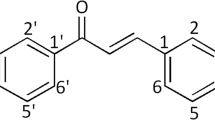
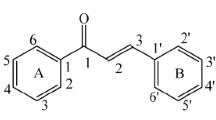
Abstract
Chalcones and their analogs have been an area of great interest in recent years. Numerous research papers have been published, and chalcones continue to show promise for new drug investigations. Researchers have explored new approaches for the synthesis of chalcone derivatives, which have revealed an array of pharmacological and biological effects. These chalcone derivatives have shown important antimicrobial, antifungal, anti-mycobacterial, antimalarial, antiviral, anti-inflammatory, antioxidant, antileishmanial anti-tumor, and anticancer properties. This review highlights the synthesis and pharmacological properties of chalcone derivatives.













































































Similar content being viewed by others
References
S.V. Kostanecki, Tambor, J. Chem. Ber. 32, 1921 (1899)
S. Maayan, N. Ohad, K. Soliman, Bioorg. Med. Chem. 13(2), 433–441 (2005)
Z.E. Nowakowska, Eur. J. Med. Chem. 42(2), 125–137 (2007)
M.L. Go, X. Wu, X.L. Liu, Curr. Med. Chem. 12(4), 481–499 (2005)
M. Cushman, D. Nagarathnamm, J. Nat. Prod. 54(6), 1656–1660 (1991)
M.A. Rahman, Chem. Sci. J. 29, 1–16 (2011)
T.S. Straub, Tetrahedron Lett. 36, 663 (1995)
S.A. Hermes, Chem. Ber. 70, 96422h (1969)
E.D. Bergman, L. Ginsibm, R. Pappo, Org. React. 10, 179 (1959)
Y.R. Prasad, P.R. Kumar, C.A. Deepti, M.V. Ramana, Eur. J. Chem. 3(13), 236–241 (2006)
M.L. Go, X. Wu, X.L. Liu, Curr. Med. Chem. 12, 483 (2005)
U.M. Liu, P. Wilairat, S.L. Croft, A.L. Tan, M. Go, Bioorg. Med. Chem. 11, 2729 (2003)
D. Binder, C.R. Noe, W. Holzer, B. Rosenwirth, Arch. Pharm. 318, 48 (1985)
B.P. Bandga, S.A. Patil, B.L. Korbad, S.H. Nile, C.N. Khobragade, Eur. J. Med. Chem. 45, 2629–2633 (2010)
N. Sunduru, A. Agarwal, S.B. Katiyar, Nishi, N. Goyal, S. Gupta, P.M.S. Chauhan, Bioorg. Med. Chem. 14, 7706–7715 (2006)
D. Kumar, N.M. Kumar, K. Akamatsu, E. Kusaka, H. Harada, T. Ito, Bioorg. Med. Chem Lett. 20, 3916–3919 (2010)
N. Tiwari, B. Dwivedi, K.F. Nizamuddin, Y. Nakanshi, K.H. Lee, Bioorg. Med. Chem. 10, 699 (2000)
J. Tatsuzaki, K.F. Bastow, K. Nakagawa-Goto, S. Nakamura, H. Itokawa, K.-H. Lee, J. Nat. Prod. 69(10), 1445–1449 (2006)
J.-M. Yun, M.-H. Kweon, H. Kwon, J.-K. Hwang, H. Mukhtar, Carcinogenesis 27(7), 1454–1464 (2006)
G.D. Carlo, N. Mascolo, A.A. Izzo, F. Capasso, Life Sci. 65, 337 (1999)
M.A. El.Hashah, M. El-Kady, M.A. Saiyed, A.A. Elaswy, Egypt J. Chem. 27, 715 (1985)
L.S. Crawley, W.J. Fanshawe, J. Het. chem. 14, 531 (1977)
E.C. Taylor, R.W. Morrison, J. Org. Chem. 32, 2379 (1967)
