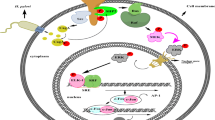
Abstract
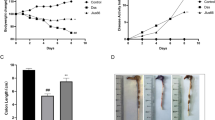
Virulence factor gamma-glutamyltransferase (GGT) of H. pylori consumes glutamine (Gln) in the stomach to decrease the tricarboxylic acid metabolite alpha-ketoglutarate (α-kg) and alter the downstream regulation of α-kg as well as cellular biological characteristics. Our previous research indicated that under H. pylori infection, mesenchymal stem cells (MSCs) migrated to the stomach and participated in gastric cancer (GC) development either by differentiating into epithelial cells or promoting angiogenesis. However, how MSCs themselves participate in H. pylori-indicated GC remains unclear. Therefore, a GGT knockout H. pylori strain (Hp-KS-1) was constructed, and downstream histone H3K9 and H3K27 methylation and the PI3K/AKT signaling pathway of α-kg were detected using Western blotting. The biological characteristics of MSCs were also examined. An additive α-kg supplement was also added to H. pylori-treated MSCs to investigate alterations in these aspects. Compared to the control and Hp-KS-1 groups, H. pylori-treated MSCs reduced Gln and α-kg, increased H3K9me3 and H3K27me3, activated the PI3K-AKT signaling pathway, and promoted the proliferation, migration, self-renewal, and pluripotency of MSCs. The addition of α-kg rescued the H. pylori-induced alterations. Injection of MSCs to nude mice resulted in the largest tumors in the H. pylori group and significantly reduced tumor sizes in the Hp-KS-1 and α-kg groups. In summary, GGT of H. pylori affected MSCs by interfering with the metabolite α-kg to increase trimethylation of histone H3K9 and H3K27, activating the PI3K/AKT signaling pathway, and promoting proliferation, migration, self-renewal, and pluripotency in tumorigenesis, elucidating the mechanisms of MSCs in GC development.
Similar content being viewed by others
References
Abraham, R.T. and Eng, C.H. 2010. A metabolic (re-)balancing act. Mol. Cell 38, 481–482.
Afarideh, M., Thaler, R., Khani, F., Tang, H., Jordan, K.L., Conley, S.M., Saadiq, I.M., Obeidat, Y., Pawar, A.S., Eirin, A., et al. 2021. Global epigenetic alterations of mesenchymal stem cells in obesity: the role of vitamin C reprogramming. Epigenetics 16, 705–717.
Allis, C.D. and Jenuwein, T. 2016. The molecular hallmarks of epigenetic control. Nat. Rev. Genet. 17, 487–500.
Altman, B.J., Stine, Z.E., and Dang, C.V. 2016. From Krebs to clinic: glutamine metabolism to cancer therapy. Nat. Rev. Cancer 16, 619–634.
Baj, J., Brzozowska, K., Forma, A., Maani, A., Sitarz, E., and Portincasa, P. 2020a. Immunological aspects of the tumor microenvironment and epithelial-mesenchymal transition in gastric carcinogenesis. Int. J. Mol. Sci. 21, 2544.
Baj, J., Korona-Głowniak, I., Forma, A., Maani, A., Sitarz, E., Rahnama-Hezavah, M., Radzikowska, E., and Portincasa, P. 2020b. Mechanisms of the epithelial-mesenchymal transition and tumor microenvironment in Helicobacter pylori-induced gastric cancer. Cells 9, 1055.
Beigier-Bompadre, M., Moos, V., Belogolova, E., Allers, K., Schneider, T., Churin, Y., Ignatius, R., Meyer, T.F., and Aebischer, T. 2011. Modulation of the CD4+ T-cell response by Helicobacter pylori depends on known virulence factors and bacterial cholesterol and cholesterol α-glucoside content. J. Infect. Dis. 204, 1339–1348.
