., & Hebsgaard, M. (2006). Phylogeny and evolution of life history strategies of the parasitic barnacles (Crustacea, Cirripedia, Rhizocephala). Molecular Phylogenetics and Evolution, 41(3), 528–538. https://doi.org/10.1016/j.ympev.2006.06.004.
Gould, S. (1989). Wonderful life: The burgess shale and the nature of history. New York, London: W. W. Norton & Company.
Haiyang, H., Masahiro, U., Song, G., Kotaro, S., Tsai-Ming, L., Fang, L., et al. (2017). Constrained vertebrate evolution by pleiotropic genes. Nature Ecology & Evolution, 1(11), 1722–1730. https://doi.org/10.1038/s41559-017-0318-0.
Hansen, T. (2003). Is modularity necessary for evolvability? Remarks on the relationship between pleiotropy and evolvability. Biosystems, 69(2–3), 83–94. https://doi.org/10.1016/S0303-2647(02)00132-6.
Hansen, T. (2016). Quantitative genetics of evolvability. In R. Kliman (Ed.), Encyclopedia of evolutionary Biology (pp. 83–89). Oxford: Elsevier Academic Press.
Hoekstra, H., & Coyne, J. (2007). The locus of evolution: Evo devo and the genetics of adaptation. Evolution, 61(5), 995–1016. https://doi.org/10.1111/j.1558-5646.2007.00105.x.
Hoffmeyer, J., & Stjernfelt, F. (2016). The great chain of Semiosis. Investigating the steps in the evolution of semiotic competence. Biosemiotics, 9(1), 7–29. https://doi.org/10.1007/s12304-015-9247-y.
Hughes, M., Gerber, S., & Wills, M. (2013). Clades reach highest morphological disparity early in their evolution. Proceedings of the National Academy of Sciences of the United States of America, 110(34), 13875–13879. https://doi.org/10.1073/pnas.1302642110.
Ikemoto, Y., & Sekiyama, K. (2014). Modular network evolution under selection for robustness to noise. Physical Review E, 89(4). https://doi.org/10.1103/PhysRevE.89.042705.
Jablonka, E., & Lamb, M. (2005). Evolution in four dimensions. Cambridge, USA: MIT Press.
Jablonski, D. (2007). Scale and hierarchy in macroevolution. Palaeontology, 50, 87–109. https://doi.org/10.1111/j.1475-4983.2006.00615.x.
Kemp, T. (2007). The concept of correlated progression as the basis of a model for the evolutionary origin of major new taxa. Proceedings of the Royal Society B-Biological Sciences, 274(1618), 1667–1673. https://doi.org/10.1098/rspb.2007.0288.
Kirschner, M., & Gerhart, J. (1998). Evolvability. Proceedings of the National Academy of Sciences of the United States of America, 95(15), 8420–8427. https://doi.org/10.1073/pnas.95.15.8420.
Kirschner, M., & Gerhart, J. (2005). The plausibility of life: Resolving Darwin's dilemma. New Haven, USA: Yale University Press.
Laland, K., Uller, T., Feldman, M., Sterelny, K., Müller, G. B., Moczek, A., Jablonka, E., Odling-Smee, J., Wray, G. A., Hoekstra, H. E., Futuyma, D. J., Lenski, R. E., Mackay, T. F. C., Schluter, D., & Strassmann, J. E. (2014). Does evolutionary theory need a rethink? Nature, 514(7521), 161–164. https://doi.org/10.1038/514161a.
Laland, K., Uller, T., Fellman, M., Sterelny, K., Muller, G., Moczek, A., et al. (2015). The extended evolutionary synthesis: Its structure, assumptions and predictions. Proceedings of the Royal Society B-Biological Sciences, 282(1813). https://doi.org/10.1098/rspb.2015.1019
Lee, M., Soubrier, J., & Edgecombe, G. (2013). Rates of phenotypic and genomic evolution during the Cambrian explosion. Current Biology, 23(19), 1889–1895. https://doi.org/10.1016/j.cub.2013.07.055.
Lewontin, R. (1978). Adaptation. Scientific American, 239(3), 212–231.
Lindholm, M. (2015). DNA dispose, but subjects decide. Learning and the extended synthesis. Biosemiotics, 8(3), 443–461. https://doi.org/10.1007/s12304-015-9242-3.