P.S. Utale, P.B. Raghuvanshi, A.G. Doshi, Asian J. Chem. 10, 597 (1998)
M. Kidwai, P. Misra, J. Org. Chem. 29(18), 3237–3250 (2007)
A. Guida, M.H. Lhouty, D. Tichit, F. Figueras, P. Geneste, Appl. Catal. 164, 251 (1997)
G. Romanelli, G. Pasquale, A. Sathicq, H. Thomas, J. Autino, P.V. azquez, J. Mol. Catal. A: Chem. 340, 24 (2011)
S. Eddarir, N. Cotelle, Y. Bakkour, C. Rolando, Tetrahedron Lett. 44, 5359 (2003)
D.N. Dhar, Chemistry of Chalcones and Related Compounds (Wiley, New York, 1981)
M.J. Climent, A. Corma, S. Iborra, A. Velty, J. Catal. 221, 474 (2004)
S. Sebti, A. Solhy, A. Smahi, A. Kossir, H. Oumimoun, Catal. Commun. 3, 335–339 (2002)
F. Micheli, F. Degiorgis, A. Feriani, A. Paio, A. Pozzan, P. Zarantonello, P.J. Seneci, Comb. Chem. 3, 224 (2001)
X. Bu, L. Zhao, Y. Li. Synthesis. 11, 1246–1248 (1997)
A.R. Alcantara, J.M. Marinas, J.V. Sinisterra, Tetrahedron Lett. 28, 1515 (1987)
J.B. Daskiewicz, G. Comte, D. Barron, A.D. Pietro, F. Thomasson, Tetrahedron Lett. 40, 7095 (1999)
N. Iranpoor, F. Kazemi, Tetrahedron Lett. 54, 9475 (1998)
N.O. Calloway, L.D. Green, J. Am. Chem. Soc. 59, 809 (1937)
T. Szell, I. Sohalr, Can. J. Chem. 47, 1254 (1969)
K. Irie, K. Watanabe, Bull. Chem. Soc. Jpn 53, 1366 (1980)
L. Mazza, A. Guaram, Synthesis. 1, 41–44 (1980)
T. Nakano, S. Irifune, S. Umano, A. Inada, Y. Ishii, M.J. Ogawa, J. Org. Chem. 52, 2239–2244 (1987)
R.E. Lyle, L.P. Paradis, J. Am. Chem. Soc. 77, 6667 (1955)
M. G. Marathey, J. Uni. Poona. 1–19 (1952)
A.A. Raval, N.M. Shah, J. Sci. Res. India 21, 234 (1962)
Szell and Sipos, Ann. 64, 113 (1961)
C. Kuroda, T. Matsukuma, Chem Abstr. 26, 2442 (1932)
L. Reichel, Nalanxlissenchaften 33, 215 (1944). doi:10.4236/oalib.1100367
G.V. Jadhav, V.G. Kulkami, Curr. Sci. India. 20, 42 (1951)
V. M. Vlasov, Izv. Sib. Old. Akd. Nauk. USSR, Set: Khim. Nauk., 2, 96 (1971),C.A., 60, 1683 (1964)
P. L. Cheng, P. Fournari and Triouflet, J. Bull. Soc. Chem. France., 1963, 10, 2248, C. A., 60, 1683 (1964)
D.S. Breslow, C.R. Hauser, J. Am. Chem. Soc. 62, 2385 (1940)
S. Fujise, H. Tatsuta, J. Chem. Soc. Jpn. 63, 632 (1942)
J.L. Guthrit, N. Rabjohn, J. Org. Chem. 22, 176 (1957)
H. Gilman, J.P. Nelson, Rec. Trav. Chem. 55, 518 (1936)
E.P. Kohler, H.M. Chandwell, Org. Synth. Coll. 2, 1 (1922)
E. Schraufstalter, S. Deutsch, Chem. Ber. 81, 489 (1948)
H.E. Smith, M.C. Paulson, J. Am. Chem. Soc. 76, 4486 (1954)
H. Obara, J. Onodera, Y. Kurihara, Bull. Chem. Soc. Jpn. 44, 289 (1971)
J. Shinoda and S. Sato, J. Pharm. Soc. Japan. 48, 791, (1928) C.A., 23,836, (1929)
E.F. Kurth, J. Am. Chem. Soc. 68, 697 (1946)
