Abstract
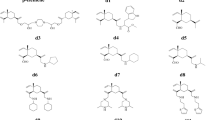
A new spiroketal, spirodienal B (1), was isolated from culture extracts of S. cellulosum. Its structure was elucidated by spectroscopic analysis. In addition to 1, the strain produced a second, closely related antibiotic, spirodienal. Both compounds demonstrated potent cytotoxicity against human tumor cells. Against human tumor cells such as HCT-15, the activity of 1 was more than 4000 times stronger than that of doxorubicin in terms of IC50.
Similar content being viewed by others
References
Ahn, J.-W., Spirodienal, a new spiroketal from Sorangium cellulosum. Bull. Korean Chem., Soc. 30, 742–744 (2009).
Ahn, J.-W., Li, X., and Zee, O.-P., Soraphinol B, a new acyloin compound produced by Sorangium cellulosum. Bull. Korean Chem. Soc., 28, 1215–1216 (2007).
Cowden, C. J. and Paterson, I., Cancer drugs better than taxol? Nature, 387, 238–239 (1997).
Gerth, K., Bedorf, N., Höfle, G., Irschik, H., and Reichenbach, H., Epothilones A and B: Antifungal and cytotoxic compounds from Sorangium cellulosum (Myxobacteria). J. Antibiot., 49, 560–563 (1996).
Hardt, I. H., Steinmetz, H., Gerth, K., Sasse, F., Reichenbach, H., and Höfle, G., New natural epothilones from Sorangium cellulosum, strains So ce90/B2 and So ce90/D13: Isolation, structure elucidation, and SAR studies. J. Nat. Prod., 64, 847–856 (2001).
Kirst, H. A., Mynderse, J. S., Martin, J. W., Baker, P. J., Paschal, J. W., Rios Steiner, J. L., Lobkovsky, E., and Clardy, J., Structure of the spiroketal-macrolide ossamycin. J. Antibiot., 49, 162–167 (1996).
Kobayashi, K., Nishino, C., Ohya, J., Sato, S., Mikawa, T., Shiobara, Y., Kodama, M., and Nishimoto, N., Oligomycin E, a new antitumor antibiotic produced by Streptomyces sp. MCI-2225. J. Antibiot., 40, 1053–1057 (1987).
Li, X., Yu, T.-K., Kwak, J. H., Son, B.-Y., Seo, Y., Zee, O.-P., and Ahn, J.-W., Soraphinol C, a new free-radical scavenger from Sorangium cellulosum. J. Microbiol. Biotechnol., 18, 520–522 (2008).
Litaudon, M., Hickford, S. J. H., Lill, R. E., Lake, R. J., Blunt, J. W., and Munro, M. H. G., Antitumor polyether macrolides: New and hemisynthetic halichondrins from the New Zealand deep-water sponge Lissodendoryx sp. J. Org. Chem., 62, 1868–1871 (1997).
McCurdy, H. D., Bergey’s Manual of Determinative Bacteriology, Buchanan, R. E., Gibbons, N. E. (eds.), Williams & Wilkins, Baltimore, 79–98 (1974).
Niggemann, J., Bedorf, N., Flörke, U., Steinmetz, H., Gerth, K., Reichenbach, H., and Höfle, G., Spirangien A and B, highly cytotoxic and antifungal spiroketals from the myxobacterium Sorangium cellulosum: Isolation, structure elucidation and chemical modifications. Eur. J. Org. Chem., 5013–5018 (2005).
Reichenbach, H. and Dworkin, M., The myxobacteria. In Balows, A., Trupper, H. G., Dworkin, M., Harder, W., and Schleifer, K. H. (eds.), The Prokaryotes, 2nd ed. Springer Verlag, New York, 3416–3487 (1992).
Skehan, P., Storeng, R., Scudiero, D., Monks, A., McMahon, J., Vistica, D., Warren, J., Bokesch, H., Kenny, S., and Boyd, M. R., New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst., 82, 1107–1112 (1990).
Someno, T., Kumagai, H., Ohba, S.-I., Amemiya, M., Naganawa, H., Ishizuka, M., and Ikeda, D., ICM0301s, New angiogenesis inhibitors from Aspegillus sp. F-1491. J. Antibiot., 57(2), 104–109 (2004).
Suarez-Gomez, B., Souto, M. L., Norte, M., and Fernandez, J. J., Isolation and structural determination of DTX-6, a new okadaic acid derivative. J. Nat. Prod., 64, 1363–1364 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kwak, J.H., Ahn, JW. A new cytotoxic spiroketal from the myxobacterium Sorangium cellulosum . Arch. Pharm. Res. 32, 841–844 (2009). https://doi.org/10.1007/s12272-009-1604-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-009-1604-4