Abstract
Chronic pain relief remains an unmet medical need. Current research points to a substantial contribution of glia-neuron interaction in its pathogenesis. Particularly, microglia play a crucial role in the development of chronic pain. To better understand the microglial contribution to chronic pain, specific regional and temporal manipulations of microglia are necessary. Recently, two new approaches have emerged that meet these demands. Chemogenetic tools allow the expression of designer receptors exclusively activated by designer drugs (DREADDs) specifically in microglia. Similarly, optogenetic tools allow for microglial manipulation via the activation of artificially expressed, light-sensitive proteins. Chemo- and optogenetic manipulations of microglia in vivo are powerful in interrogating microglial function in chronic pain. This review summarizes these emerging tools in studying the role of microglia in chronic pain and highlights their potential applications in microglia-related neurological disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neuropathic pain is a chronic condition that results in pain hypersensitivity and allodynia (pain responses to normally innocuous stimuli) after nerve damage that can occur after a host of insults, such as physical injury, diabetes, or autoimmune diseases [1]. When tissue damage has healed, however, neuropathic pain does not resolve [2]. A growing body of evidence indicates that microglia, as central nervous system (CNS) resident immune cells, play an important role in the pathogenesis of neuropathic pain [3,4,5]. Indeed, specific ablation or inhibition of microglia prevents the development of neuropathic pain [6,7,8]. In addition, recent progress highlights intimate microglia-neuron interactions in chronic pain [5, 9].
Microglia undergo functional changes during chronic pain states. In homeostatic conditions, microglia dynamically respond to changes in the microenvironment with their remarkably motile processes [10,11,12,13]. However, in response to peripheral nerve injury, microglia become activated and promote chronic pain. A major known mechanism for microglia to contribute to this process is through the release of cytokines and other mediators, such as interleukin-1 beta (IL-1β), IL-6, tumor necrosis factor alpha (TNFα), prostaglandin E2, brain-derived neurotrophic factor, and reactive oxygen species. These signals can lead to chronic pain [5, 14]. In addition to diffusible molecules, microglia also contribute to chronic pain hypersensitivity by adopting new functional roles, such as altered transcriptional activation and phagocytosis [9]. However, it is important to note that microglial activation during chronic pain states is not always detrimental because they are indeed heterogeneous [15, 16]. Recent findings identified a subpopulation of activated microglia playing a beneficial role in resolving chronic pain after peripheral nerve injury [17].
The cellular mechanisms of microglia in chronic pain have been investigated through pharmacological approaches. For example, systemic inhibition of microglia and macrophages by the broad inhibitor minocycline attenuates pain hypersensitivity [7, 8]. However, minocycline also has inhibitory effects on other cells, such as neurons, astrocytes, and T-cells [18,19,20]. Inhibitors of microglia through the colony-stimulating factor 1 receptor (CSF-1R), such as PLX5622 [21, 22] and neutralizing colony-stimulating factor 1 (CSF-1) antibody, also reduce microglial activation and proliferation in the spinal dorsal horn after nerve injury and alter pain responses [21, 23]. Specifically, mechanical allodynia and thermal hyperalgesia are attenuated in CSF-1 inhibitor-treated mice with chronic pain [21, 23,24,25,26]. However, CSF-1R inhibition induces off-target effects in other peripheral immune cells expressing the receptor [22, 27].
The importance of spinal microglia in the development of chronic pain has also been demonstrated by using genetic approaches to remove key microglial genes such as P2X4 [28], P2X7 [29], CX3CR1 [30], TRPM2 [31], P2Y12 [32], and Hv1 [33]. In addition, multiple Cre lines including CX3CR1 [34], Sal1 [35], TMEM119 [36], HexB [37], and P2Y12 [38] have been developed to target microglia. However, the development of advanced tools that provide temporal accuracy and spatial specificity is still needed. In the past few years, precise and selective methods for manipulating microglia have been used to study their involvement in chronic pain. Here, we introduce recent advances in how microglia control the pathophysiology of pain by using chemogenetic and optogenetic approaches.