T.A. Gaissman, R.O. Clinton, J. Am. Chem. Soc. 68, 697 (1946)
G. J. Martin, J. M. Beler and S. Avakian, U. S. Patent,2, 769, 817 (1956), C.A., 51, 14815 (1957)
Falcao de Fonseca L and Bol Fac Farm, Univ. Coimbra, Ed. Cient, 28, 49,(1968), C. A., 72, 121124p (1970)
S. Fujise, H. Tatsuta, J. Chem. Soc. Japan. 63, 932 (1942)
D.N. Dhar, J.B. Lal, J. Org. Chem. 23, 1159 (1958)
W. Davey and J. R. Gwilt, J. Chem. Soc. 1008 (1957)
R.T. Morrison, R.N. Boyd, Organic Chemistry, 6th edn. (Pearson Education Publishers, 2004), pp. 971–990, 997–1020
S.M. Mukherji, S.P. Singh, R.P. Kapoor, Organic Chemistry, vol. II. (New Age International (P) Limited Publishers, 2003), pp. 586–587
M. Palaniandavar, C. Natarajan, Aust. J. Chem. 33, 737 (1980)
J. Russel, H. Clarke, J. Am. Chem. Soc. 61, 3651 (1939)
S. Eddarir, N. Cotelle, Y. Bakkour, C. Rolando, Tetrahedron Lett. 44(28), 5359–5363 (2003)
A. Bianco, C. Cavarischia, A. Farina, M. Guiso, C.A. Marra, Tetrahedron Lett. 44(51), 9107–9109 (2003)
V.R. Venkatraman, S. Nagrajan, J. Indian Chem. Soc. 62, 787 (1985)
V.M. Patel, Ph.D. Thesis, South Gujarat University, Surat, Dec.2004
T. Szell, I. Sohar, Acta. Chim. (Budapest) 62, 429 (1969)
Onoda and T. Sasaki, Akita Daigaku Kyoiku Gakubu Kiyo, Shizen Kagaku. 25, 7 (1975)
Onoda, T. Sasaki, Chem. Abstr. 85, 5450 (1976)
S. Fujise, H. Tatsuta, J. Chem. Soc. Jpn. 63, 932 (1942)
H. Gilman, L. Cason, J. Am. Chem. Soc. 72, 3469 (1950)
H. Bestmann, B. Arnason, J. Chem. Ber. 95, 1513 (1962)
S. Trippett, D.M. Walker, Chem. Ind. (London) 933 (1960)
W.S. Wadsworth, W.D. Emmons, J. Am. Chem. Soc. 83, 1733 (1961)
S. Trippett, D.M. Walker, J. Chem. Soc. 1266–1272 (1961)
A.K. Bose, M.S. Manhas, M.S. Ghosh, J. Org. Chem. 56, 6965 (1991)
R. Gupta, S. Paul, A. Gupta, Indian J. Chem. 33, 888 (1994)
V.R. Naik, H.B. Naik, Asian J. Chem. 12, 305 (2000)
A.R. Jagdale, A. Sudalai, Tetrahedron Lett. 48(28), 4895–4898 (2007)
D.H. Choi, Y.K. Cha, Bull. Korean Chem. Soc. 23(3), 469–476 (2002)
K.M. Johnston, J.F. Jones, J. Chem. Soc. 814–817 (1969)
V.T. Ramakrishan, J.J. Kagan, Org. Chem. 35(9), 2898–2900 (1970)
Z. Zheng, H. Alper, Org. Lett. 11(15), 3278–3281 (2009)
I. Abe, Y. Sano, Y. Takahashi, H. Noguchi, J. Biol. Chem. 278(27), 25218–25226 (2003)
T. Baba, H. Kizuka, H. Handa, Y. Ono, Appl Catal. 194–195, 203 (2000)
R.N. Mistry, K.R. Desai, Asian J. Chem. 16(1), 201 (2004)
E.L. Gall, F. Texier-Boullet, J. Hamelin, Syn. Commun. 29(20), 3651–3657 (1999)
B.E. Blass, Tetrahedron Lett. 58(46), 9301–9320 (2002)
U. Bora, A. Saikia, R.C. Boruah, Ind. J. Chem. 44B(12), 2523–2526 (2005)
