Abstract
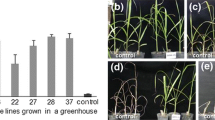
To produce high quality, glyphosate-resistant soybeans, we crossed Jinda 73 and glyphosate-resistant RR1 (Roundup Ready First Generation) (RR1) resulting in 34 hybrid strains. To determine the effects of glyphosate on soybean metabolism, we grew the two parents upto the seedling stage, and measured chlorophyll, soluble sugar, malondialdehyde (MDA), relative conductivity and proline. Then, we treated the plants with glyphosate and measured the same factors again. Results showed that the chlorophyll content of Jinda 73 and RR1 decreased after spraying glyphosate. Glyphosate increased the level of soluble sugar, MDA, relative conductivity and proline in Jinda 73, but had no significant effect on RR1. We determined glyphosate resistance of the parents and the 34 hybrid, offspring strains by documenting the growth response in the field after treatment with glyphosate. Results showed that 29 hybrid, offspring strains have complete glyphosate resistance. Polymerase chain reaction (PCR) shows that the strains which have complete resistance to glyphosate have imported the CP4 5-enolpyhruvylshikimate-3- phosphate synthase (CP4 EPSPS) gene successfully. We selected three high quality, glyphosate-resistant strains (F7-3, F7-16 and F7-21), which had higher protein and oil levels as compared with Jinda 73.



Similar content being viewed by others
References
Abraham E, Hourton-Cabassa C, Erdei L, Szabados L (2010) Methods for determination of proline in plants. Methods Mol Biol (Clifton, NJ) 639:317–331. https://doi.org/10.1007/978-1-60761-702-0_20
Anton FA, Cuadra LM, Gutierrez P, Laborda E, Laborda P (1993) Degradational behavior of the pesticides glyphosate and diflubenzuron in water. Bull Environ Contam Toxicol 51:881–888
Arregui MC, Lenardon A, Sanchez D, Maitre MI, Scotta R, Enrique S (2004) Monitoring glyphosate residues in transgenic glyphosate-resistant soybean. Pest Manag Sci 60:163–166. https://doi.org/10.1002/ps.775
Bernal J, Martin MT, Soto ME, Nozal MJ, Marotti I, Dinelli G, Bernal JL (2012) Development and application of a liquid chromatography-mass spectrometry method to evaluate the glyphosate and aminomethylphosphonic acid dissipation in maize plants after foliar treatment. J Agric Food Chem 60:4017–4025. https://doi.org/10.1021/jf3006504
Botsoglou NA, Fletouris DJ, Papageorgiou GE, Vassilopoulos VN, Mantis AJ, Trakatellis AG (1994) Rapid, sensitive, and specific thiobarbituric acid method for measuring lipid peroxidation in animal tissue, food, and feedstuff samples. J Agric Food Chem 42:1931–1937
Cerdeira AL, Duke SO (2006) The current status and environmental impacts of glyphosate-resistant crops: a review. J Environ Qual 35:1633–1658. https://doi.org/10.2134/jeq2005.0378
Chen LQ, Cheung LS, Feng L, Tanner W, Frommer WB (2015) Transport of sugars. Annu Rev Biochem 84:865–894. https://doi.org/10.1146/annurev-biochem-060614-033904
Clive J (2007) The global status of the commercialized biotechnological/genetically modified crops: 2006. Tsitol Genet 41:10–12
Coalova I, de Molina Rios, Mdel C, Chaufan G (2014) Influence of the spray adjuvant on the toxicity effects of a glyphosate formulation. Toxicology In Vitro 28:1306–1311. https://doi.org/10.1016/j.tiv.2014.06.014
Couee I, Sulmon C, Gouesbet G, El Amrani A (2006) Involvement of soluble sugars in reactive oxygen species balance and responses to oxidative stress in plants. J Exp Bot 57:449–459. https://doi.org/10.1093/jxb/erj027
Crockett PA, Bhalla PL, Lee CK, Singh MB (2000) RAPD analysis of seed purity in a commercial hybrid cabbage (Brassica oleracea var. capitata) cultivar. Genome 43:317–321
Dhir SK, Widholm JM (1992) Regeneration of transgenic soybean (Glycine max) plants from electroporated protoplasts. Plant Physiol 99:81
Dill GM (2005) Glyphosate-resistant crops: history, status and future. Pest Manag Sci 61:219–224. https://doi.org/10.1002/ps.1008