Lynch, V., & Wagner, G. (2010). Did egg-laying boas break dollo's law? Phylogenetic evidence for reversal to oviparity in sand boas (Eryx: Boidae). Evolution, 64(1), 207–216. https://doi.org/10.1111/j.1558-5646.2009.00790.x.
Lynch, M., Burger, R., Butcher, D., & Gabriel, W. (1993). The mutational meltdown in asexual populations. Journal of Heredity, 84(5), 339–344.
Manser, T. (1990). The efficiency of antibody affinity maturation: Can the rate of B-cell division be limiting? Immunology Today, 11(9), 305–309. https://doi.org/10.1016/0167-5699(90)90124-R.
Margulis, L., & Fester, R. (1991). Symbiosis as a source of evolutionary innovation: Speciation and morphogenesis. Cambridge, USA: MIT Press.
Markoš, A. (2014). Biosphere as semiosphere: Variations on Lotman. Sign System Studies, 42(4), 487–498.
Markoš, A. (2015). The birth and life of species–cultures. Biosemiotics, 9(1), 73–84. https://doi.org/10.1007/s12304-015-9252-1.
Markoš, A., & Cvrčková, F. (2013). The Meaning(s) of Information, Code … and Meaning. Biosemiotics, 6(1), 61–75. https://doi.org/10.1007/s12304-012-9155-3.
Markoš, A., & Das, P. (2016). Levels or domains of life? Biosemiotics, 9(3), 319–330. https://doi.org/10.1007/s12304-016-9271-6.
Markoš, A., & Faltýnek, D. (2011). Language metaphors of life. Biosemiotics, 4(2), 171–200. https://doi.org/10.1007/s12304-010-9097-6.
Marshall, C., & Valentine, J. (2010). The importance of preadapted genomes in the origin of the animal bodyplans and the cambrian explosion. Evolution, 64(5), 1189–1201. https://doi.org/10.1111/j.1558-5646.2009.00908.x.
Maynard Smith, J., & Szathmáry, E. (2010). The major transitions in evolution. New York: Oxford University Press Inc..
Mayr, E. (2003). The growth of biological thought: Diversity, evolution, and inheritance. In Cambridge, Massacusetts. London, UK: The Belknap Press of Harvard University Press.
McShea, D. (1996). Metazoan complexity and evolution: Is there a trend? Perspective. Evolution, 50(2), 477–492. https://doi.org/10.2307/2410824.
McShea, D. (2000). Functional complexity in organisms: Parts as proxies. Biology and Philosophy, 15(5), 641–668. https://doi.org/10.1023/A:1006695908715.
McShea, D. (2001a). The hierarchical structure of organisms: A scale and documentation of a trend in the maximum. Paleobiology, 27(2), 405–423. https://doi.org/10.1666/0094-8373(2001)027<0405:THSOOA>2.0.CO;2.
McShea, D. (2001b). The minor transitions in hierarchical evolution and the question of a directional bias. Journal of Evolutionary Biology, 14(3), 502–518. https://doi.org/10.1046/j.1420-9101.2001.00283.x.
Meyer, A., & Van de Peer, Y. (2005). From 2R to 3R: Evidence for a fish-specific genome duplication (FSGD). BioEssays, 27(9), 937–945. https://doi.org/10.1002/bies.20293.
Muller, H. (1964). The relation of recombination to mutational advance. Mutation Research, 1(1), 2–9.
Murchison, E. (2008). Clonally transmissible cancers in dogs and Tasmanian devils. Oncogene, 27, 19–30.
OED Online. (2017). "evolvability, n.". Online: Oxford University Press. www.oed.com/view/Entry/269743. Accessed 25 September 2017.
Ostdiek, G. (2011). Cast in plastic: Semiotic plasticity and the pragmatic reading of Darwin. Biosemiotics, 4(1), 69–82. https://doi.org/10.1007/s12304-010-9108-7.
Pepper, J. (2000). The evolution of modularity in genome architecture. In C. Maley, & E. Boudreau (Eds.), Artificial Life 7 Workshop Proceedings (pp. 9–12).
Pigliucci, M. (2008). Opinion - is evolvability evolvable? Nature Reviews Genetics, 9(1), 75–82. https://doi.org/10.1038/nrg2278.
Pigliucci, M. (2009). An extended synthesis for evolutionary Biology. Year in Evolutionary Biology, 2009(1168), 218–228. https://doi.org/10.1111/j.1749-6632.2009.04578.x.
Pigliucci, M., & Müller, G. (2010). Evolution: The extended synthesis. USA: MIT Press.