W. Wei, W. Qunrong, D. Liqin, Z. Aiquing, W. Duoyauan, Ultrasonics Sonochem. 12(6), 411–414 (2005)
A. Fuentes, J.M. Marinas, J.V. Sinisterra, Tetrahedron Lett. 28(39), 4541–4544 (1987)
V. Calvino, M. Picallo, A.J. Lpoz-Peinado, R.M. Martin-Aranda, C.J. Duran-Valle, Appl. Surface Sci. 252(17), 6071–6074 (2006)
G. Rothenberg, A.P. Downie, C.L. Raston, J.L. Scott, J. Am. Chem. Soc. 123, 8701–8708 (2001)
Z.N. Siddiqui, Arab. J. Chem (2015). doi:10.1016/j.arabjc.2015.06.013. (Article in Press)
U.C. Rajesh, G. Purohit, D.S. Rawat, ACS Sustain. Chem. Eng. 3(10), 2397–2404 (2015)
E. Coombs, D.P. Evans, J. Chem. Soc. 1295–1930 (1940)
E.K. Nikitin, J. Gen. Chem. USSR. 6, 1278 (1936)
P.L. Nayak, M.K. Rout, J. Ind. Chem. Soc. 52, 809 (1975)
G. Sipos, A. Fuka, T. Szell, Monatsh. Chem. 91, 643 (1960)
D.S. Noyce, W.A. Pryor, J. Am. Chem. Soc. 11, 1397 (1955)
A.B. Linke, D.E. Eveleigh, Z. Naturorsch 30B, 740 (1975)
R.M. Horowitz and B. Gentili, U. S. Patent, 1975, 3, 890, 298
R.M. Horowitz, B. Gentili, Chem. Abstr. 83, 147706g (1975)
J.H. Adams, British Patent, 1971, 1, 250, 338
J.H. Adams, Chem. Abstr. 16, 46956 (1972)
G.P. Rizzi and J.S. Neely, German Patent, 1972, 2, 148, 332
G.P. Rizzi, J.S. Neely, Chem. Abstr. 11, 86777h (1972)
J. Fukomoto and S. Okada, German Patent, 1922, 2, 204, 716
J. Fukomoto, S. Okada, Chem. Abstr. 11, 152515z (1972)
M. Delcarmen, G. Barrio, J.R. Barrio, G. Walker, A. Noveli, N.J. Leonard, J. Am. Chem. Soc. 95, 4891 (1973)
Mitsubishi Petrochemical Co. Ltd., British Patent. 1968, 1, 128, 090
Mitsubishi Petrochemical Co. Ltd., Chem. Abstr. 69, 97342y (1968)
Hercules Powder Co., British Patent. 1959, 873, 021
Hercules Powder Co., Chem. Abstr. 56, 6185 h (1962)
G. Hayakawa and T. Inoue, Japanese Patent 7, 107, 386 (1971)
G. Hayakawa, T. Inoue, Chem. Abstr. 74, 14332y (1971)
N.V. Phillip’s, Gloeilampenfabrieken, Netherland Patent. 6, 501, 841 (1966)
N.V. Phillip’s, Chem. Abstr. 66, 16034n (1967)
P. Baeyens and G. Krijil, Trans. Inst. Metal. Finish, 45, 115 (1967)
P. Baeyens, G. Krijil, Chem. Abstr. 67, 10450a (1967)
J.H. Adams, British Patent. 1, 250, 388 (1971)
J.H. Adams, Chem. Abstr. 76, 46956p (1972)
D.A. Akhmedzade, V.D. Yasnopol’skii and Y.I. Golovanova, Azerb KhimZh., 1968, 117
D.A. Akhmedzade, V.D. Yasnopol’skii, Y.I. Golovanova, Chem. Abstr. 69, 107333n (1968)
T.H. Simpson and N. Uri, British Patent, 875, 164 (1956)
T.H. Simpson, N. Uri, Chem. Abstr. 56, 3582b (1962)
V. Karel, Brauwisscnchaft. 14, 411 (1961)
V. Karel, Chem. Abstr. 56, 5220a (1962)
E.K. Fields, U.S. Patent. 2, 799, 652 (1957)
E.K. Fields, Chem. Abstr. 51, 15113c (1957)
