Abstract
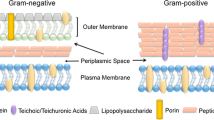
ClpP protease is essential for virulence and survival under stress conditions in several pathogenic bacteria. The clpP mutation in a murine infection model has demonstrated both attenuation of virulence and a sensitivity to hydrogen peroxide. However, the underlying mechanisms for these changes have not been resolved. Because macrophages play a major role in immune response and activated macrophages can kill microbes via oxygen-dependant mechanisms, we investigated the effect of the clpP mutation on its sensitivity to macrophage-mediated oxygen-dependant mechanisms. The clpP mutant derived from D39 (serotype 2) exhibited a higher sensitivity to oxidative stresses such as reactive oxygen intermediates, reactive nitrogen intermediates, and H2O2, but no sensitivity to osmotic stress (NaCl) and pH. Moreover, viability of the clpP mutant was significantly increased in murine macrophage cells by treatment with S-methylisothiourea sulfate, which inhibits inducible nitric oxide synthase (iNOS) activity and subsequently elicits lower level secretions of nitric oxide (NO). However, viability of wild type was unchanged. Taken together, these results indicate that ClpP is involved in the resistance to oxidative stresses after entrapment by macrophages and subsequently contributes to virulence via NO mediated pathway.
Similar content being viewed by others
References
Adamou, J.E., J.H. Heinrichs, A.L. Erwin, W. Walsh, T. Gayle, M. Dormitzer, R. Dagan, and et al. 2001. Identification and characterization of a novel family of pneumococcal proteins that are protective against sepsis. Infect. Immun. 69, 949–958.
Akira, S., S. Uematsu, and O. Takeuchi. 2006. Pathogen recognition and innate immunity. Cell 124, 783–801.
Borezée, E., E. Pellegrini, J.L. Beretti, and P. Berche. 2001. SvpA, a novel surface virulence-associated protein required for intracellular survival of Listeria monocytogenes. Microbiology 147 (Pt 11), 2913–2923.
Calandra, T. and T. Roger. 2003. Macrophage migration inhibitory factor: a regulator of innate immunity. Nat. Rev. Immunol. 3, 791–800.
Cao, J., T. Chen, D. Li, C.K. Wong, D. Chen, W. Xu, X. Zhang, C.W. Lam, and Y. Yin. 2008. Mucosal immunization with purified ClpP could elicit protective efficacy against pneumococcal pneumonia and sepsis in mice. Microbes Infect. 10, 1536–1542.
Cao, J., D. Chen, W. Xu, T. Chen, S. Xu, J. Luo, Q. Zhao, B. Liu, D. Wang, X. Zhang, Y. Shan, and Y. Yin. 2007. Enhanced protection against pneumococcal infection elicited by immunization with the combination of PspA, PspC, and ClpP. Vaccine 25, 4996–5005.
Craig, E.A., B.D. Gambill, and R.J. Nelson. 1993. Heat shock proteins: Molecular chaperones of protein biogenesis. Microbiol. Rev. 57, 402–414.
Ding, A.H., C.F. Nathan, and D.J. Stuehr. 1988. Release of reactive nitrogen intermediates and reactive oxygen intermediates from mouse peritoneal macrophages: comparison of activating cytokines and evidence for independent production. J. Immunol. 141, 2407–2412.
Frees, D., S.N. Qazi, P.J. Hill, and H. Ingmer. 2003. Alternative roles of ClpX and ClpP in Staphylococcus aureus stress tolerance and virulence. Mol. Microbiol. 48, 1565–1578.
Gaillot, O., S. Bregenholt, F. Jaubert, J.P. Di Santo, and P. Berche. 2001. Stress-induced ClpP serine protease of Listeria monocytogenes is essential for induction of listeriolysin O-dependent protective immunity. Infect. Immun. 69, 4938–4943.
Hava, D.L. and A. Camilli. 2002. Large-scale identification of serotype 4 Streptococcus pneumoniae virulence factors. Mol. Microbiol. 45, 1389–1406.
Kerr, A.R., X.Q. Wei, P.W. Andrew, and T.J. Mitchell. 2004. Nitric oxide exerts distinct effects in local and systemic infections with Streptococcus pneumoniae. Microb. Pathog. 36, 303–310.
Knapp, S., J.C. Leemans, S. Florquin, J. Branger, N.A. Maris, J. Pater, N. van Rooijen, and T. van der Poll. 2003. Alveolar macrophages have a protective antiinflammatory role during murine pneumococcal pneumonia. Am. J. Respir. Crit. Care Med. 167, 171–179.
Ko, K.S., S. Park, W.S. Oh, J.Y. Suh, T. Oh, S. Ahn, J. Chun, and J.H. Song. 2006. Comparative analysis of growth-phase-dependent gene expression in virulent and avirulent Streptococcus pneumoniae using a high-density DNA microarray. Mol. Cells 21, 82–88.
Kwon, H.Y., S.W. Kim, M.H. Choi, A.D. Ogunniyi, J.C. Paton, S.H. Park, S.N. Pyo, and D.K. Rhee. 2003. Effect of heat shock and mutations in ClpL and ClpP on virulence gene expression in Streptococcus pneumoniae. Infect. Immun. 71, 3757–3765.
Kwon, H.Y., A.D. Ogunniyi, M.H. Choi, S.N. Pyo, D.K. Rhee, and J.C. Paton. 2004. The ClpP protease of Streptococcus pneumoniae modulates virulence gene expression and protects against fatal pneumococcal challenge. Infect. Immun. 72, 5646–5653.