Queller, D. (1997). Cooperators since life began. The Quarterly Review of Biology, 72(2), 184–188.
Queller, D. (2000). Relatedness and the fraternal major transitions. Philosophical Transactions of the Royal Society of London Series B-Biological Sciences, 355(1403), 1647–1655. https://doi.org/10.1098/rstb.2000.0727.
Rasnicyn, A. (2005). Collected works in evolutionary biology (Izbrannye trudy po evolucionnoj biologii). Moscow: Tovarisevstvo naucnych izdanii KMK.
Rasskin-Gutman, D. (2005). Modularity: Jumping forms within morphospace. In W. Callebaut, & R.-G. D (Eds.), Modularity: Understanding the development and evolution of natural complex systems (pp. 207–219). Cambridge, London: MIT Press.
Riedl, R. (1977). A systems-analytical approach to macro-evolutionary phenomena. Quarterly Review of Biology, 52(4), 351–370. https://doi.org/10.1086/410123.
Riedl, R. (1978). Order in living organisms: A systems analysis of evolution. New York, USA: Wiley.
Rosa, D. (1899). La Riduzione progressiva della variabilità e i suoi rapporti coll'estinzione e coll'origine delle specie. Torino: Clausen.
Rutherford, S., & Lindquist, S. (1998). Hsp90 as a capacitor for morphological evolution. Nature, 396(6709), 336–342. https://doi.org/10.1038/24550.
Schank, J., & Wimsatt, W. (1986). (1986). Generative entrenchment and evolution. PSA: Proceedings of the Biennial Meeting of the Philosophy of Science Association Number Two: Symposia and Invited Papers, 1986, 33–60.
Schlosser, G. (2002). Modularity and the units of evolution. Theory in Biosciences, 121(1), 1–80. https://doi.org/10.1078/1431-7613-00049.
Schlosser, G. (2004). The role of modules in development and evolution. In G. Schlosser & G. Wagner (Eds.), Modularity in development and evolution (pp. 519–582). Chicago, London: The University of Chicago Press.
Schlosser, G., & Wagner, G. (2004). Modularity in development and evolution. Chicago, USA: University of Chicago Press.
Schoch, R. (2010). Riedl's burden and the body plan: Selection, constraint, and deep time. Journal of Experimental Zoology Part B-Molecular and Developmental Evolution, 314B(1), 1–10. https://doi.org/10.1002/jez.b.21300.
Sharov, A. (2014). Evolutionary constraints or opportunities? Biosystems, 123, 9–18. https://doi.org/10.1016/j.biosystems.2014.06.004.
Sharov, A. (2016). Evolutionary biosemiotics and multilevel construction networks. Biosemiotics, 9(3), 399–416. https://doi.org/10.1007/s12304-016-9269-0.
Shcherbakov, V. (2012). Stasis is an inevitable consequence of every successful evolution. Biosemiotics, 5(2), 227–245. https://doi.org/10.1007/s12304-011-9122-4.
Shcherbakov, V. (2013). Biological species as a form of existence, the higher form. In I. Pavlinov (Ed.), The species problem - ongoing issues (pp. 65–91). Rijeka, Croatia: InTech.
Simon, H. (1962). The architecture of complexity. Proceedings of the American Philosophical Society, 106(6), 467–482.
Stern, D., & Orgogozo, V. (2008). The loci of evolution: How predictable is genetic evolution ? Evolution, 62(9), 2155–2177. https://doi.org/10.1111/j.1558-5646.2008.00450.x.
Stern, D., & Orgogozo, V. (2009). Is genetic evolution predictable? Science, 323(5915), 746–751. https://doi.org/10.1126/science.1158997.
Szathmáry, E. (2015). Toward major evolutionary transitions theory 2.0. Proceedings of the National Academy of Sciences of the United States of America, 112(33), 10104–10111. https://doi.org/10.1073/pnas.1421398112.
Szathmáry, E., & Maynard Smith, J. (1995). The major evolutionary transitions. Nature, 374(6519), 227–232. https://doi.org/10.1038/374227a0.
Thomas, R. (2005). Hierarchical integration of modular structures in the evolution of animal skeletons. In W. Callebaut, & R.-G. D (Eds.), Modularity: Understanding the development and evolution of natural complex systems (pp. 239–258). Cambridge, London: MIT Press.
Toman, J., & Flegr, J. (2017). Stability-based sorting: The forgotten process behind (not only) biological evolution. Journal of Theoretical Biology, 435, 29–41.