International Minerals and Chemical Corpn., British Patent. 1, 189, 573 (1970)
International Minerals and Chemical Corpn, Chem. Abstr. 73, 45798q (1970)
A. Russell, J. Todd, J. Chem. Soc. 1506–1508 (1934)
P. Mitter, S. Saha, J. Indian Chem. Soc. 11, 257 (1934)
J. Shinoda, S. Sato, M. Kawagoe, J. Pharm. Soc. Japan. 24, 1459 (1904)
J. Shinoda, S. Sato, J. Pharm. Soc Japan. 49, 64 (1929)
E. Schraufstater, S. Deutch, Chem. Abstr. 44, 3563 (1950)
M. Larsen, H. Kromann, A. Kharazmi, S.F. Nielsen, Bioorg. Med. Chem. Lett. 15, 4858 (2005)
G. Zongru, H. Rui, F.Z. Shenquarg, G. Shuminhshu, Chem. Abstr. 125, 10376r (1996)
H. Serre, J. Rey, J.P. Tarayre, Chem Abstr. 91, 9494a (1979)
S.F. Nielsen, T. Boesen, M. Larsen, H. Kromann, Bioorg. Med. Chem. Lett. 12, 3047 (2004)
A.K. Padersen, G.A. Fitz, J. Pharm. Sci. 74, 188 (1985)
Y. Satomi, Ind. J. Cancer. 120, 208071 (1994)
A. Vall, B. Vall, D. Cartier, R.L. Guillou, et al., Eur. J. Med. Chem. 41, 142–146 (2006)
Tashio pharmaceutical Ltd., Chem. Abstr. 101, 54722j (1984)
M. Chen. S.B. Christensen, L. Blom, K. Fich, Antimicrob. Agents Chemother. 37, 2550 (1993)
M. Chen, S.B. Christensen, T.G. Theander, Antimicrob. Agents chemother. 38, 139 (1994)
C.M. Christensen, S.B. Zhai, M.H. Theander, J. Infect. Dis. 176, 1327 (1997)
R.J. Anto, K. Sukumaran, G. Kuttan, M.N. Rao, A. Cancer Lett. 97, 33 (1997)
F. Severi, S. Benvenuti, L. Costantino, L. Antolini, Eur. J. Med. Chem. 33, 359 (1998)
J.N. Dominguez, J.E. Charris, G. Lobo, P.J. Rosenthal, Eur. J. Med. Chem. 36, 555 (2001)
O. Nerya, O.R. Musa, S. Khatib, J. Vaya, Phytochemistry 65, 1389 (2004)
S.R. Modi, H.B. Naik, Orient. J. Chem. 10, 85 (1994)
R. Vincenzo, S.G. Panici, P. Benedess, F.O. Remelletti, Anticancer Drug Res. 10, 481 (1995)
O. Torn, O. Yoshihito, S.B. Shoji, Jpn. Kokai Tokyo JP. 11(349), 521 (1991)
O. Toru, O. Yoshihito, S. Shoji, Chem. Abstr. 132, 22752t (2000)
W.D. Seo, J.H. Kim, J.E. Kang, K.H. Park, Bioorg. Med. Chem. Lett. 15, 5514 (2005)
W. Xiang, T.R. Edward, K. Louri, et al., E. J. Pharm. Sci. 27, 175–187 (2005)
B.A. Bhatt, K.L. Dhar, S.C. Puri, G.N. Qazi, J. Pharm. Sci. 15, 3177 (2005)
S.J. Won, T.C.T. Liu, L.T. Tsao, J.R. Weng, H.H. Ko, J.P. Wang, C.N. Lim, Eur. J. Med. Chem. 40, 103 (2005)
F. Zhao, H. Nozawa, A. Daikonnya, K. Kondo, S. Kitanaka, Biol. Pharm. Bull. 26(1), 61–65 (2003)
Y. Ito, Y. Miyake. K. Okada, WO2007052330. A1 (2007)
B.A. Baviskar, B.B. Baviskar, M.R. Shiradkar, U.A. Deokate, S.S. Khadabadi, Eur. J. Chem. 6(1), 196–200 (2009)
G. Devaux, A. Nuhrich, V. Dargelos, Fr. Demande 2(357), 247 (1978)
G. Devaux, A. Nuhrich, V. Dargelos, Chem. Abstr. 89, 193384e (1978)