Chemogenetic Approaches in Microglia
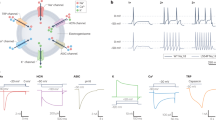
Chemogenetic approaches refer to the expression and activation of DREADDs [39, 40]. DREADDs allow the selective interrogation of multiple G-protein-coupled receptor (GPCR) signaling cascades, including Gq, Gi, and Gs in various cell types [40]. DREADDs can be specifically activated in a cell type of choice by locally or systemically applying a specific ligand, such as clozapine N-oxide (CNO), with minimal off-target effects. Chemogenetic approaches have historically been used in neurons to interrogate the neuronal circuitry underlying behaviors [41,42,43]. Similarly, numerous studies have also applied DREADD approaches in astrocytes to investigate their physiological alterations in GPCR-mediated Ca2+ signaling [44], memory [45,46,47], neuroinflammation [48], and pain [49, 50]. Microglia express a number of GPCRs that are important for various microglial functions [51]. In particular, the microglial signature P2Y12 receptor is a Gi-coupled GPCR involved in the chemotaxis of processes towards ATP/ADP, which can occur after injury [52] and during the development of neuropathic pain [32, 53, 54]. Of the available chemogenetic GPCRs, Gi- (e.g. hM4Di) and Gq-signaling (e.g. hM3Dq) DREADDs have been used to investigate the functions of microglia in the CNS (Fig. 1).
Chemogenetic and optogenetic approaches in microglia. A Viral-vector-mediated (e.g. AVV) or gene knock-in approaches (e.g. Cx3cr1cre) introduce DREADDs or opsins specifically in microglia. B Activation of DREADDs (chemogenetics) or opsins (optogenetics) in microglia by a DREADD ligand (e.g. CNO) or specific light stimulation. C In microglia, a DREADD ligand binds to hM3Dq or hM4Di, to activate Gq- or Gi-coupled signaling; light stimulation of opsins opens non-selective cation channels (such as ReaChR or ChETA) or proton pumps (such as ArchT)
Chemogenetic Manipulation of CNS Microglia
Several studies have used chemogenetic approaches in microglia (Table 1). The Watkins’s lab was the first to use Gi and Gq DREADDs in rat microglia by viral expression [55, 56]. Spinal microglia were transfected with adeno-associated virus (AAV) 9 containing DREADDs driven under the CD68 promoter. AAVs have been successfully used to target various cell types in the CNS. However, microglial transduction in vivo is complicated, and it may not achieve robust transfection levels [57]. However, in the Watkins studies, microglial DREADD transfection by AAV led to functional DREADD expression in the spinal cord. Microglial Gi DREADD activation can attenuate pro-inflammatory signaling including through the nuclear factor of the kappa light polypeptide gene enhancer in B-cell inhibitor alpha, NLR family pyrin domain-containing 3 (NLRP3), and IL-1β [55]. On the other hand, Gq DREADDs mediate microglial activation and cytokine release [58, 59], potentially through the mobilization of intracellular Ca2+. Thus, Gq DREADD activation induces pro-inflammatory mediator production, while Gi DREADD activation inhibits lipopolysaccharide- (LPS) and chemokine (C-C motif) ligand 2-induced inflammatory signaling in vitro [56]. One potential confounder in these studies is the possibility that microglia might react with an immune response to AAV transfection [57]. However, it has been reported that AAV vectors (in contrast to adenovirus-based vectors) cause minimal immune reactions [60]. Nonetheless, careful use of adequate controls (DREADD expression without DREADD ligand administration) is essential to distinguish the effects of chemogenetic manipulation from the side effects of immune reaction to viral infection.
Other studies have more commonly applied gene knock-in approaches to selectively express Gi/Gq DREADD in microglia [61,62,63]. The C-X3-C motif chemokine receptor 1 (CX3CR1) is highly expressed by microglia in the CNS and cells of mononuclear origin in the periphery [64]. The use of constitutive Cx3cr1cre/+:R26LSL-hM4Di/+ mice results in Gi DREADD expression in all CX3CR1-expressing cells including microglia and monocytes [61]. To exclusively express Gi DREADD in microglia but not peripheral monocytes, researchers have used inducible Cx3cr1creER/+:R26LSL-hM4Di/+ mice [63]. Due to the fact that blood CX3CR1+ cells have rapid turnover while microglia are longer-lived, it is possible to achieve greater microglial specificity by waiting for cell turnover after tamoxifen administration [34]. Indeed, DREADDs are expressed and co-localized only with Iba1+ microglia in the spinal cord and brain 4 weeks after tamoxifen injection [63]. Using these genetic knock-in mice expressing Gi/Gq DREADDs in microglia, further studies have interrogated their function and underlying mechanisms in chronic pain (Fig. 2) [61,62,63].