Bradley, K.C., Finsterbusch, K., Schnepf, D., Crotta, S., Llorian, M., Davidson, S., Fuchs, S.Y., Staeheli, P., and Wack, A. 2019. Microbiota-driven tonic interferon signals in lung stromal cells protect from influenza virus infection. Cell Rep. 28, 245–256.
Carey, B.W., Finley, L.W.S., Cross, J.R., Allis, C.D., and Thompson, C.B. 2015. Intracellular α-ketoglutarate maintains the pluripotency of embryonic stem cells. Nature 518, 413–416.
Chevalier, C., Thiberge, J.M., Ferrero, R.L., and Labigne, A. 1999. Essential role of Helicobacter pylori γ-glutamyltranspeptidase for the colonization of the gastric mucosa of mice. Mol. Microbiol. 31, 1359–1372.
Chi, P., Allis, C.D., and Wang, G.G. 2010. Covalent histone modifications—miswritten, misinterpreted and mis-erased in human cancers. Nat. Rev. Cancer 10, 457–469.
Correa, P. and Houghton, J. 2007. Carcinogenesis of Helicobacter pylori. Gastroenterology 133, 659–672.
Cruzat, V., Macedo Rogero, M., Noel Keane, K., Curi, R., and Newsholme, P. 2018. Glutamine: metabolism and immune function, supplementation and clinical translation. Nutrients 10, 1564.
Curthoys, N.P. and Watford, M. 1995. Regulation of glutaminase activity and glutamine metabolism. Annu. Rev. Nutr. 15, 133–159.
Gerhard, M., Schmees, C., Voland, P., Endres, N., Sander, M., Reindl, W., Rad, R., Oelsner, M., Decker, T., Mempel, M., et al. 2005. A secreted low-molecular-weight protein from Helicobacter pylori induces cell-cycle arrest of T cells. Gastroenterology 128, 1327–1339.
Gu, H., Ji, R., Zhang, X., Wang, M., Zhu, W., Qian, H., Chen, Y., Jiang, P., and Xu, W. 2016. Exosomes derived from human mesenchymal stem cells promote gastric cancer cell growth and migration via the activation of the Akt pathway. Mol. Med. Rep. 14, 3452–3458.
Guo, J., Dai, X., Laurent, B., Zheng, N., Gan, W., Zhang, J., Guo, A., Yuan, M., Liu, P., Asara, J.M., et al. 2019. Akt methylation by SETDB1 promotes Akt kinase activity and oncogenic functions. Nat. Cell Biol. 21, 226–237.
Ha, H., Debnath, B., and Neamati, N. 2017. Role of the CXCL8-CXCR1/2 axis in cancer and inflammatory diseases. Theranostics 7, 1543–1588.
He, L., Wang, W., Shi, H., Jiang, C., Yao, H., Zhang, Y., Qian, W., and Lin, R. 2021. THBS4/integrin a2 axis mediates BM-MSCs to promote angiogenesis in gastric cancer associated with chronic Helicobacter pylori infection. Aging 13, 19375–19396.
Hemming, S., Cakouros, D., Isenmann, S., Cooper, L., Menicanin, D., Zannettino, A., and Gronthos, S. 2014. EZH2 and KDM6A act as an epigenetic switch to regulate mesenchymal stem cell lineage specification. Stem Cells 32, 802–815.
Hensley, C.T., Wasti, A.T., and DeBerardinis, R.J. 2013. Glutamine and cancer: cell biology, physiology, and clinical opportunities. J. Clin. Invest. 123, 3678–3684.
Herlofsen, S.R., Bryne, J.C., Høiby, T., Wang, L., Issner, R., Zhang, X., Coyne, M.J., Boyle, P., Gu, H., Meza-Zepeda, L.A., et al. 2013. Genome-wide map of quantified epigenetic changes during in vitro chondrogenic differentiation of primary human mesenchymal stem cells. BMC Genomics 14, 105.
