Abstract
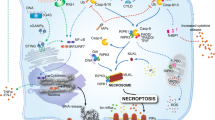
Cell death occurs spontaneously or in response to external stimuli, and can be largely subdivided into apoptosis and necrosis by the distinct morphological and biochemical features. Unlike apoptosis, necrosis was recognized as the passive and unwanted cell demise committed in a non-regulated and disorganized manner. However, under specific conditions such as caspase intervention, necrosis has been proposed to be regulated in a well-orchestrated way as a backup mechanism of apoptosis. The term programmed necrosis has been coined to describe such an alternative cell death. Recently, at least some regulators governing programmed necrosis have been identified and demonstrated to be interconnected via a wide network of signal pathways by further extensive studies. There is growing evidence that programmed necrosis is not only associated with pathophysiological diseases, but also provides innate immune response to viral infection. Here, we will introduce recent updates on the molecular mechanism and physiological significance of programmed necrosis.
Similar content being viewed by others
References
Adams, S., Pankow, S., Werner, S., and Munz, B. (2007). Regulation of NF-kappaB activity and keratinocyte differentiation by the RIP4 protein: implications for cutaneous wound repair. J. Invest. Dermatol. 127, 538–544.
Agostinis, P., Buytaert, E., Breyssens, H., and Hendrickx, N. (2004). Regulatory pathways in photodynamic therapy induced apoptosis. Photochem. Photobiol. Sci. 3, 721–729.
Cauwels, A., Janssen, B., Waeytens, A., Cuvelier, C., and Brouckaert, P. (2003). Caspase inhibition causes hyperacute tumor necrosis factor-induced shock via oxidative stress and phospholipase A2. Nat. Immunol. 4, 387–393.
Chan, F.K., Shisler, J., Bixby, J.G., Felices, M., Zheng, L., Appel, M., Orenstein, J., Moss, B., and Lenardo, M.J. (2003). A role for tumor necrosis factor receptor-2 and receptor-interacting protein in programmed necrosis and antiviral responses. J. Biol. Chem. 278, 51613–51621.
Cho, Y.S., Challa, S., Moquin, D., Genga, R., Ray, T.D., Guildford, M., and Chan, F.K. (2009). Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virusinduced inflammation. Cell 137, 1112–1123.
Cinquepalmi, L., Boni, L., Dionigi, G., Rovera, F., Diurni, M., Benevento, A., and Dionigi, R. (2006). Long-term results and quality of life of patients undergoing sequential surgical treatment for severe acute pancreatitis complicated by infected pancreatic necrosis. Surg. Infect. 7, S113–116.
de Murcia, G., Schreiber, V., Molinete, M., Saulier, B., Poch, O., Masson, M., Niedergang, C., and Menissier de Murcia, J. (1994). Structure and function of poly(ADP-ribose) polymerase. Mol. Cell. Biochem. 138, 15–24.
Degterev, A., Hitomi, J., Germscheid, M., Ch’en, I.L., Korkina, O., Teng, X., Abbott, D., Cuny, G.D., Yuan, C., Wagner, G., et al. (2008). Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat. Chem. Biol. 4, 313–321.
Eigenbrod, T., Park, J.H., Harder, J., Iwakura, Y., and Nunez, G. (2008). Cutting edge: critical role for mesothelial cells in necrosisinduced inflammation through the recognition of IL-1 alpha released from dying cells. J. Immunol. 181, 8194–8198.
Galluzzi, L., and Kroemer, G. (2008). Necroptosis: a specialized pathway of programmed necrosis. Cell 135, 1161–1163.
Galluzzi, L., and Kroemer, G. (2009). Shigella targets the mitochondrial checkpoint of programmed necrosis. Cell Host Microbe 5, 107–109.
Han, W., Xie, J., Li, L., Liu, Z., and Hu, X. (2009). Necrostatin-1 reverts shikonin-induced necroptosis to apoptosis. Apoptosis 14, 674–686.
Hartwig, A., Asmuss, M., Blessing, H., Hoffmann, S., Jahnke, G., Khandelwal, S., Pelzer, A., and Burkle, A. (2002). Interference by toxic metal ions with zinc-dependent proteins involved in maintaining genomic stability. Food Chem. Toxicol. 40, 1179–1184.
He, S., Wang, L., Miao, L., Wang, T., Du, F., Zhao, L., and Wang, X. (2009). Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell 137, 1100–1111.
Hitomi, J., Christofferson, D.E., Ng, A., Yao, J., Degterev, A., Xavier, R.J., and Yuan, J. (2008). Identification of a molecular signaling network that regulates a cellular necrotic cell death pathway. Cell 135, 1311–1323.
Holler, N., Zaru, R., Micheau, O., Thome, M., Attinger, A., Valitutti, S., Bodmer, J.L., Schneider, P., Seed, B., and Tschopp, J. (2000). Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat. Immunol. 1, 489–495.