A. Dandia, V. Sehgal, P. Singh, Ind. J. Chem. 33(B), 1288 (1993)
Y.R. Prasad, K.P. Ravi, C.H. Asha Deepthi, M. Venkata Ramana, Asian J. Chem. 19, 4799 (2007)
M.S. Karthikeyan, B.S. Holla, N.S. Kumari, Eur. J. Med. Chem. 42(1), 30–36 (2007)
S.F. Nielsen, T. Boesen, M. Larsen, K. Schonning, H. Kromann, Bioorg. Med. Chem. 12(11), 3047–3054 (2004)
R. Tsukiyama, H. Katsura, N. Tokuriki, M. Kobayashi, Antimicrob. Agents Chemother. 46(5), 1226–1230 (2002)
Y.R. Prasad, K.P. Ravi, C.H. AshaDeepti, M.E. Venkata Ramana, Eur. J. Chem. 3(4), 236–241 (2006)
L. Stevaz, A. Tapia, S.N. Lopez, R.L. Furlan, E. Petenatti et al., J. Agric. Food Chem. 52(11), 3297–3300 (2004)
P. Boeck, P.C. Leal, R.A. Yunes, S. Zacchino, Arch. Pharm. 338(2–3), 87–95 (2005)
N.R. Rao, G.S. Rao, P. Mukkanti, Pharm. Rev. 117, 39 (2004)
H.N.E. Sohly, A.S. Joshi, A.C. Nimrod, A.M. Clark, Planta Med. 67(1), 87–89 (2001)
T.B. Machodo, I.C. Leal, R.M. Kuster, A.C. Amaral, V. Kokis, Phytother Res. 19(6), 519–525 (2005)
Z. Nowakowaska, B. Kedzia, G.E. Schroeder, Eur. J. Med. Chem. 43(4), 707–713 (2008)
S. Burmaoglu et al., J. Enzyme Inhib. Med. Chem. 32(1), 490–495 (2017)
C.L. Miranda, G.L.M. Aponso, J.F. Stevens, M.L. Deinzer, D.R. Buhler, J. Agric. Food Chem. 48(9), 3876–3884 (2000)
A.T. Dinkova-Kostova, M.A. Massiah, R.E. Bozak, R.J. Hicks, P. Talalay, Proc. Nat. Acad. Sci. 98(6), 3404–3409 (2001)
M.R.E.S. Aly, H.A.E.R. Fodah, S.Y. Saleh, Eur. J. Med. Chem. 76, 517 (2014)
T.N. Doan, D.T. Tran, Pharmacol. Pharm. 2, 282 (2011)
P.M. Sivakumar, P.K. Prabhakar, M. Doble, Med. Chem. Res. 20, 482 (2011)
Y.P. Qian, Y.J. Shang, Q.F. Teng, J. Chang, G.J. Fan et al., Food Chem. 126, 241 (2011)
S. Padhye, A. Ahmad, N. Oswal, P. Dandawate et al., Bioorg. Med. Chem. Lett. 20, 5818 (2010)
E.C. Torres-Santos, D.L. Moreira, M.A. Kaplan, M.N. Meirelles, B. Rossi-Bergmann, Antimicrob. Agents Chemother. 43(5), 1234–1241 (1999)
A. Hermoso, I.A. Jimenez, Z.A. Mamani, I.L. Bazzocchi, B. Valladares, Bioorg. Med. Chem. 11(8), 3975–3980 (2003)
M.L. Bello, L.D. Chiaradia, L.R.S. Dias, L.K. Pacheco, T.R. Stump, A. Mascarello, M. Steindel, R.A. Yunes, H.C. Castro, R.J. Nunes, C.R. Rodrigues, Bioorg. Med. Chem. Lett. 19, 5046 (2011)
Z. Nazarian, S. Emami, S. Heydari, S.K. Ardestani, M. Nakhjiri, F. Poorrajab, A. Shafiee, A. Foroumadi, Eur. J. Med. Chem. 45, 1424 (2010)
A. Foroumadi, S. Emami, M. Sorkhi, M. Nakhjiri, Z. Nazarian, S. Heydari, S.K. Ardestani, F. Poorrajab, A. Shafiee, Chem. Biol. Drug Des. 75, 590 (2010)
M. Chen, T.G. Theander, S.B. Christensen, L. Hviid, L. Zhai, A. Kharazmi, Antimicrob. Agents Chemother. 38, 1470 (1994)