Houghton, J., Stoicov, C., Nomura, S., Rogers, A.B., Carlson, J., Li, H., Cai, X., Fox, J.G., Goldenring, J.R., and Wang, T.C. 2004. Gastric cancer originating from bone marrow-derived cells. Science 306, 1568–1571.
Huang, W.C. and Hung, M.C. 2009. Induction of Akt activity by chemotherapy confers acquired resistance. J. Formos. Med. Assoc. 108, 180–194.
Huang, B., Li, G., and Jiang, X.H. 2015. Fate determination in mesenchymal stem cells: a perspective from histone-modifying enzymes. Stem Cell Res. Ther. 6, 35.
Iyer, L.M., Tahiliani, M., Rao, A., and Aravind, L. 2009. Prediction of novel families of enzymes involved in oxidative and other complex modifications of bases in nucleic acids. Cell Cycle 8, 1698–1710.
Jang, M.W., Yun, S.P., Park, J.H., Ryu, J.M., Lee, J.H., and Han, H.J. 2012. Cooperation of Epac1/Rap1/Akt and PKA in prostaglandin E2-induced proliferation of human umbilical cord blood derived mesenchymal stem cells: involvement of c-Myc and VEGF expression. J. Cell. Physiol. 227, 3756–3767.
Kaelin, W.G.Jr. 2011. Cancer and altered metabolism: potential importance of hypoxia-inducible factor and 2-oxoglutarate-dependent dioxygenases. Cold Spring Harb. Symp. Quant. Biol. 76, 335–345.
Karnoub, A.E., Dash, A.B., Vo, A.P., Sullivan, A., Brooks, M.W., Bell, G.W., Richardson, A.L., Polyak, K., Tubo, R., and Weinberg, R.A. 2007. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449, 557–563.
Khatoon, J., Rai, R.P., and Prasad, K.N. 2016. Role of Helicobacter pylori in gastric cancer: updates. World J. Gastrointest. Oncol. 8, 147–158.
Kiaris, H., Chatzistamou, I., Papavassiliou, A.G., and Schally, A.V. 2011. Growth hormone-releasing hormone: not only a neurohormone. Trends Endocrinol. Metab. 22, 311–317.
Kim, K.M., Lee, S.G., Park, M.G., Song, J.Y., Kang, H.L., Lee, W.K., Cho, M.J., Rhee, K.H., Youn, H.S., and Baik, S.C. 2007. γ-Glutamyltranspeptidase of Helicobacter pylori induces mitochondria-mediated apoptosis in AGS cells. Biochem. Biophys. Res. Commun. 355, 562–567.
Kouzarides, T. 2007. Chromatin modifications and their function. Cell 128, 693–705.
Li, Y., Guo, D., Sun, R., Chen, P., Qian, Q., and Fan, H. 2019. Methylation patterns of Lys9 and Lys27 on Histone H3 correlate with patient outcome in gastric cancer. Dig. Dis. Sci. 64, 439–446.
Liang, F., Yue, J., Wang, J., Zhang, L., Fan, R., Zhang, H., and Zhang, Q. 2015. GPCR48/LGR4 promotes tumorigenesis of prostate cancer via PI3K/Akt signaling pathway. Med. Oncol. 32, 49.
Liberti, M.V. and Locasale, J.W. 2016. The Warburg effect: how does it benefit cancer cells? Trends Biochem. Sci. 41, 211–218.
Liu, S., He, L., and Yao, K. 2018. The antioxidative function of alpha-ketoglutarate and its applications. BioMed Res. Int. 2018, 3408467.
Liu, X., Li, Z., Song, Y., Wang, R., Han, L., Wang, Q., Jiang, K., Kang, C., and Zhang, Q. 2016. AURKA induces EMT by regulating histone modification through Wnt/β-catenin and PI3K/Akt signaling pathway in gastric cancer. Oncotarget 7, 33152–33164.
