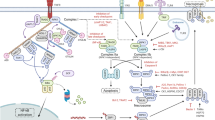
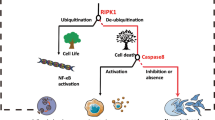
Abstract
Necroptosis is an active and well-orchestrated necrosis, distinctive from apoptosis in microscopic structure, and biochemical and molecular features. Unlike apoptosis-undergoing cells, which are removed by macrophage or neighboring cells, necrotic cell death releases danger signals and provokes inflammation, and further a severe damage to neighbor tissue. A regulated necrosis, termed as necroptosis or programmed necrosis, is emerging as a new paradigm of cell death that can be activated when apoptotic machinery is genetically or pathogenically defective. It plays biological significances in pathogenesis of a variety of inflammatory diseases as well as in a beneficial innate immune defense mechanism. This review highlights the identification of hits against necroptosis, and comprehensive approaches to discovery of small molecules that regulate necroptotic cell death. Also, the signaling molecular mechanism of necroptosis and future clinical uses of necroptosis inhibitor will be described in brief.



Similar content being viewed by others
References
Benedict, C.A., P.S. Norris, and C.F. Ware. 2002. To kill or be killed: Viral evasion of apoptosis. Nature Immunology 3: 1013–1018.
Breckenridge, D.G., B.H. Kang, D. Kokel, S. Mitani, L.A. Staehelin, and D. Xue. 2008. Caenorhabditis elegans drp-1 and fis-2 regulate distinct cell-death execution pathways downstream of ced-3 and independent of ced-9. Molecular Cell 31: 586–597.
Cheng, G., R.H. Kong, L.M. Zhang, and J.N. Zhang. 2012. Mitochondria in traumatic brain injury and mitochondrial-targeted multipotential therapeutic strategies. British Journal of Pharmacology 167: 699–719.
Cho, Y.S., S. Challa, D. Moquin, R. Genga, T.D. Ray, M. Guildford, and F.K. Chan. 2009. Phosphorylation-driven assembly of the RIP1–RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137: 1112–1123.
Cho, Y.S., S.Y. Park, H.S. Shin, and F.K. Chan. 2010. Physiological consequences of programmed necrosis, an alternative form of cell demise. Molecules and Cells 29: 327–332.
Dantzer, F., J.C. Ame, V. Schreiber, J. Nakamura, J. Menissier-De Murcia, and G. De Murcia. 2006. Poly(ADP-ribose) polymerase-1 activation during DNA damage and repair. Methods in Enzymology 409: 493–510.
Degterev, A., J. Hitomi, M. Germscheid, I.L. Ch’en, O. Korkina, X. Teng, D. Abbott, G.D. Cuny, C. Yuan, G. Wagner, S.M. Hedrick, S.A. Gerber, A. Lugovskoy, and J. Yuan. 2008. Identification of RIP1 kinase as a specific cellular target of necrostatins. Nature Chemical Biology 4: 313–321.
Degterev, A., Z. Huang, M. Boyce, Y. Li, P. Jagtap, N. Mizushima, G.D. Cuny, T.J. Mitchison, M.A. Moskowitz, and J. Yuan. 2005. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nature Chemical Biology 1: 112–119.
Frank, S., B. Gaume, E.S. Bergmann-Leitner, W.W. Leitner, E.G. Robert, F. Catez, C.L. Smith, and R.J. Youle. 2001. The role of dynamin-related protein 1, a mediator of mitochondrial fission, in apoptosis. Developmental Cell 1: 515–525.
Gerl, R., and D.L. Vaux. 2005. Apoptosis in the development and treatment of cancer. Carcinogenesis 26: 263–270.
Grimm, S., B.Z. Stanger, and P. Leder. 1996. RIP and FADD: Two “death domain”-containing proteins can induce apoptosis by convergent, but dissociable, pathways. Proceedings of the National Academy of Sciences of the United States of America 93: 10923–10927.
Gunther, C., E. Martini, N. Wittkopf, K. Amann, B. Weigmann, H. Neumann, M.J. Waldner, S.M. Hedrick, S. Tenzer, M.F. Neurath, and C. Becker. 2011. Caspase-8 regulates TNF-alpha-induced epithelial necroptosis and terminal ileitis. Nature 477: 335–339.
Hanahan, D., and R.A. Weinberg. 2011. Hallmarks of cancer: The next generation. Cell 144: 646–674.
Hitomi, J., D.E. Christofferson, A. Ng, J. Yao, A. Degterev, R.J. Xavier, and J. Yuan. 2008. Identification of a molecular signaling network that regulates a cellular necrotic cell death pathway. Cell 135: 1311–1323.