Mechanisms underlying chronic pain regulation by chemogenetic and optogenetic manipulation of microglia. A Chemogenetic activation of Gi DREADD attenuates neuropathic pain after peripheral nerve injury. After L4 spinal nerve transection (SNT) (1), CNO activation of Gi DREADD-expressing microglia (2) leads to microglial inhibition. Subsequently, SNT-induced microglial upregulation of IRF8 and IL-1β is inhibited (3). Thus, chemogenetic inhibition of microglia normalizes neuronal hyperactivity (4) and attenuates chronic pain behaviors after SNT (5). B Optogenetic activation of spinal microglia triggers chronic “microgliogenic” pain. Optogenetic stimulation of spinal microglia expressing ReaChR with red light (625 nm) (1) activates microglia and increases the Ca2+-dependent release of IL-1β (2), which sensitizes neuronal activity (3), leading to chronic pain behaviors (4)
In addition to pain research, chemogenetic methods were also recently applied to microglia to investigate their functions in inflammation, vascular interaction, and aversive behaviors. For example, inducible Cx3cr1creER/+:R26LSL-hM3Dq/+ mice were used for the functional expression of microglial Gq DREADDs [65]. Gq DREADD activation induced intracellular Ca2+ elevation and the phagocytosis of FluoSpheres in primary microglia. Unexpectedly, chronic Gq DREADD activation attenuated the LPS-induced increase of pro-inflammatory cytokines, including TNFα, IL-1β, and IL-6 in the mouse brain. In line with these results, chronic Gq DREADD activation in microglia robustly increased social exploration 2 h after LPS-induced inflammation [65]. Recently, a Cre-inducible lentiviral vector has been used to express DREADDs in dorsal striatal microglia of Cx3cr1creER/+ mice [66]. Using this approach, both microglial Gq DREADD activation in naïve mice or LPS administration led to conditioned place aversion. Interestingly, microglial Gi DREADD activation before LPS administration prevents the development of conditioned place aversion without affecting markers of systemic inflammation [66]. Chemogenetic methods have been also applied to microglia to investigate neurovascular coupling changes [67]. Cx3cr1creER/+:R26LSL-hM3Dq-CGaMP5g–tdTomato/+ mice were used for the expression of microglial Gq DREADDs. Activation of Gq DREADDs in microglia that interact with arterioles and microvessels in the cortex led to the withdrawal of perivascular microglial processes around arterioles and reduced the cerebral blood flow in response to whisker stimulation. Taken together, these bi-directional chemogenetic approaches have proven to be powerful tools in interrogating microglial function in the brain.
Chemogenetic Manipulation of Microglia in Chronic Pain
The first study applying chemogenetic approaches through the viral expression of DREADDs in microglia studied the contribution of microglia to morphine-induced persistent sensitization in rats [55]. While opiates have been regularly used for pain treatment, they paradoxically induce nociceptive sensitization known as opioid-induced hyperalgesia [68]. Exposure to opioids in healthy individuals has been demonstrated to lead to hyperalgesia in many clinical studies, although large-scale trials cannot be performed in human subjects for ethical reasons [69, 70] Morphine-induced persistent sensitization is associated with microglial inflammasome activation in the spinal cord [55]. Microglial Gi DREADD activation reduces pro-inflammatory signaling and prevents morphine-induced persistent sensitization. Similar to microglial Gi DREADD activation, pharmacological blockade of toll-like receptor 4, P2X7, or the inflammasome can all independently block morphine-induced sensitization [55]. Thus, the mechanism underlying the action of Gi DREADD in microglia may be related to Gi inhibition of Ca2+ elevation for pro-inflammatory cytokine production or release [58, 71].
Using viral expression of both Gq and Gi DREADD specifically in microglia, the group further studied the role of microglia in chronic pain in rats [56]. Gi DREADD activation in microglia rapidly reversed allodynia in neuropathic pain conditions. Mechanistically, microglial Gi DREADD activation attenuated the level of inflammatory mediators including nitric oxide (NO) and IL-1β. Microglial Gq DREADD activation, on the other hand, was able to induce allodynia in naïve male rats and increased the expression of pro-inflammatory mediators, such as NO, TNFα, Il-1β, and IL-6 [56].