Loenarz, C. and Schofield, C.J. 2008. Expanding chemical biology of 2-oxoglutarate oxygenases. Nat. Chem. Biol. 4, 152–156.
Long, M.W., Robinson, J.A., Ashcraft, E.A., and Mann, K.G. 1995. Regulation of human bone marrow-derived osteoprogenitor cells by osteogenic growth factors. J. Clin. Invest. 95, 881–887.
Martinez, S. and Hausinger, R.P. 2015. Catalytic mechanisms of Fe(II)- and 2-oxoglutarate-dependent oxygenases. J. Biol. Chem. 290, 20702–20711.
McGovern, K.J., Blanchard, T.G., Gutierrez, J.A., Czinn, S.J., Krakowka, S., and Youngman, P. 2001. γ-Glutamyltransferase is a Helicobacter pylori virulence factor but is not essential for colonization. Infect. Immun. 69, 4168–4173.
Meier, J.L. 2013. Metabolic mechanisms of epigenetic regulation. ACS Chem. Biol. 8, 2607–2621.
Meng, L., Shi, H., Wang, Z., Fan, M., Pang, S., and Lin, R. 2021. The gamma-glutamyltransferase gene of Helicobacter pylori can promote gastric carcinogenesis by activating Wnt signal pathway through upregulating TET1. Life Sci. 267, 118921.
Nan, L.P., Wang, F., Ran, D., Zhou, S.F., Liu, Y., Zhang, Z., Huang, Z.N., Wang, J.C., Feng, X.M., and Zhang, L. 2020. Naringin alleviates H2O2-induced apoptosis via the PI3K/Akt pathway in rat nucleus pulposus-derived mesenchymal stem cells. Connect. Tissue Res. 61, 554–567.
Nishikawa, G., Kawada, K., Nakagawa, J., Toda, K., Ogawa, R., Inamoto, S., Mizuno, R., Itatani, Y., and Sakai, Y. 2019. Bone marrow-derived mesenchymal stem cells promote colorectal cancer progression via CCR5. Cell Death Dis. 10, 264.
Numakura, S., Uozaki, H., Kikuchi, Y., Watabe, S., Togashi, A., and Watanabe, M. 2019. Mesenchymal stem cell marker expression in gastric cancer stroma. Anticancer Res. 39, 387–393.
Oertli, M., Noben, M., Engler, D.B., Semper, R.P., Reuter, S., Maxeiner, J., Gerhard, M., Taube, C., and Müller, A. 2013. Helicobacter pylori γ-glutamyl transpeptidase and vacuolating cytotoxin promote gastric persistence and immune tolerance. Proc. Natl. Acad. Sci. USA 110, 3047–3052.
Quante, M., Tu, S.P., Tomita, H., Gonda, T., Wang, S.S., Takashi, S., Baik, G.H., Shibata, W., Diprete, B., Betz, K.S., et al. 2011. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 19, 257–272.
Ren, J., Huang, D., Li, R., Wang, W., and Zhou, C. 2020. Control of mesenchymal stem cell biology by histone modifications. Cell Biosci. 10, 11.
Ricci, V., Giannouli, M., Romano, M., and Zarrilli, R. 2014. Helicobacter pylori gamma-glutamyl transpeptidase and its pathogenic role. World J. Gastroenterol. 20, 630–638.
Rick, F.G., Seitz, S., Schally, A.V., Szalontay, L., Krishan, A., Datz, C., Stadlmayr, A., Buchholz, S., Block, N.L., and Hohla, F. 2012. GHRH antagonist when combined with cytotoxic agents induces S-phase arrest and additive growth inhibition of human colon cancer. Cell Cycle 11, 4203–4210.
Rossi, M., Bolz, C., Revez, J., Javed, S., El-Najjar, N., Anderl, F., Hyytiäinen, H., Vuorela, P., Gerhard, M., and Hänninen, M.L. 2012. Evidence for conserved function of γ-glutamyltranspeptidase in Helicobacter genus. PLoS ONE 7, e30543.