Holler, N., R. Zaru, O. Micheau, M. Thome, A. Attinger, S. Valitutti, J.L. Bodmer, P. Schneider, B. Seed, and J. Tschopp. 2000. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nature Immunology 1: 489–495.
Iyer, S.S., W.P. Pulskens, J.J. Sadler, L.M. Butter, G.J. Teske, T.K. Ulland, S.C. Eisenbarth, S. Florquin, R.A. Flavell, J.C. Leemans, and F.S. Sutterwala. 2009. Necrotic cells trigger a sterile inflammatory response through the Nlrp3 inflammasome. Proceedings of the National Academy of Sciences of the United States of America 106: 20388–20393.
Kennedy, C.L., D.J. Smith, D. Lyras, A. Chakravorty, and J.I. Rood. 2009. Programmed cellular necrosis mediated by the pore-forming alpha-toxin from Clostridium septicum. PLoS Pathogens 5: e1000516.
Kroemer, G., L. Galluzzi, and C. Brenner. 2007. Mitochondrial membrane permeabilization in cell death. Physiological Reviews 87: 99–163.
Krumschnabel, G., H.L. Ebner, M.W. Hess, and A. Villunger. 2010. Apoptosis and necroptosis are induced in rainbow trout cell lines exposed to cadmium. Aquatic Toxicology 99: 73–85.
Li, Y., X. Yang, C. Ma, J. Qiao, and C. Zhang. 2008. Necroptosis contributes to the NMDA-induced excitotoxicity in rat’s cultured cortical neurons. Neuroscience Letters 447: 120–123.
Lo, S.C., and M. Hannink. 2006. PGAM5, a Bcl-XL-interacting protein, is a novel substrate for the redox-regulated Keap1-dependent ubiquitin ligase complex. Journal of Biological Chemistry 281: 37893–37903.
Los, M., M. Mozoluk, D. Ferrari, A. Stepczynska, C. Stroh, A. Renz, Z. Herceg, Z.Q. Wang, and K. Schulze-Osthoff. 2002. Activation and caspase-mediated inhibition of PARP: A molecular switch between fibroblast necrosis and apoptosis in death receptor signaling. Molecular Biology of the Cell 13: 978–988.
Luo, X., I. Budihardjo, H. Zou, C. Slaughter, and X. Wang. 1998. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94: 481–490.
Moquin, D.M., T. Mcquade, and F.K. Chan. 2013. CYLD deubiquitinates RIP1 in the TNFalpha-induced necrosome to facilitate kinase activation and programmed necrosis. PLoS ONE 8: e76841.
Narayan, N., I.H. Lee, R. Borenstein, J. Sun, R. Wong, G. Tong, M.M. Fergusson, J. Liu, I.I. Rovira, H.L. Cheng, G. Wang, M. Gucek, D. Lombard, F.W. Alt, M.N. Sack, E. Murphy, L. Cao, and T. Finkel. 2012. The NAD-dependent deacetylase SIRT2 is required for programmed necrosis. Nature 492: 199–204.
Otera, H., and K. Mihara. 2011. Molecular mechanisms and physiologic functions of mitochondrial dynamics. Journal of Biochemistry 149: 241–251.
Park, S., H. Shin, and Y. Cho. 2013. Shikonin induces programmed necrosis-like cell death through the formation of receptor interacting protein 1 and 3 complex. Food and Chemical Toxicology 55: 36–41.
Petit, F., D. Arnoult, L. Viollet, and J. Estaquier. 2003. Intrinsic and extrinsic pathways signaling during HIV-1 mediated cell death. Biochimie 85: 795–811.
Qinli, Z., L. Meiqing, J. Xia, X. Li, G. Weili, J. Xiuliang, J. Junwei, Y. Hailan, Z. Ce, and N. Qiao. 2013. Necrostatin-1 inhibits the degeneration of neural cells induced by aluminum exposure. Restorative neurology and neuroscience 31: 543–555.
Rangamani, P., and L. Sirovich. 2007. Survival and apoptotic pathways initiated by TNF-alpha: Modeling and predictions. Biotechnology and Bioengineering 97: 1216–1229.
Robinson, N., S. Mccomb, R. Mulligan, R. Dudani, L. Krishnan, and S. Sad. 2012. Type I interferon induces necroptosis in macrophages during infection with Salmonella enterica serovar Typhimurium. Nature Immunology 13: 954–962.
Scholz, C., T. Wieder, L. Starck, F. Essmann, K. Schulze-Osthoff, B. Dorken, and P.T. Daniel. 2005. Arsenic trioxide triggers a regulated form of caspase-independent necrotic cell death via the mitochondrial death pathway. Oncogene 24: 1904–1913.