Turney, P. (1999) 'Increasing Evolvability considered as a large-scale trend in evolution' Proceedings of the 1999 genetic and evolutionary computation conference (GECCO-99). Orlando, FL: National Research Council of Canada.
Valentine, J. (1995). Why no new phyla after the cambrian? Genome and ecospace hypotheses revisited. PALAIOS, 10(2), 190–194. https://doi.org/10.2307/3515182.
Vermeij, G. (1973). Biological versatility and earth history. Proceedings of the National Academy of Sciences of the United States of America, 70(7), 1936–1938. https://doi.org/10.1073/pnas.70.7.1936.
von Uexküll, J. (1909). Umwelt und Innenwelt der Tiere. Berlin: J. Springer.
Wagner, G. (1995). The biological role of homologues: A building block hypothesis. Neues Jahrbuch für Geologie und Paläontologie - Abhandlungen, 19, 279–288.
Wagner, A. (2005). Robustness and evolvability in living systems. Princeton, USA: University Press Princeton.
Wagner, G., & Altenberg, L. (1996). Perspective: Complex adaptations and the evolution of evolvability. Evolution, 50(3), 967–976. https://doi.org/10.2307/2410639.
Wagner, G., & Laubichler, M. (2004). Rupert Riedl and the re-synthesis of evolutionary and developmental biology: Body plans and evolvability. Journal of Experimental Zoology Part B-Molecular and Developmental Evolution, 302B(1), 92–102. https://doi.org/10.1002/jez.b.20005.
Wagner, G., Mezey, J., & Calabretta, R. (2005). Natural selection and the origin of modules. In W. Callebaut & D. Rasskin-Gutman (Eds.), Modularity: Understanding the development and evolution of natural complex systems (pp. 33–49). Cambridge, USA: MIT Press.
Wagner, G., Pavlicev, M., & Cheverud, J. (2007). The road to modularity. Nature Reviews Genetics, 8(12), 921–931. https://doi.org/10.1038/nrg2267.
Watson, R., & Szathmary, E. (2016). How can evolution learn? Trends in Ecology & Evolution, 31(2), 147–157. https://doi.org/10.1016/j.tree.2015.11.009.
Watson, R., Wagner, G., Pavlicev, M., Weinreich, D., & Mills, R. (2014). The evolution of phenotypic correlations and “developmental memory”. Evolution, 68(4), 1124–1138. https://doi.org/10.1111/evo.12337.
Watson, R., Mills, R., Buckley, C., Kouvaris, K., Jackson, A., Powers, S., et al. (2016). Evolutionary connectionism: Algorithmic principles underlying the evolution of biological organisation in Evo-Devo, Evo-eco and evolutionary transitions. Evolutionary Biology, 43(4), 553–581. https://doi.org/10.1007/s11692-015-9358-z.
Webster, M. (2007). A Cambrian peak in morphological variation within trilobite species. Science, 317(5837), 499–502. https://doi.org/10.1126/science.1142964.
West-Eberhard, M. (2003). Developmental plasticity and evolution. Oxford, UK: Oxford University Press.
Whiting, M., Bradler, S., & Maxwell, T. (2003). Loss and recovery of wings in stick insects. Nature, 421(6920), 264–267. https://doi.org/10.1038/nature01313.
Wimsatt, W. (2013). The role of generative entrenchment and robustness in the evolution of complexity. In C. Lineweaver, P. Davies, & M. Ruse (Eds.), Complexity and the arrow of time (pp. 308–331). New York, USA: Cambridge University Press.
Wimsatt, W., & Schank, J. (2004). Generative entrenchment, modularity, and evolvability: When genic selection meets the whole organism. In G. Schlosser & G. Wagner (Eds.), Modularity in development and evolution (pp. 359–394). Chicago, London: The University of Chicago Press.
Acknowledgements
We thank Charlie Lotterman for the final revisions of our text.
Funding
This work was supported by the Grant Agency of the Charles University in Prague (project no: 578416); and the Charles University Research Centre (UNCE 204004). The funding sources had no role in study design, the collection, analysis and interpretation of data, the writing of the report and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Toman, J., Flegr, J. Macroevolutionary Freezing and the Janusian Nature of Evolvability: Is the Evolution (of Profound Biological Novelty) Going to End?. Biosemiotics 11, 263–285 (2018). https://doi.org/10.1007/s12304-018-9326-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12304-018-9326-y