Shao, L., Chen, Z., Soutto, M., Zhu, S., Lu, H., Romero-Gallo, J., Peek, R., Zhang, S., and El-Rifai, W. 2019. Helicobacter pylori-induced miR-135b-5p promotes cisplatin resistance in gastric cancer. FASEB J. 33, 264–274.
Shen, X., Wu, S., Zhang, J., Li, M., Xu, F., Wang, A., Lei, Y., and Zhu, G. 2020. Wild-type IDH1 affects cell migration by modulating the PI3K/AKT/mTOR pathway in primary glioblastoma cells. Mol. Med. Rep. 22, 1949–1957.
Shi, H., Qi, C., Meng, L., Yao, H., Jiang, C., Fan, M., Zhang, Q., Hou, X., and Lin, R. 2021. Bone marrow-derived mesenchymal stem cells promote Helicobacter pylori-associated gastric cancer progression by secreting thrombospondin-2. Cell Prolif. 54, e13114.
Shibayama, K., Kamachi, K., Nagata, N., Yagi, T., Nada, T., Doi, Y., Shibata, N., Yokoyama, K., Yamane, K., Kato, H., et al. 2003. A novel apoptosis-inducing protein from Helicobacter pylori. Mol. Microbiol. 47, 443–451.
Shibayama, K., Wachino, J., Arakawa, Y., Saidijam, M., Rutherford, N.G., and Henderson, P.J.F. 2007. Metabolism of glutamine and glutathione via gamma-glutamyltranspeptidase and glutamate transport in Helicobacter pylori: possible significance in the pathophysiology of the organism. Mol. Microbiol. 64, 396–406.
Song, Y., Li, Z.X., Liu, X., Wang, R., Li, L.W., and Zhang, Q. 2017. The Wnt/β-catenin and PI3K/Akt signaling pathways promote EMT in gastric cancer by epigenetic regulation via H3 lysine 27 acetylation. Tumour Biol. 39, 1–10. doi: https://doi.org/10.1177/1010428317712617.
Straussman, R., Morikawa, T., Shee, K., Barzily-Rokni, M., Qian, Z.R., Du, J., Davis, A., Mongare, M.M., Gould, J., Frederick, D.T., et al. 2012. Tumor microenvironment induces innate RAF-inhibitor resistance through HGF secretion. Nature 487, 500–504.
Sun, L., Huang, C., Zhu, M., Guo, S., Gao, Q., Wang, Q., Chen, B., Li, R., Zhao, Y., Wang, M., et al. 2020. Gastric cancer mesenchymal stem cells regulate PD-L1-CTCF enhancing cancer stem cell-like properties and tumorigenesis. Theranostics 10, 11950–11962.
Sun, L., Wang, Q., Chen, B., Zhao, Y., Shen, B., Wang, H., Xu, J., Zhu, M., Zhao, X., Xu, C., et al. 2018. Gastric cancer mesenchymal stem cells derived IL-8 induces PD-L1 expression in gastric cancer cells via STAT3/mTOR-c-Myc signal axis. Cell Death Dis. 9, 928.
Tsukada, Y., Fang, J., Erdjument-Bromage, H., Warren, M.E., Borchers, C.H., Tempst, P., and Zhang, Y. 2006. Histone demethylation by a family of JmjC domain-containing proteins. Nature 439, 811–816.
Valenzuela, M., Bravo, D., Canales, J., Sanhueza, C., Díaz, N., Almarza, O., Toledo, H., and Quest, A.F.G. 2013. Helicobacter pylori-induced loss of survivin and gastric cell viability is attributable to secreted bacterial gamma-glutamyl transpeptidase activity. J. Infect. Dis. 208, 1131–1141.
