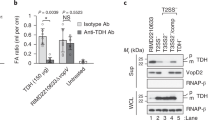
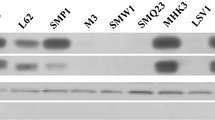
Abstract
Vibrio parahaemolyticus, a Gram-negative marine bacterial pathogen, is emerging as a major cause of food-borne illnesses worldwide due to the consumption of raw seafood leading to diseases including gastroenteritis, wound infection, and septicemia. The bacteria utilize toxins and type III secretion system (T3SS) to trigger virulence. T3SS is a multi-subunit needle-like apparatus used to deliver bacterial proteins, termed effectors, into the host cytoplasm which then target various eukaryotic signaling pathways. V. parahaemolyticus carries two T3SSs in each of its two chromosomes, named T3SS1 and T3SS2, both of which play crucial yet distinct roles during infection: T3SS1 causes cytotoxicity whereas T3SS2 is mainly associated with enterotoxicity. Each T3SS secretes a unique set of effectors that contribute to virulence by acting on different host targets and serving different functions. Emerging studies on T3SS2 of V. parahaemolyticus, reveal its regulation, translocation, discovery, characterization of its effectors, and development of animal models to understand the enterotoxicity. This review on recent findings for T3SS2 of V. parahaemolyticus highlights a novel mechanism of invasion that appears to be conserved by other marine bacteria.
Similar content being viewed by others
References
Barbieri, J.T. and Sun, J. 2004. Pseudomonas aeruginosa ExoS and ExoT. Rev. Physiol. Biochem. Pharmacol. 152, 79–92.
Begley, M., Gahan, C.G., and Hill, C. 2005. The interaction between bacteria and bile. FEMS Microbiol. Rev. 29, 625–651.
Bette-Bobillo, P., Giro, P., Sainte-Marie, J., and Vidal, M. 1998. Exoenzyme S from P. aeruginosa ADP ribosylates rab4 and inhibits transferrin recycling in SLO-permeabilized reticulocytes. Biochem. Biophys. Res. Commun. 244, 336–341.
Boutin, B.K., Townsend, S.F., Scarpino, P.V., and Twedt, R.M. 1979. Demonstration of invasiveness of Vibrio parahaemolyticus in adult rabbits by immunofluorescence. Appl. Environ. Microbiol. 37, 647–653.
Broberg, C.A., Zhang, L., Gonzalez, H., Laskowski-Arce, M.A., and Orth, K. 2010. A Vibrio effector protein is an inositol phosphatase and disrupts host cell membrane integrity. Science 329, 1660–1662.
Burdette, D.L., Seemann, J., and Orth, K. 2009. Vibrio VopQ induces PI3-kinase-independent autophagy and antagonizes phagocytosis. Mol. Microbiol. 73, 639–649.
Buttner, D. and Bonas, U. 2002. Port of entry-the type III secretion translocon. Trends Microbiol. 10, 186–192.
Coburn, J. and Gill, D.M. 1991. ADP-ribosylation of p21ras and related proteins by Pseudomonas aeruginosa exoenzyme S. Infect. Immun. 59, 4259–4262.
Fraylick, J.E., Rucks, E.A., Greene, D.M., Vincent, T.S., and Olson, J.C. 2002. Eukaryotic cell determination of ExoS ADP-ribosyltransferase substrate specificity. Biochem. Biophys. Res. Commun. 291, 91–100.
Gotoh, K., Kodama, T., Hiyoshi, H., Izutsu, K., Park, K.S., Dryselius, R., Akeda, Y., Honda, T., and Iida, T. 2010. Bile acid-induced virulence gene expression of Vibrio parahaemolyticus reveals a novel therapeutic potential for bile acid sequestrants. PLoS ONE 5, e13365.
Gupta, S. and Chowdhury, R. 1997. Bile affects production of virulence factors and motility of Vibrio cholerae. Infect. Immun. 65, 1131–1134.
Hiyoshi, H., Kodama, T., Saito, K., Gotoh, K., Matsuda, S., Akeda, Y., Honda, T., and Iida, T. 2011. VopV, an F-actin-binding type III secretion effector, is required for Vibrio parahaemolyticus-induced enterotoxicity. Cell Host Microbe 10, 401–409.
Honda, T., Ni, Y.X., Hata, A., Yoh, M., Miwatani, T., Okamoto, T., Goshima, K., Takakura, H., Tsunasawa, S., and Sakiyama, F. 1990. Properties of a hemolysin related to the thermostable direct hemolysin produced by a Kanagawa phenomenon negative, clinical isolate of Vibrio parahaemolyticus. Can. J. Microbiol. 36, 395–399.
Joseph, S.W., Colwell, R.R., and Kaper, J.B. 1982. Vibrio parahaemolyticus and related halophilic Vibrios. Crit. Rev. Microbiol. 10, 77–124.