Lee, M.R., S.M. Bae, T.S. Kim, and K.J. Lee. 2006. Proteomic analysis of protein expression in Streptococcus pneumoniae in response to temperature shift. J. Microbiol. 44, 375–382.
MacMicking, J., Q.W. Xie, and C. Nathan. 1997. Nitric oxide and macrophage function. Annu. Rev. Immunol. 15, 323–350.
Mandell, L.A., R.G. Wunderink, A. Anzueto, J.G. Bartlett, G.D. Campbell, N.C. Dean, S.F. Dowell, and et al. 2007. Infectious Diseases Society of America; American Thoracic Society. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin. Infect. Dis. 44, 27–72.
Marriott, H.M., F. Ali, R.C. Read, T.J. Mitchell, M.K. Whyte, and D.H. Dockrell. 2004. Nitric oxide levels regulate macrophage commitment to apoptosis or necrosis during pneumococcal infection. FASEB J. 18, 1126–1128.
Nathan, C.F. 1987. Neutrophil activation on biological surfaces. Massive secretion of hydrogen peroxide in response to products of macrophages and lymphocytes. J. Clin. Invest. 80, 1550–1560.
Oggioni, M.R., C. Trappetti, A. Kadioglu, M. Cassone, F. Iannelli, S. Ricci, P.W. Andrew, and G. Pozzi. 2006. Switch from planktonic to sessile life: a major event in pneumococcal pathogenesis. Mol. Microbiol. 61, 1196–1210.
Orihuela, C.J., G. Gao, K.P. Francis, J. Yu, and E.I. Tuomanen. 2004. Tissue-specific contributions of pneumococcal virulence factors to pathogenesis. J. Infect. Dis. 190, 1661–1669.
Orihuela, C.J., G. Gao, M. McGee, J. Yu, K.P. Francis, and E. Tuomanen. 2003. Organ-specific models of Streptococcus pneumoniae disease. Scand. J. Infect. Dis. 35, 647–652.
Paton, J.C. and P. Giammarinaro. 2001. Genome-based analysis of pneumococcal virulence factors: the quest for novel vaccine antigens and drug targets. Trends Microbiol. 9, 515–518.
Polissi, A., A. Pontiggia, G. Feger, M. Altieri, H. Mottl, L. Ferrari, and D. Simon. 1998. Large-scale identification of virulence genes from Streptococcus pneumoniae. Infect. Immun. 66, 5620–5629.
Robertson, G.T., W.L. Ng, J. Foley, R. Gilmour, and W.E. Winkler. 2002. Global transcriptional analysis of clpP mutations of type 2 Streptococcus pneumoniae and their effects on physiology and virulence. J. Bacteriol. 184, 3508–3520.
Robertson, G.T., W.L. Ng, J. Foley, and M.E. Winkler. 2003. Essentiality of clpX, but not clpP, clpL, clpC, or clpE, in Streptococcus pneumoniae R6. J. Bacteriol. 185, 2961–2966.
Szabó, C., G.J. Southan, and C. Thiemermann. 1994. Beneficial effects and improved survival in rodent models of septic shock with S-methylisothiourea sulfate, a potent and selective inhibitor of inducible nitric oxide synthase. Proc. Natl. Acad. Sci. USA 91, 12472–12476.
Thomsen, L.E., J.E. Olsen, J.W. Foster, and H. Ingmer. 2002. ClpP is involved in the stress response and degradation of misfolded proteins in Salmonella enterica serovar Typhimurium. Microbiology 148, 2727–2733.
Throup, J.P., K.K. Koretke, A.P. Bryant, K.A. Ingraham, A.F. Chalker, Y. Ge, A. Marra, and et al. 2000. A genomic analysis of two-component signal transduction in Streptococcus pneumoniae. Mol. Microbiol. 35, 566–576.
Tu, L.N., H.Y. Jeong, H.Y. Kwon, A.D. Ogunniyi, J.C. Paton, S.N. Pyo, and D.K. Rhee. 2007. Modulation of adherence, invasion, and tumor necrosis factor alpha secretion during the early stages of infection by Streptococcus pneumoniae ClpL. Infect. Immun. 75, 2996–3005.
Wizemann, T.M., J.H. Heinrichs, J.E. Adamou, A.L. Erwin, C. Kunsch, G.H. Choi, S.C. Barash, and et al. 2001. Use of a whole genome approach to identify vaccine molecules affording protection against Streptococcus pneumoniae infection. Infect. Immun. 69, 1593–1598.
Wright, R.M., L.A. Ginger, N. Kosila, N.D. Elkins, B. Essary, J.L. McManaman, and J.E. Repine. 2004. Mononuclear phagocyte xanthine oxidoreductase contributes to cytokine-induced acute lung injury. Am. J. Respir. Cell Mol. Biol. 30, 479–490.
Yamamoto, T., H. Sashinami, A. Takaya, T. Tomoyasu, H. Matsui, Y. Kikuchi, T. Hanawa, S. Kamiya, and A. Nakane. 2001. Disruption of the genes for ClpXP protease in Salmonella enterica serovar Typhimurium results in persistent infection in mice, and development of persistence requires endogenous gamma interferon and tumor necrosis factor alpha. Infect. Immun. 69, 3164–3174.
Yu, A.Y. and W.A. Houry. 2007. ClpP: a distinctive family of cylindrical energy-dependent serine proteases. FEBS Lett. 581, 3749–3757.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Park, CY., Kim, EH., Choi, SY. et al. Virulence attenuation of Streptococcus pneumoniae clpP mutant by sensitivity to oxidative stress in macrophages via an NO-mediated pathway. J Microbiol. 48, 229–235 (2010). https://doi.org/10.1007/s12275-010-9300-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-010-9300-0