After Cre-inducible DREADD mice were generated in 2016 [72], both Kiguchi’s group and our group investigated microglial function in chronic pain in mice selectively expressing DREADDs in microglia. Using constitutive Cx3cr1cre/+:R26LSL-hM4Di/+ mice, Kiguchi’s group showed that microglia expressing Gi DREADDs in the spinal cord, upon activation, alleviate pain sensitization after partial sciatic nerve ligation (PSL) [61]. Using inducible Cx3cr1creER/+:R26LSL-hM4Di/+ mice, we showed that Gi DREADDs are specifically expressed in microglia in adult mice [63]. Microglial Gi DREADD activation 3 days before L4 spinal nerve transection (SNT) delayed the development of allodynia while activation 3 days after SNT attenuated mechanical allodynia [63].
Several potential mechanisms underlying microglial Gi DREADD in pain attenuation have been proposed [63]. First, activation of microglial Gi DREADD signaling prevents microglial proliferation, known as the main source of microgliosis in neuropathic pain [4, 23]. Second, SNT-upregulated expression of interferon regulatory factor (IRF) 8, a transcription factor implicated in microglial transition to a reactive state [73], is prevented by Gi DREADD activation. Similarly, Il-1β as a critical mediator of neuropathic pain [73, 74], is also decreased by microglial Gi DREADD activation. Third, C-fiber-evoked field potentials in vivo are reduced upon activation of microglial Gi DREADD. These results complement and expand previous findings that selective microglial activation in the spinal cord promotes synaptic strengthening and synaptic plasticity between primary afferent C-fibers and spinal neurons [25, 75]. Together, these results indicate that Gi DREADD manipulation in microglia attenuates chronic pain by inhibiting microglial proliferation, neuroinflammation, and synaptic potentiation (Fig. 2A).
Microglial function in chronic pain was further investigated by using Gq DREADD in constitutive Cx3cr1cre/+:R26LSL-hM3Dq/+ mice [62]. After Gq DREADD activation, naïve male mice displayed allodynia and hyperalgesia. Further analysis showed that Gq DREADD activation led to a significant upregulation of inflammatory mediators (IL-1β, TNFα, CCL3, and CCL4) and microglial markers (Iba1, CD11b, IRF5, and IRF7). Importantly, these results point to a sex-specific mechanism, as both the behavioral correlates of pain and their potential underlying inflammation occurred in male but not female mice. After microglial ablation by PLX3397, an inhibitor of CSF1-R, Gq DREADD activation by CNO administration does not induce chronic pain hypersensitivity or the upregulation of inflammatory markers in male mice, further providing evidence that the chemogenetic activation of microglia is necessary for the development of chronic pain.
These findings are part of a larger literature finding an interesting sex-dependent microglial function in chronic pain. Although microglial activation (microgliosis) develops in both sexes in neuropathic pain models, emerging reports suggest that microglial inhibition resolves pain only in male mice. For example, microglia-targeted inhibitors (minocycline as well as inhibitors of TLR4, P2X4, or p38 mitogen-activated protein kinases) are effective in attenuating neuropathic pain in male but not female rodents [76,77,78]. One potential explanation is the involvement of T-cells in the development of chronic pain in females only, while males depend on microglia-related mechanisms [76]. In line with this idea, activation of Gi DREADD in CX3CR1+ cells attenuates mechanical allodynia after PSL only in male mice [61]. While Gq DREADD microglia can initiate sex-dependent differences in pain responses, we have recently found that microglial Gi DREADD activation attenuates neuropathic pain in both male and female mice after SNT [63]. This discrepancy might be explained by different genetic manipulations (constitutive Cx3cr1cre/+ versus inducible Cx3cr1creER/+ mouse lines impacting different sets of cell classes) or different CNO dose paradigms (one time-point only at 10 mg/kg vs three times at 5 mg/kg/day). Further, no sex differences have been reported in chronic pain attenuation after CX3CR1+ cells are genetically ablated [6]. Future studies are needed to understand these potential discrepancies and determine the circumstances under which microglia may engage in sex-dependent chronic pain responses.