Dill GM, Cajacob CA, Padgette SR (2008) Glyphosate-resistant crops: adoption, use and future considerations. Pest Manag Sci 64:326–331. https://doi.org/10.1002/ps.1501
Dun BQ et al (2007) Reconstitution of glyphosate resistance from a split 5-enolpyruvyl shikimate-3-phosphate synthase gene in Escherichia coli and transgenic tobacco. Appl Environ Microbiol 73:7997–8000. https://doi.org/10.1128/aem.00956-07
Funke T, Yang Y, Han H, Healy-Fried M, Olesen S, Becker A, Schonbrunn E (2009) Structural basis of glyphosate resistance resulting from the double mutation Thr97 - > Ile and Pro101 - > Ser in 5-enolpyruvylshikimate-3-phosphate synthase from Escherichia coli. J Biol Chem 284:9854–9860. https://doi.org/10.1074/jbc.M809771200
Gianessi LP (2005) Economic and herbicide use impacts of glyphosate-resistant crops. Pest Manag Sci 61:241–245. https://doi.org/10.1002/ps.1013
Gimsing AL, Borggaard OK, Sestoft P (2004) Modeling the kinetics of the competitive adsorption and desorption of glyphosate and phosphate on goethite and gibbsite and in soils. Environ Sci Technol 38:1718–1722
Gomes MP et al (2014) Alteration of plant physiology by glyphosate and its by-product aminomethylphosphonic acid: an overview. J Exp Bot 65:4691–4703. https://doi.org/10.1093/jxb/eru269
Haberhauer G, Pfeiffer L, Gerzabek MH (2000) Influence of molecular structure on sorption of phenoxyalkanoic herbicides on soil and its particle size fractions. J Agric Food Chem 48:3722–3727
Healy-Fried ML, Funke T, Priestman MA, Han H, Schonbrunn E (2007) Structural basis of glyphosate tolerance resulting from mutations of Pro101 in Escherichia coli 5-enolpyruvylshikimate-3-phosphate synthase. J Biol Chem 282:32949–32955. https://doi.org/10.1074/jbc.M705624200
Jiang Y, Cao Z, Jia R, Qi H, Chen M (2012) Determination of glyphosate and aminomethylphosphonic acid in rice using hydrophilic interaction chromatography-tandem mass spectrometry. Se Pu 30:39–44
Jiao L, Wang L, Qiu Z, Wang Q, Zhou Q, Huang X (2015) Effects of bisphenol A on chlorophyll synthesis in soybean seedlings. Environ Sci Pollut Res Int 22:5877–5886. https://doi.org/10.1007/s11356-014-3764-0
Lin YH, Huang LF, Hase T, Huang HE, Feng TY (2015) Expression of plant ferredoxin-like protein (PFLP) enhances tolerance to heat stress in Arabidopsis thaliana. New Biotechnol 32:235–242. https://doi.org/10.1016/j.nbt.2014.12.001
Ow DW, DE Wet JR, Helinski DR, Howell SH, Wood KV, Deluca M (1986) Transient and stable expression of the firefly luciferase gene in plant cells and transgenic plants. Science (New York, NY) 234:856–859. https://doi.org/10.1126/science.234.4778.856
Ozturk L, Yazici A, Eker S, Gokmen O, Romheld V, Cakmak I (2008) Glyphosate inhibition of ferric reductase activity in iron deficient sunflower roots. New Phytol 177:899–906. https://doi.org/10.1111/j.1469-8137.2007.02340.x
Pessagno RC, Torres Sanchez RM, dos Santos Afonso M (2008) Glyphosate behavior at soil and mineral-water interfaces. Environ Pollut (Barking, Essex: 1987) 153:53–59. https://doi.org/10.1016/j.envpol.2007.12.025
Pipke R, Amrhein N (1988) Degradation of the phosphonate herbicide glyphosate by Arthrobacter atrocyaneus ATCC 13752. Appl Environ Microbiol 54:1293–1296
Rosa M, Prado C, Podazza G, Interdonato R, Gonzalez JA, Hilal M, Prado FE (2009) Soluble sugars–metabolism, sensing and abiotic stress: a complex network in the life of plants. Plant Signal Behav 4:388–393
Saibi W, Feki K, Ben Mahmoud R, Brini F (2015) Durum wheat dehydrin (DHN-5) confers salinity tolerance to transgenic Arabidopsis plants through the regulation of proline metabolism and ROS scavenging system. Planta 242:1187–1194. https://doi.org/10.1007/s00425-015-2351-z
Schonbrunn E, Eschenburg S, Shuttleworth WA, Schloss JV, Amrhein N, Evans JN, Kabsch W (2001) Interaction of the herbicide glyphosate with its target enzyme 5-enolpyruvylshikimate 3-phosphate synthase in atomic detail. Proc Natl Acad Sci USA 98:1376–1380. https://doi.org/10.1073/pnas.98.4.1376
