Abstract
Background
Cancer stem cells (CSCs) are believed to form metastases. We sought to determine whether CD70 + subpopulation in human breast cancers represents the CSCs accounting for distant metastasis.
Methods
We measured the expression levels of CD70 in breast cancer cell lines and 122 primary breast cancer samples. We characterized the functional roles of CD70 + subpopulation in distant metastasis of breast cancers.
Results
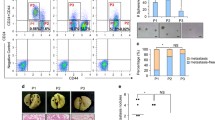
We observed a distinct pattern of CD70 expression in a panel of primary breast carcinoma samples, indicating that CD70 serves as a biomarker of lung-specific metastasis. CD70− and CD70+ cell populations isolated from breast cancer cell lines exhibited epithelial and mesenchymal phenotypes, respectively. CD70+ cells, but not CD70− cells, possessed self-renewal and differentiation potentials. Tumorsphere formation in suspension cultures and in vivo tumorigenicity were significantly greater in CD70+ cells than in CD70− cells. Furthermore, the development of lung metastases induced by orthotopic injection was markedly increased in mice inoculated with CD70 + cells. CD70 contributed to the promotion of lung metastases by enhancing self-renewal potential of CD70 + cells.
Conclusions
We isolated CSCs from primary human breast cancers and found that CD70 + subpopulations mediate lung-specific metastasis. These findings might be used to aid in selection of patients for postoperative adjuvant therapy.





Similar content being viewed by others
References
Nguyen DX, Bos PD, Massague J. Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer. 2009;9: 274–84.
Lu X, Kang Y. Organotropism of breast cancer metastasis. J Mammary Gland Biol Neoplasia. 2007;12: 153–62.
Al-Hajj M, Clarke MF. Self-renewal and solid tumor stem cells. Oncogene. 2004;23:7274–82.
Tang DG. Understanding cancer stem cell heterogeneity and plasticity. Cell Res. 2012;22: 457–72.
Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007;1: 313–23.
Pang R, Law WL, Chu AC, Poon JT, Lam CS, Chow AK. A subpopulation of CD26 + cancer stem cells with metastatic capacity in human colorectal cancer. Cell Stem Cell. 2010;6: 603–15.
Bowman MR, Crimmins MA, Yetz-Aldape J, Kriz R, Kelleher K, Herrmann S. The cloning of CD70 and its identification as the ligand for CD27. J Immunol. 1994;152:1756–61.
Goodwin RG, Alderson MR, Smith CA, Armitage RJ, VandenBos T, Jerzy R. Molecular and biological characterization of a ligand for CD27 defines a new family of cytokines with homology to tumor necrosis factor. Cell. 1993;73: 447–56.
Hintzen RQ, Lens SM, Lammers K, Kuiper H, Beckmann MP, van Lier RA. Engagement of CD27 with its ligand CD70 provides a second signal for T cell activation. J Immunol. 1995;154:2612–23.
Grewal IS. CD70 as a therapeutic target in human malignancies. Expert Opin Ther Targets. 2008;12:341–51.
Wischhusen J, Jung G, Radovanovic I, Beier C, Steinbach JP, Rimner A. Identification of CD70-mediated apoptosis of immune effector cells as a novel immune escape pathway of human glioblastoma. Cancer Res. 2002;62: 2592–9.
Riether C, Schurch CM, Buhrer ED, Hinterbrandner M, Huguenin AL, Hoepner S. CD70/CD27 signaling promotes blast stemness and is a viable therapeutic target in acute myeloid leukemia. J Exp Med l. 2017;214:359–80.
Wu Z, Wei D, Gao W, Xu Y, Hu Z, Ma Z. TPO-induced metabolic reprogramming drives liver metastasis of colorectal cancer CD110 + tumor-initiating cells. Cell Stem Cell l. 2015;17:47–59.
Minn AJ, Gupta GP, Padua D, Bos P, Nguyen DX, Nuyten D. Lung metastasis genes couple breast tumor size and metastatic spread. Proc Natl Acad Sci USA. 2007;104: 6740–5.
Song KH, Park MS, Nandu TS, Gadad S, Kim SC, Kim MY. GALNT14 promotes lung-specific breast cancer metastasis by modulating self-renewal and interaction with the lung microenvironment. Nat Commun l. 2016;7:13796.
Oskarsson T, Batlle E, Massague J. Metastatic stem cells: sources, niches, and vital pathways. Cell Stem Cell. 2014;14:306–21.
Balic M, Lin H, Young L, Hawes D, Giuliano A, McNamara G. Most early disseminated cancer cells detected in bone marrow of breast cancer patients have a putative breast cancer stem cell phenotype. Clin Cancer Res. 2006;12:5615–21. l .
Gao W, Chen L, Ma Z, Du Z, Zhao Z, Hu Z. Isolation and phenotypic characterization of colorectal cancer stem cells with organ-specific metastatic potential. Gastroenterology. 2013;145:636–46.e5.
May CD, Sphyris N, Evans KW, Werden SJ, Guo W, Mani SA. Epithelial-mesenchymal transition and cancer stem cells: a dangerously dynamic duo in breast cancer progression. Breast Cancer Res. 2011;13:202.
Tam WL, Weinberg RA. The epigenetics of epithelial-mesenchymal plasticity in cancer. Nat Med. 2013;19:1438–49.
Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3:453–8.
Gupta GP, Nguyen DX, Chiang AC, Bos PD, Kim JY, Nadal C. Mediators of vascular remodelling co-opted for sequential steps in lung metastasis. Nature. 2007;446: 765–770.
Acknowledgements
This work was supported by grants from the National Nature Science Foundation of China (81372350, 81172099, 81572886 and 81501435).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Liu, L., Yin, B., Yi, Z. et al. Breast cancer stem cells characterized by CD70 expression preferentially metastasize to the lungs. Breast Cancer 25, 706–716 (2018). https://doi.org/10.1007/s12282-018-0880-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-018-0880-6