Optogenetic Approaches in Microglia
Optogenetics fuse genetic and optical procedures to allow the manipulation of specific cell populations, conferring the unique capability to sense and respond to light through light-sensitive proteins in behaving animals [79]. All known organisms express photon-sensitive receptor proteins, called rhodopsins. The main types of opsins found in microorganisms are bacteriorhodopsins and halorhodopsins, which are light-driven ion pumps/channels such as channelrhodopsins (ChRs), and sensors such as sensory rhodopsin [80]. Optogenetic approaches have been widely used to drive the depolarization or hyperpolarization of selected neurons in response to specific wavelengths of light, allowing scientists to interrogate complex circuits underlying behavior [81, 82] including pain sensation [83]. Recent advances have also enabled optogenetic approaches to be applied to glial cells such as astrocytes and microglia [84,85,86,87]. For example, optogenetic approaches have been used to dissect astrocyte functions in breathing [84], memory [45], and epilepsy [85]. Furthermore, a recent study used ChR2, a non-selective, depolarizing cation channel, to selectively activate astrocytes. Depolarized spinal astrocytes elicited chronic pain behaviors by inducing ATP release [86]. In the periphery, optogenetic activation of ChR2-expressing macrophages in the heart improves the electrical connections underlying conduction [88]. Here, we introduce how optogenetics has been applied to microglia to dissect their function in the CNS, with a particular focus on chronic pain (Fig. 1).
Optogenetic Manipulation of CNS Microglia
Ionotropic signaling is an overlook mechanism underlying microglial interactions with the brain microenvironment [89]. Unlike neurons, microglia have few voltage-gated Na+ or Ca2+ channels in vivo. Microglia mediate ionic fluxes using multiple ion channels including K+ channels [90, 91], proton channels [92], transient receptor potential channels [93], pannexin-1 [94, 95], and purinergic ionotropic receptors [96]. The changes in microglial membrane potential in response to ion channel activation under pathological conditions are associated with the reactive microglial transition. For instance, prolonged increased K+ channel conductance often precedes the reactive state transition [51, 97]. K+ channels are also essential for microglial process surveillance and chemotactic responses to extracellular ATP/tissue injury [91, 98, 99]. However, it is not known whether the changes in membrane potential are either necessary or sufficient for microglial activation. Recent advances applying optogenetic approaches to microglia allow us to address these early questions [87, 100].
Only a few studies have used optogenetic approaches in microglia so far (Table 2). In a proof-of-concept study by Yamanaka’s group, ChR2 was expressed specifically in microglia by using transgenic Iba1-tTA:tetOChR2(CS128S)-EYFP mice [101]. Blue light stimulation depolarized microglia indicating its functional expression, but no further studies were conducted using these mice. For the first time, red-activated ChR (ReaChR) was expressed in microglia using Cx3cr1creER/+:R26LSL-ReaChR/+ mice [87]. The advantage of using ReaChR (a newer generation of non-selective cation channels) compared with ChR2 is its activation by red light, which has better penetration deeper into tissue with less light scatter than the blue/green light for ChR2 activation. In addition, the ReaChR current can be maintained with far less inactivation occurring during light stimulation [102]. Selective microglial ReaChR expression can be achieved using Cx3cr1creER/+:R26LSL-ReaChR/+ mice. In the spinal cord, rhodopsin protein is only co-localized with Iba1+ microglia [87]. ReaChR expression is also functional as red-light stimulation induces inward currents in spinal microglia, resulting in their depolarization. In addition, pro-inflammatory cytokines such as IL-1β are secreted by primary microglia after red-light stimulation and this requires extracellular Ca2+ influx [87]. Thus, optogenetic depolarization of spinal microglia is sufficient for Ca2+-dependent cytokine release.
