Abstract
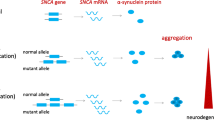
Alpha-synuclein (α-syn) deposition in Lewy bodies (LB) is one of the main neuropathological hallmarks of Parkinson’s disease (PD). LB accumulation is considered a causative factor of PD, which suggests that strategies aimed at reducing α-syn levels could be relevant for its treatment. In the present study, we developed novel nanocarriers suitable for systemic delivery of small interfering ribonucleic acid (siRNA) that were specifically designed to reduce neuronal α-syn by RNA interference. Anionic liposomes loaded with an siRNA–protamine complex for α-syn gene silencing and decorated with a rabies virus glycoprotein (RVG)-derived peptide as a targeting agent were prepared. The nanoparticles were characterized for their ability to load, protect, and deliver the functional siRNA to mouse primary hippocampal and cortical neurons as well as their efficiency to induce gene silencing in these cells. Moreover, the nanocarriers were evaluated for their stability in serum. The RVG-decorated liposomes displayed suitable characteristics for future in vivo applications and successfully induced α-syn gene silencing in primary neurons without altering cell viability. Collectively, our results indicate that RVG-decorated liposomes may be an ideal tool for further studies aimed at achieving efficient in vivo α-syn gene silencing in mouse models of PD.

Similar content being viewed by others
Change history
28 June 2017
The name of the fourth author in the original version of this article was unfortunately wrongly written on the first page.
References
Bendor, J. T.; Logan, T. P.; Edwards, R. H. The function of α-synuclein. Neuron 2013, 79, 1044–1066.
McLean, P. J.; Kawamata, H.; Ribich, S.; Hyman, B. T. Membrane association and protein conformation of α-synuclein in intact neurons. Effect of parkinson’s disease-linked mutations. J. Biol. Chem. 2000, 275, 8812–8816.
Lashuel, H. A.; Overk, C. R.; Oueslati, A.; Masliah, E. The many faces of α-synuclein: From structure and toxicity to therapeutic target. Nat. Rev. Neurosci. 2013, 14, 38–48.
Bellucci, A.; Mercuri, N. B.; Venneri, A.; Faustini, G.; Longhena, F.; Pizzi, M.; Missale, C.; Spano, P. Parkinson’s disease: From synaptic loss to connectome dysfunction. Neuropathol. Appl. Neurobiol. 2016, 42, 77–94.
Bellucci, A.; Zaltieri, M.; Navarria, L.; Grigoletto, J.; Missale, C.; Spano, P. From α-synuclein to synaptic dysfunctions: New insights into the pathophysiology of Parkinson’s disease. Brain Res. 2012, 1476, 183–202.
Maraganore, D. M. Rationale for therapeutic silencing of alpha-synuclein in Parkinson’s disease. J. Mov. Disord. 2011, 4, 1–7.
Specht, C. G.; Schoepfer, R. Deletion of the alpha-synuclein locus in a subpopulation of C57BL/6J inbred mice. BMC Neurosci. 2001, 2, 11.
Spillantini, M. G.; Crowther, R. A.; Jakes, R.; Hasegawa, M.; Goedert, M. α-synuclein in filamentous inclusions of Lewy bodies from Parkinson’s disease and dementia with Lewy bodies. Proc. Natl. Acad. Sci. USA 1998, 95, 6469–6473.
Braak, H.; DelTredici, K.; Rü b, U.; de Vos, R. A.; Jansen Steur, E. N.; Braak, E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol. Aging 2003, 24, 197–211.
Eslamboli, A.; Romero Ramos, M.; Burger, C.; Bjorklund, T.; Muzyczka, N.; Mandel, R. J.; Baker, H.; Ridley, R. M.; Kirik, D.Long-term consequences of human alpha-synuclein overexpression in the primate ventral midbrain. Brain 2007, 130, 799–815.
Uversky, V. N. Neuropathology, biochemistry, and biophysics of α-synuclein aggregation. J. Neurochem. 2007, 103, 17–37.
Sapru, M. K.; Yates, J. W.; Hogan, S.; Jiang, L. X.; Halter, J.; Bohn, M. C. Silencing of human α-synuclein in vitro and in rat brain using lentiviral-mediated RNAi. Exp. Neurol. 2006, 198, 382–390.
O’Mahony, A. M.; Godinho, B. M. D. C.; Cryan, J. F.; O’Driscoll, C. M. Non-viral nanosystems for gene and small interfering RNA delivery to the central nervous system: Formulating the solution. J. Pharm. Sci. 2013, 102, 3469–3484.
Lewis, J.; Melrose, H.; Bumcrot, D.; Hope, A.; Zehr, C.; Lincoln, S.; Braithwaite, A.; He, Z.; Ogholikhan, S.; Hinkle, K. et al. In vivo silencing of alpha-synuclein using naked siRNA. Mol. Neurodegener. 2008, 3, 19.
McCormack, A. L.; Mak, S. K.; Henderson, J. M.; Bumcrot, D.; Farrer, M. J.; Di Monte, D. A. α-synuclein suppression by targeted small interfering RNA in the primate substantia nigra. PLoS One 2010, 5, e12122.
Gorbatyuk, O. S.; Li, S. D.; Nash, K.; Gorbatyuk, M.; Lewin, A. S.; Sullivan, L. F.; Mandel, R. J.; Chen, W. J.; Meyers, C.; Manfredsson, F. P. et al. In vivo RNAi-mediated α-synuclein silencing induces nigrostriatal degeneration. Mol. Ther. 2010, 18, 1450–1457.