Kaufman, M.R., Jia, J., Zeng, L., Ha, U., Chow, M., and Jin, S. 2000. Pseudomonas aeruginosa mediated apoptosis requires the ADP-ribosylating activity of exoS. Microbiology 146, 2531–2541.
Kodama, T., Gotoh, K., Hiyoshi, H., Morita, M., Izutsu, K., Akeda, Y., Park, K.S., Cantarelli, V.V., Dryselius, R., Iida, T., and et al. 2010. Two regulators of Vibrio parahaemolyticus play important roles in enterotoxicity by controlling the expression of genes in the Vp-PAI region. PLoS ONE 5, e8678.
Kodama, T., Hiyoshi, H., Gotoh, K., Akeda, Y., Matsuda, S., Park, K.S., Cantarelli, V.V., Iida, T., and Honda, T. 2008. Identification of two translocon proteins of Vibrio parahaemolyticus type III secretion system 2. Infect. Immun. 76, 4282–4289.
Kodama, T., Rokuda, M., Park, K.S., Cantarelli, V.V., Matsuda, S., Iida, T., and Honda, T. 2007. Identification and characterization of VopT, a novel ADP-ribosyltransferase effector protein secreted via the Vibrio parahaemolyticus type III secretion system 2. Cell. Microbiol. 9, 2598–2609.
Liverman, A.D., Cheng, H.C., Trosky, J.E., Leung, D.W., Yarbrough, M.L., Burdette, D.L., Rosen, M.K., and Orth, K. 2007. Arp2/3-independent assembly of actin by Vibrio type III effector VopL. Proc. Natl. Acad. Sci. USA 104, 17117–17122.
Makino, K., Oshima, K., Kurokawa, K., Yokoyama, K., Uda, T., Tagomori, K., Iijima, Y., Najima, M., Nakano, M., Yamashita, A., and et al. 2003. Genome sequence of Vibrio parahaemolyticus: a pathogenic mechanism distinct from that of V. cholerae. Lancet 361, 743–749.
Matsuda, S., Okada, N., Kodama, T., Honda, T., and Iida, T. 2012. A cytotoxic type III secretion effector of Vibrio parahaemolyticus targets vacuolar H+-ATPase subunit c and ruptures host cell lysosomes. PLoS Pathog. 8, e1002803.
Mattei, P.J., Faudry, E., Job, V., Izore, T., Attree, I., and Dessen, A. 2011. Membrane targeting and pore formation by the type III secretion system translocon. FEBS J. 278, 414–426.
McCarter, L. 1999. The multiple identities of Vibrio parahaemolyticus. J. Mol. Microbiol. Biotechnol. 1, 51–57.
McGuffie, E.M., Frank, D.W., Vincent, T.S., and Olson, J.C. 1998. Modification of Ras in eukaryotic cells by Pseudomonas aeruginosa exoenzyme S. Infect. Immun. 66, 2607–2613.
Mueller, C.A., Broz, P., and Cornelis, G.R. 2008. The type III secretion system tip complex and translocon. Mol. Microbiol. 68, 1085–1095.
Mukherjee, S., Hao, Y.H., and Orth, K. 2007. A newly discovered post-translational modification-the acetylation of serine and threonine residues. Trends Biochem. Sci. 32, 210–216.
Mukherjee, S., Keitany, G., Li, Y., Wang, Y., Ball, H.L., Goldsmith, E.J., and Orth, K. 2006. Yersinia YopJ acetylates and inhibits kinase activation by blocking phosphorylation. Science 312, 1211–1214.
Namgoong, S., Boczkowska, M., Glista, M.J., Winkelman, J.D., Rebowski, G., Kovar, D.R., and Dominguez, R. 2011. Mechanism of actin filament nucleation by Vibrio VopL and implications for tandem W domain nucleation. Nat. Struct. Mol. Biol. 18, 1060–1067.
Nishibuchi, M., Fasano, A., Russell, R.G., and Kaper, J.B. 1992. Enterotoxigenicity of Vibrio parahaemolyticus with and without genes encoding thermostable direct hemolysin. Infect. Immun. 60, 3539–3545.
Nishibuchi, M. and Kaper, J.B. 1995. Thermostable direct hemolysin gene of Vibrio parahaemolyticus: a virulence gene acquired by a marine bacterium. Infect. Immun. 63, 2093–2099.
Ohnishi, K., Nakahira, K., Unzai, S., Mayanagi, K., Hashimoto, H., Shiraki, K., Honda, T., and Yanagihara, I. 2011. Relationship between heat-induced fibrillogenicity and hemolytic activity of thermostable direct hemolysin and a related hemolysin of Vibrio parahaemolyticus. FEMS Microbiol. Lett. 318, 10–17.