J.N. Dominguez, C. Leon, J. Rodrigues, P.J. Rosenthal, J. Med. Chem. 48(10), 3654–3658 (2005)
J.N. Dominguez, C. Leon, J. Rodrigues, P.J. Rosenthal, Farmaco 60(4), 307–311 (2005)
N. Yadav, S.K. Dixit, A. Bhattacharya, L.C. Mishra, M. Sharma, S.K. Awasthi, V.K. Bhasin, Chem. Biol. Drug Des. 80, 340 (2012)
N. Sharma, D. Mohanakrishnan, U.K. Sharma, R. Kumar, A.K. Richa, D.Sahal Sinha, Eur. J. Med. Chem. 79, 350 (2014)
E.M. Guantai, K. Ncokazi, T.J. Egan, J. Gut, P.J. Rosenthal, R. Bhampidipati, A. Kopinathan, P.J. Smith, K. Chibale, J. Med. Chem. 54, 3637 (2011)
G. Wanare, R. Aher, N. Kawathekar, R. Ranjan, N.K. Kaushik, D. Sahal, Bioorg. Med. Chem. Lett. 20, 4675 (2010)
V. Tomar, G. Bhattacharjee, S. Kamaluddin, K. Rajakumar, S.K. Srivastava, E.J. Puri, J. Med. Chem. 45, 745 (2010)
X. Wu, P. Wilairat, M.L. Go, Bioorg. Med. Chem. Lett. 12(17), 2299–2302 (2002)
P.M. Sivakumar, S.P. Sreenivasan, V. Kumar, M. Doble, Bioorg. Med. Chem. Lett. 17(6), 1695–1700 (2007)
P.M. Sivakumar, S.K. Geetha Babu, D. Mukesh, Chem. Pharm. Bull. 55(1), 44–49 (2007)
G.M. Cunha, J.B. Fontenele, F.C. De Souza, E.R. Silveira, N.A. Nogueira et al., Phytotherap. Res. 17(2), 155–159 (2003)
T. Sato, T. Taguchi, I. Umezawa, T. Inoue, N. Kawasaki, WO2000051959, A1 (2000)
A. Phrutivorapongkul, V. Lipipun, N. Ruangrungsi, K. Kirtikara, K. Nishikawa et al., Chem. Pharm. Bull. 51(2), 187–190 (2003)
N.J. Lawrence, A.T. McGown, S. Ducki, J.A. Hadfield, Anticancer Drug Des. 15(2), 135–141 (2000)
F. Epifano, S. Genovese, L. Menghini, M. Curini, Phytochemistry 68(7), 939–953 (2007)
C. Nakamura, N. Kawasaki, H. Miyataka, E. Jayachandran, I.H. Kim et al., Bioorg. Med. Chem. Lett. 10(3), 699–706 (2002)
D. Salvie, F. Richard, A. John, Bioorg. Med. Chem. Lett. 9, 8 (1998)
E. Bombardeli, P. Valenti, Chem. Abstr. 134, 222628k (2001)
M. Uenaka, K. Kawata, M. Nagai, T. Endoh, Chem. Abstr. 134, 29421j (2001)
H.X. Xu, M. Wan, H. Dong, P.P. But, L.Y. Foo, Biol. Pharm. Bull. 23(9), 1072–1076 (2000)
G. Nakagawa, K. Lee, Tetrahedron Lett. 47(47), 8263–8266 (2006)
T. Arslan et al., Arch. Pharm. (weinheim) 349(9), 741–748 (2016)
T. Arslan et al., Bioorg. Med. Chem. Lett. 26(24), 5867–5870 (2016)
H-Y Qiu et al., Chem. Med. Chem. Article in press (2017). doi:10.1002/cmdc.201700001
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gaonkar, S.L., Vignesh, U.N. Synthesis and pharmacological properties of chalcones: a review. Res Chem Intermed 43, 6043–6077 (2017). https://doi.org/10.1007/s11164-017-2977-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-2977-5