Wang, G.G. and Allis, C.D. 2009. “Misinterpretation” of a histone mark is linked to aberrant stem cells and cancer development. Cell Cycle 8, 1982–1983.
Wang, L., Bo, X., Yi, X., Xiao, X., Zheng, Q., Ma, L., and Li, B. 2020. Exosome-transferred LINC01559 promotes the progression of gastric cancer via PI3K/AKT signaling pathway. Cell Death Dis. 11, 723.
Wang, J., Hevi, S., Kurash, J.K., Lei, H., Gay, F., Bajko, J., Su, H., Sun, W., Chang, H., Xu, G., et al. 2009. The lysine demethylase LSD1 (KDM1) is required for maintenance of global DNA methylation. Nat. Genet. 41, 125–129.
Whetstine, J.R., Nottke, A., Lan, F., Huarte, M., Smolikov, S., Chen, Z., Spooner, E., Li, E., Zhang, G., Colaiacovo, M., et al. 2006. Reversal of histone lysine trimethylation by the JMJD2 family of histone demethylases. Cell 125, 467–481.
Xiao, M., Yang, H., Xu, W., Ma, S., Lin, H., Zhu, H., Liu, L., Liu, Y., Yang, C., Xu, Y., et al. 2012. Inhibition of α-KG-dependent histone and DNA demethylases by fumarate and succinate that are accumulated in mutations of FH and SDH tumor suppressors. Genes Dev. 26, 1326–1338.
Xiao, D., Zeng, L., Yao, K., Kong, X., Wu, G., and Yin, Y. 2016. The glutamine-alpha-ketoglutarate (AKG) metabolism and its nutritional implications. Amino Acids 48, 2067–2080.
Zeng, X.Q., Wang, J., and Chen, S.Y. 2017. Methylation modification in gastric cancer and approaches to targeted epigenetic therapy (Review). Int. J. Oncol. 50, 1921–1933.
Zhang, G.F., Jensen, M.V., Gray, S.M., El, K., Wang, Y., Lu, D., Becker, T.C., Campbell, J.E., and Newgard, C.B. 2021. Reductive TCA cycle metabolism fuels glutamine- and glucose-stimulated insulin secretion. Cell Metab. 33, 804–817.
Zhang, J., Zhang, J., Zhang, N., Li, T., Zhou, X., Jia, J., Liang, Y., Sun, X., and Chen, H. 2020. The effects of platelet-rich and platelet-poor plasma on biological characteristics of BM-MSCs in vitro. Anal. Cell. Pathol. 2020, 8546231.
Zheng, X.B., He, X.W., Zhang, L.J., Qin, H.B., Lin, X.T., Liu, X.H., Zhou, C., Liu, H.S., Hu, T., Cheng, H.C., et al. 2019. Bone marrow-derived CXCR4-overexpressing MSCs display increased homing to intestine and ameliorate colitis-associated tumorigenesis in mice. Gastroenterol. Rep. 7, 127–138.
Acknowledgements
This study was supported by the National Natural Science Foundation of Hubei Province (No. 2017CFA061).
Author information
Authors and Affiliations
Contributions
ZW: performed project design, most of the experiments/data analysis, and wrote the manuscript; WW and HS: assisted in project design, manuscript revision and helped in experiments; LM: helped in project design and some of the experiments/data analysis, SP, XJ, and MF: helped in some of the experiments; RL: performed the project design, supervision, and manuscript revision.
Corresponding author
Ethics declarations
All animal experiments were conducted under the approval of the Medical Ethics Committee of Wuhan Union Hospital of Huazhong University of Science and Technology (S-028.19-12-12).
Additional information
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, Z., Wang, W., Shi, H. et al. Gamma-glutamyltransferase of Helicobacter pylori alters the proliferation, migration, and pluripotency of mesenchymal stem cells by affecting metabolism and methylation status. J Microbiol. 60, 627–639 (2022). https://doi.org/10.1007/s12275-022-1575-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-022-1575-4