Shimoda Y, Ito H, Tanaka A (2012) Conversion of chlorophyll b to chlorophyll a precedes magnesium dechelation for protection against necrosis in Arabidopsis. Plant J 72:501–511. https://doi.org/10.1111/j.1365-313X.2012.05095.x
Shinabarger DL, Braymer HD (1986) Glyphosate catabolism by Pseudomonas sp. strain PG2982. J Bacteriol 168:702–707
Smart CC, Johanning D, Muller G, Amrhein N (1985) Selective overproduction of 5-enol-pyruvylshikimic acid 3-phosphate synthase in a plant cell culture which tolerates high doses of the herbicide glyphosate. J Biol Chem 260:16338–16346
Vivancos PD et al (2011) Perturbations of amino acid metabolism associated with glyphosate-dependent inhibition of shikimic acid metabolism affect cellular redox homeostasis and alter the abundance of proteins involved in photosynthesis and photorespiration. Plant Physiol 157:256–268. https://doi.org/10.1104/pp.111.181024
Wan MT, Watts RG, Moul DJ (1989) Effects of different dilution water types on the acute toxicity to juvenile Pacific salmonids and rainbow trout of glyphosate and its formulated products. Bull Environ Contam Toxicol 43:378–385
Wang YJ, Zhou DM, Luo XS, Sun RJ, Chen HM (2004) Cadmium adsorption in montmorillonite as affected by glyphosate. J Environ Sci (China) 16:881–884
Wang CJ et al (2012) Induction of drought tolerance in cucumber plants by a consortium of three plant growth-promoting rhizobacterium strains. PLoS ONE 7:e52565. https://doi.org/10.1371/journal.pone.0052565
Xu J et al (2003) The first intron of rice EPSP synthase enhances expression of foreign gene. Sci China Ser C Life Sci 46:561–569. https://doi.org/10.1360/02yc0120
Xun F, Xie B, Liu S, Guo C (2015) Effect of plant growth-promoting bacteria (PGPR) and arbuscular mycorrhizal fungi (AMF) inoculation on oats in saline-alkali soil contaminated by petroleum to enhance phytoremediation. Environ Sci Pollut Res Int 22:598–608. https://doi.org/10.1007/s11356-014-3396-4
Yu Y, Zhou Q, He Z (2005) Effects of methamidophos and glyphosate on copper sorption-desorption behavior in soils. Sci China Ser C Life Sci 48(Suppl 1):67–75
Yuan XY, Guo PY, Zhang LG, Wang X, Zhao R, Guo X, Song X (2010) Glyphosate and post-drought rewatering on protective enzyme activities and membrane lipid peroxidation in leaves of glyphosate-resistant soybean [Glycine max (L.) Merr.] seedlings. Scientia Agricultura Sinica 43:698–705
Zelaya IA, Anderson JA, Owen MD, Landes RD (2011) Evaluation of spectrophotometric and HPLC methods for shikimic acid determination in plants: models in glyphosate-resistant and -susceptible crops. J Agric Food Chem 59:2202–2212. https://doi.org/10.1021/jf1043426
Zhang H, Li G (2005) Study on the relationships between soybean drought-resistance and physiological and ecological indicators. Chin Agric Sci Bull
Zhang Y, Zhang Y, Qu Q, Wang G, Wang C (2013) Determination of glyphosate and aminomethylphosphonic acid in soybean samples by high performance liquid chromatography using a novel fluorescent labeling reagent. Anal Methods 5:6465–6472
Zhu Y, Yin J, Li D, Wang F (2005) Quantitative PCR method for detecting glyphosate- tolerant gene transfer in soybeans. J China Agric Univ 10
Zobiole LHS, Kremer RJ, Oliveira Júnior RSD, Constantin J (2010) Glyphosate affects photosynthesis in first and second generation of glyphosate-resistant soybeans. Plant Soil 336:251–265
Acknowledgements
This work was partly supported by Beijing Outstanding Talent Training for Young Backbone Individual Projects (No. 2016000020124G049) and Dabeinong Youth Education Foundation (No. 1014115004/020).
Author information
Authors and Affiliations
Contributions
Wei-yu Li and Gui-quan Li designed the study and prepared the manuscript. Ping Lu, Hao Xie, Jing-xuan Wang, Dong-yu Guo and Xing-yu Liang conducted the experimental work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Li, Wy., Lu, P., Xie, H. et al. Effects of glyphosate on soybean metabolism in strains bred for glyphosate-resistance. Physiol Mol Biol Plants 25, 523–532 (2019). https://doi.org/10.1007/s12298-018-0597-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-018-0597-1