Nayak, S.; Herzog, R. W. Progress and prospects: Immune responses to viral vectors. Gene Ther. 2010, 17, 295–304.
Li, C. X.; Parker, A.; Menocal, E.; Xiang, S. L.; Borodyansky, L.; Fruehauf, J. H. Delivery of RNA interference. Cell Cycle 2006, 5, 2103–2109.
Haussecker, D. Current issues of RNAi therapeutics delivery and development. J. Control. Release 2014, 195, 49–54.
Grimm, D. Small silencing RNAs: State-of-the-art. Adv. Drug Deliv. Rev. 2009, 61, 672–703.
Yang, J.; Liu, H. M.; Zhang, X. Design, preparation and application of nucleic acid delivery carriers. Biotechnol. Adv. 2014, 32, 804–817.
David, S.; Pitard, B.; Benoît, J. P.; Passirani, C. Non-viral nanosystems for systemic siRNA delivery. Pharmacol. Res. 2010, 62, 100–114.
Cooper, J. M.; Wiklander, P. B. O.; Nordin, J. Z.; Al Shawi, R.; Wood, M. J.; Vithlani, M.; Schapira, A. H. V.; Simons, J. P.; El Andaloussi, S.; Alvarez Erviti, L. Systemic exosomal siRNA delivery reduced alpha-synuclein aggregates in brains of transgenic mice. Mov. Disord. 2014, 29, 1476–1485.
Alvarez Erviti, L.; Seow, Y.; Yin, H. F.; Betts, C.; Lakhal, S.; Wood, M. J. A. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011, 29, 341–345.
Kumar, P.; Wu, H. Q.; McBride, J. L.; Jung, K. E.; Kim, M. H.; Davidson, B. L.; Lee, S. K.; Shankar, P.; Manjunath, N. Transvascular delivery of small interfering RNA to the central nervous system. Nature 2007, 448, 39–43.
Robbins, P. D.; Morelli, A. E. Regulation of immune responses by extracellular vesicles. Nat. Rev. Immunol. 2014, 14, 195–208.
Vader, P.; Mol, E. A.; Pasterkamp, G.; Schiffelers, R. M. Extracellular vesicles for drug delivery. Adv. Drug Deliv. Rev. 2016, 106, 148–156.
De Luca, M. A.; Lai, F.; Corrias, F.; Caboni, P.; Bimpisidis, Z.; Maccioni, E.; Fadda, A. M.; Di Chiara, G. Lactoferrin- and antitransferrin-modified liposomes for brain targeting of the NK3 receptor agonist senktide: Preparation and in vivo evaluation. Int. J. Pharm. 2015, 479, 129–137.
Ozpolat, B.; Sood, A. K.; Lopez Berestein, G. Liposomal siRNA nanocarriers for cancer therapy. Adv. Drug Deliv. Rev. 2014, 66, 110–116.
Li, W. J.; Szoka, F. C. Lipid-based nanoparticles for nucleic acid delivery. Pharm. Res. 2007, 24, 438–449.
Buyens, K.; Demeester, J.; De Smedt, S. S.; Sanders, N. N. Elucidating the encapsulation of short interfering RNA in PEGylated cationic liposomes. Langmuir 2009, 25, 4886–4891.
Huo, H.; Gao, Y. K.; Wang, Y.; Zhang, J. H.; Wang, Z. Y.; Jiang, T. Y.; Wang, S. L. Polyion complex micelles composed of pegylated polyasparthydrazide derivatives for siRNA delivery to the brain. J. Colloid Interface Sci. 2015, 447, 8–15.
Hamidi, M.; Azadi, A.; Rafiei, P. Pharmacokinetic consequences of pegylation. Drug Deliv. 2006, 13, 399–409.
Lafon, M. Rabies virus receptors. J. Neurovirol. 2005, 11, 82–87.
Bauer, M.; Kristensen, B. W.; Meyer, M.; Gasser, T.; Widmer, H. R.; Zimmer, J.; Ueffing, M. Toxic effects of lipid-mediated gene transfer in ventral mesencephalic explant cultures. Basic Clin. Pharmacol. Toxicol. 2006, 98, 395–400.
Zaltieri, M.; Grigoletto, J.; Longhena, F.; Navarria, L.; Favero, G.; Castrezzati, S.; Colivicchi, M. A.; Della Corte, L.; Rezzani, R.; Pizzi, M. et al. α-synuclein and synapsin III cooperatively regulate synaptic function in dopamine neurons. J. Cell Sci. 2015, 128, 2231–2243.
Acknowledgements
The authors gratefully acknowledge Micaela Morelli for supporting the establishment of the collaboration between the groups participating to this study. M. S. thanks Angela Corona for fruitful discussions about the design and implementation of the project. A. B. is grateful to “Ambrosini Arredamenti SNC” for funding support within the project “Molecular Mechanisms, associated with Neurodegenerative Diseases” and the Italian Ministry of Education, University and Scientific Research—University of Brescia Ex 60% Research Funds.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at https://doi.org/10.1007/s12274-017-1726-9.
Rights and permissions
About this article
Cite this article
Schlich, M., Longhena, F., Faustini, G. et al. Anionic liposomes for small interfering ribonucleic acid (siRNA) delivery to primary neuronal cells: Evaluation of alpha-synuclein knockdown efficacy. Nano Res. 10, 3496–3508 (2017). https://doi.org/10.1007/s12274-017-1561-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1561-z