Olive, A.J., Kenjale, R., Espina, M., Moore, D.S., Picking, W.L., and Picking, W.D. 2007. Bile salts stimulate recruitment of IpaB to the Shigella flexneri surface, where it colocalizes with IpaD at the tip of the type III secretion needle. Infect. Immun. 75, 2626–2629.
Orth, K., Palmer, L.E., Bao, Z.Q., Stewart, S., Rudolph, A.E., Bliska, J.B., and Dixon, J.E. 1999. Inhibition of the mitogen-activated protein kinase kinase superfamily by a Yersinia effector. Science 285, 1920–1923.
Park, K.S., Ono, T., Rokuda, M., Jang, M.H., Okada, K., Iida, T., and Honda, T. 2004. Functional characterization of two type III secretion systems of Vibrio parahaemolyticus. Infect. Immun. 72, 6659–6665.
Piñeyro, P., Zhou, X., Orfe, L.H., Friel, P.J., Lahmers, K., and Call, D.R. 2010. Development of two animal models to study the function of Vibrio parahaemolyticus type III secretion systems. Infect. Immun. 778, 4551–4559.
Prouty, A.M. and Gunn, J.S. 2000. Salmonella enterica serovar typhimurium invasion is repressed in the presence of bile. Infect. Immun. 68, 6763–6769.
Raimondi, F., Kao, J.P., Fiorentini, C., Fabbri, A., Donelli, G., Gasparini, N., Rubino, A., and Fasano, A. 2000. Enterotoxicity and cytotoxicity of Vibrio parahaemolyticus thermostable direct hemolysin in in vitro systems. Infect. Immun. 68, 3180–3185.
Ritchie, J.M., Rui, H., Zhou, X., Iida, T., Kodoma, T., Ito, S., Davis, B.M., Bronson, R.T., and Waldor, M.K. 2012. Inflammation and disintegration of intestinal villi in an experimental model for Vibrio parahaemolyticus-induced diarrhea. PLoS Pathog. 8, e1002593.
Schuhmacher, D.A. and Klose, K.E. 1999. Environmental signals modulate ToxT-dependent virulence factor expression in Vibrio cholerae. J. Bacteriol. 181, 1508–1514.
Su, Y.C. and Liu, C. 2007. Vibrio parahaemolyticus: a concern of seafood safety. Food Microbiol. 24, 549–558.
Sun, J. and Barbieri, J.T. 2003. Pseudomonas aeruginosa ExoT ADP-ribosylates CT10 regulator of kinase (Crk) proteins. J. Biol. Chem. 278, 32794–32800.
Takai, Y., Sasaki, T., and Matozaki, T. 2001. Small GTP-binding proteins. Physiol. Rev. 81, 153–208.
Trosky, J.E., Li, Y., Mukherjee, S., Keitany, G., Ball, H., and Orth, K. 2007. VopA inhibits ATP binding by acetylating the catalytic loop of MAPK kinases. J. Biol. Chem. 282, 34299–34305.
Trosky, J.E., Mukherjee, S., Burdette, D.L., Roberts, M., McCarter, L., Siegel, R.M., and Orth, K. 2004. Inhibition of MAPK signaling pathways by VopA from Vibrio parahaemolyticus. J. Biol. Chem. 279, 51953–51957.
Yarbrough, M.L., Li, Y., Kinch, L.N., Grishin, N.V., Ball, H.L., and Orth, K. 2009. AMPylation of Rho GTPases by Vibrio VopS disrupts effector binding and downstream signaling. Science 323, 269–272.
Yeung, P.S. and Boor, K.J. 2004. Epidemiology, pathogenesis, and prevention of foodborne Vibrio parahaemolyticus infections. Foodborne Pathog. Dis. 1, 74–88.
Yu, B., Cheng, H.C., Brautigam, C.A., Tomchick, D.R., and Rosen, M.K. 2011. Mechanism of actin filament nucleation by the bacterial effector VopL. Nat. Struct. Mol. Biol. 18, 1068–1074.
Zhang, L., Krachler, A.M., Broberg, C.A., Li, Y., Mirzaei, H., Gilpin, C.J., and Orth, K. 2012. Type III effector VopC mediates invasion for Vibrio species. Cell Rep. 1, 453–460.
Zhou, X., Ritchie, J.M., Hiyoshi, H., Iida, T., Davis, B.M., Waldor, M.K., and Kodama, T. 2012. The hydrophilic translocator for Vibrio parahaemolyticus, T3SS2, is also translocated. Infect. Immun. 80, 2940–2947.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ham, H., Orth, K. The role of type III secretion System 2 in Vibrio parahaemolyticus pathogenicity. J Microbiol. 50, 719–725 (2012). https://doi.org/10.1007/s12275-012-2550-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-2550-2