Hsu, T.S., Yang, P.M., Tsai, J.S., and Lin, L.Y. (2009). Attenuation of cadmium-induced necrotic cell death by necrostatin-1: potential necrostatin-1 acting sites. Toxicol. Appl. Pharmacol. 235, 153–162.
Joza, N., Susin, S.A., Daugas, E., Stanford, W.L., Cho, S.K., Li, C.Y., Sasaki, T., Elia, A.J., Cheng, H.Y., Ravagnan, L., et al. (2001). Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 410, 549–554.
Kang, Y.H., Yi, M.J., Kim, M.J., Park, M.T., Bae, S., Kang, C.M., Cho, C.K., Park, I.C., Park, M.J., Rhee, C.H., et al. (2004). Caspase-independent cell death by arsenic trioxide in human cervical cancer cells: reactive oxygen species-mediated poly(ADP-ribose) polymerase-1 activation signals apoptosis-inducing factor release from mitochondria. Cancer Res. 64, 8960–8967.
Krysko, D.V., D’Herde, K., and Vandenabeele, P. (2006). Clearance of apoptotic and necrotic cells and its immunological consequences. Apoptosis 11, 1709–1726.
Lautier, D., Lagueux, J., Thibodeau, J., Menard, L., and Poirier, G.G. (1993). Molecular and biochemical features of poly (ADP-ribose) metabolism. Mol. Cell. Biochem. 122, 171–193.
Lin, Y., Choksi, S., Shen, H.M., Yang, Q.F., Hur, G.M., Kim, Y.S., Tran, J.H., Nedospasov, S.A., and Liu, Z.G. (2004). Tumor necrosis factor-induced nonapoptotic cell death requires receptor-interacting protein-mediated cellular reactive oxygen species accumulation. J. Biol. Chem. 279, 10822–10828.
Micheau, O., and Tschopp, J. (2003). Induction of TNF receptor Imediated apoptosis via two sequential signaling complexes. Cell 114, 181–190.
Moubarak, R.S., Yuste, V.J., Artus, C., Bouharrour, A., Greer, P.A., Menissier-de Murcia, J., and Susin, S.A. (2007). Sequential activation of poly(ADP-ribose) polymerase 1, calpains, and Bax is essential in apoptosis-inducing factor-mediated programmed necrosis. Mol. Cell. Biol. 27, 4844–4862.
Nakagawa, T., Shimizu, S., Watanabe, T., Yamaguchi, O., Otsu, K., Yamagata, H., Inohara, H., Kubo, T., and Tsujimoto, Y. (2005). Cyclophilin D-dependent mitochondrial permeability transition regulates some necrotic but not apoptotic cell death. Nature 434, 652–658.
Pardo, J., Galvez, E.M., Koskinen, A., Simon, M.M., Lobigs, M., Regner, M., and Mullbacher, A. (2009). Caspase-dependent inhibition of mousepox replication by gzmB. PloS one 4, e7512.
Petit, F., Arnoult, D., Viollet, L., and Estaquier, J. (2003). Intrinsic and extrinsic pathways signaling during HIV-1 mediated cell death. Biochimie 85, 795–811.
Porcelli, S.A., and Jacobs, W.R., Jr. (2008). Tuberculosis: unsealing the apoptotic envelope. Nat. Immunol. 9, 1101–1102.
Riedl, S.J., and Salvesen, G.S. (2007). The apoptosome: signalling platform of cell death. Nat. Rev. 8, 405–413.
Strasser, A., O’Connor, L., and Dixit, V.M. (2000). Apoptosis signaling. Ann. Rev. Biochem. 69, 217–245.
Xu, Y., Huang, S., Liu, Z.G., and Han, J. (2006). Poly(ADP-ribose) polymerase-1 signaling to mitochondria in necrotic cell death requires RIP1/TRAF2-mediated JNK1 activation. J. Biol. Chem. 281, 8788–8795.
Yang, P.M., Chen, H.C., Tsai, J.S., and Lin, L.Y. (2007). Cadmium induces Ca2+-dependent necrotic cell death through calpaintriggered mitochondrial depolarization and reactive oxygen species-mediated inhibition of nuclear factor-kappaB activity. Chem. Res. Toxicol. 20, 406–415.
Zhang, D.W., Shao, J., Lin, J., Zhang, N., Lu, B.J., Lin, S.C., Dong, M.Q., and Han, J. (2009). RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325, 332–336.
Zong, W.X., Ditsworth, D., Bauer, D.E., Wang, Z.Q., and Thompson, C.B. (2004). Alkylating DNA damage stimulates a regulated form of necrotic cell death. Genes Dev. 18, 1272–1282.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Cho, Y.S., Park, S.Y., Shin, H.S. et al. Physiological consequences of programmed necrosis, an alternative form of cell demise. Mol Cells 29, 327–332 (2010). https://doi.org/10.1007/s10059-010-0066-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-010-0066-3