Smith, C.C., S.M. Davidson, S.Y. Lim, J.C. Simpkin, J.S. Hothersall, and D.M. Yellon. 2007. Necrostatin: A potentially novel cardioprotective agent? Cardiovascular Drugs and Therapy 21: 227–233.
Sosna, J., S. Voigt, S. Mathieu, A. Lange, L. Thon, P. Davarnia, T. Herdegen, A. Linkermann, A. Rittger, F.K. Chan, D. Kabelitz, S. Schutze, and D. Adam. 2014. TNF-induced necroptosis and PARP-1-mediated necrosis represent distinct routes to programmed necrotic cell death. Cellular and Molecular Life Sciences 71: 331–348.
Stanger, B.Z., P. Leder, T.H. Lee, E. Kim, and B. Seed. 1995. RIP: A novel protein containing a death domain that interacts with Fas/APO-1 (CD95) in yeast and causes cell death. Cell 81: 513–523.
Sugioka, R., S. Shimizu, and Y. Tsujimoto. 2004. Fzo1, a protein involved in mitochondrial fusion, inhibits apoptosis. Journal of Biological Chemistry 279: 52726–52734.
Sun, L., H. Wang, Z. Wang, S. He, S. Chen, D. Liao, L. Wang, J. Yan, W. Liu, X. Lei, and X. Wang. 2012. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell 148: 213–227.
Tenev, T., K. Bianchi, M. Darding, M. Broemer, C. Langlais, F. Wallberg, A. Zachariou, J. Lopez, M. Macfarlane, K. Cain, and P. Meier. 2011. The Ripoptosome, a signaling platform that assembles in response to genotoxic stress and loss of IAPs. Molecular Cell 43: 432–448.
Trichonas, G., Y. Murakami, A. Thanos, Y. Morizane, M. Kayama, C.M. Debouck, T. Hisatomi, J.W. Miller, and D.G. Vavvas. 2010. Receptor interacting protein kinases mediate retinal detachment-induced photoreceptor necrosis and compensate for inhibition of apoptosis. Proceedings of the National Academy of Sciences of the United States of America 107: 21695–21700.
Upton, J.W., W.J. Kaiser, and E.S. Mocarski. 2010. Virus inhibition of RIP3-dependent necrosis. Cell Host & Microbe 7: 302–313.
Vanlangenakker, N., M.J. Bertrand, P. Bogaert, P. Vandenabeele, and T. Vanden Berghe. 2011. TNF-induced necroptosis in L929 cells is tightly regulated by multiple TNFR1 complex I and II members. Cell Death and Disease 2: e230.
Wang, Z., H. Jiang, S. Chen, F. Du, and X. Wang. 2012. The mitochondrial phosphatase PGAM5 functions at the convergence point of multiple necrotic death pathways. Cell 148: 228–243.
Wong, R.S. 2011. Apoptosis in cancer: From pathogenesis to treatment. Journal of Experimental & Clinical Cancer Research 30: 87.
Xu, Y., S. Huang, Z.G. Liu, and J. Han. 2006. Poly(ADP-ribose) polymerase-1 signaling to mitochondria in necrotic cell death requires RIP1/TRAF2-mediated JNK1 activation. Journal of Biological Chemistry 281: 8788–8795.
Zhang, D.W., J. Shao, J. Lin, N. Zhang, B.J. Lu, S.C. Lin, M.Q. Dong, and J. Han. 2009. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325: 332–336.
Zhao, J., S. Jitkaew, Z. Cai, S. Choksi, Q. Li, J. Luo, and Z.G. Liu. 2012. Mixed lineage kinase domain-like is a key receptor interacting protein 3 downstream component of TNF-induced necrosis. Proceedings of the National Academy of Sciences of the United States of America 109: 5322–5327.
Zheng, W., A. Degterev, E. Hsu, J. Yuan, and C. Yuan. 2008. Structure-activity relationship study of a novel necroptosis inhibitor, necrostatin-7. Bioorganic & Medicinal Chemistry Letters 18: 4932–4935.
Zong, W.X., D. Ditsworth, D.E. Bauer, Z.Q. Wang, and C.B. Thompson. 2004. Alkylating DNA damage stimulates a regulated form of necrotic cell death. Genes & Development 18: 1272–1282.
Acknowledgments
This research was fully supported by college of pharmacy-specialized research fund (the Institute for New Drug Development) of Keimyung University in 2012.
Conflict of interest
The author declares no directly or indirectly conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bae, J.H., Shim, JH. & Cho, Y.S. Chemical regulation of signaling pathways to programmed necrosis. Arch. Pharm. Res. 37, 689–697 (2014). https://doi.org/10.1007/s12272-014-0385-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-014-0385-6