Similarly, ChETA (a modified form of ChR2) was expressed in microglia using Cx3cr1creER/+:R26LSL-ChETA-tdTomato/+ mice [100]. ChETA activated by blue-light induced microglial depolarization and slowed the chemotaxis of processes in response to laser-induced tissue damage [100]. Microglia rapidly hyperpolarize when sensing ATP or neuronal hyperactivity [103]. Indeed, P2Y12-coupled K+ channel activation is part of the mechanism for rapid chemotactic reactions to laser injury or basal motility [91, 98, 99]. A slower chemotaxis response induced by optogenetic microglial depolarization indicates that ATP-mediated hyperpolarization is not only a concomitant phenomenon of microglial activation but is required for the rapid expansion of microglial processes towards injury. One caveat of using blue light stimulation is the potential off-target effects. For instance, a study showed that microglia alter inflammatory-related gene expression with different levels of blue light stimulation [104]. Nevertheless, the results using ChETA indicate a potential correlation between the membrane potential and the chemotaxis of microglial processes. The light-activated proton pump archaerhodopsin (ArchT) has also been expressed in microglia using Cx3cr1creER/+:R26LSL-ArchT-EGFP/+ mice [100]. Unlike ChR2 and its variants, ArchT activation by yellow/green light results in hyperpolarization. However, the ArchT-mediated hyperpolarization of microglia does not alter the electrophysiological responses of microglial to laser-induced tissue damage, nor does it affect chemotactic responses.
Microglia sense neuronal activity and the brain environment via Ca2+ signaling [59, 105]. Indeed, increased Ca2+ in microglia is strongly correlated with pathophysiological activation such as neuroinflammation [106], seizures [59], stroke [107], and neurodegeneration [108]. Since ChR2 and its derivatives are Ca2+-permeable ion channels [109], optogenetic activation of microglia allows for the direct manipulation of Ca2+ influx. As a result, ReaChR activation of microglia leads to Ca2+-independent cytokine release [87]. On the contrary, in Ca2+-free extracellular solutions, microglial chemotaxis to damage sites is significantly slowed, similar to the increased ionic influx during ChETA activation [100]. Therefore, these results suggest that optogenetic depolarization of microglia inhibits Ca2+ elevation, thus slowing the chemotaxis of microglial processes. The underlying mechanism might be due to the reduction of the driving force for Ca2+ during depolarization in microglia. Interestingly, previous studies found that the removal of extracellular Ca2+ alone induces the convergence of microglial processes, similar to that reported in seizures and stroke [110, 111]. Future experiments using in vivo Ca2+ imaging are needed to directly investigate whether optogenetic activation of microglia increases or decreases microglial Ca2+ signaling.
Optogenetic Manipulation of Microglia in Chronic Pain
By using Cx3cr1creER/+:R26LSL-ReaChR/+ mice to exclusively express ReaChR in microglia, spinal microglia can be depolarized in real time to examine their function in pain behaviors (Fig. 2B). Red-light stimulation can be delivered locally to the lumbar spinal cord through optic fibers. After light stimulation (30 min at 20 Hz,) mechanical allodynia is evident one hour after stimulation and lasts for up to one week in mice [87]. These results are remarkable in that short-term optogenetic stimulation of spinal microglia alone induced long-lasting pain behaviors. The mere stimulation of spinal microglia through optogenetics in the absence of any inflammatory challenge, or nerve damage-elicited chronic pain [87, 112], suggests the intriguing possibility of “microgliogenic” pain that originates from microglial activation in the CNS.
Mechanistically, optogenetic stimulation of microglial ReaChR increases microglial proliferation, neuronal activity, and nociceptive transmission [87]. For example, C-fiber-evoked field potentials and neuronal C-fos expression in the dorsal horn are significantly increased after microglial optogenetic stimulation. Interestingly, IL-1β expression is increased 1–3 days after light stimulation of microglial ReaChR, which could be due to increased expression of NLRP3 inflammasome components and caspase-1. The IL-1 receptor antagonist IL-1ra is sufficient to prevent increased C-fiber-evoked field potentials by light stimulation and alleviate light-induced mechanical allodynia. Thus, optogenetic stimulation of spinal microglia triggers IL-1β release, which increases the neuronal activity underlying chronic pain behaviors (Fig. 2B). In sum, optogenetics allows specific and temporally-controlled manipulation of microglia to study their function in pain. This may provide additional benefit over chemogenetic approaches in that the optical stimulation has better spatial and temporal resolution.
Conclusions and Outlook
Chemogenetics and optogenetics are two emerging approaches recently applied in the field of microglia research. DREADDs and opsin expression can be limited to microglia either by viral injection (e.g., AAV) or by promoter-driven conditional expression (e.g., CX3CR1). Unless activated, these proteins have no biological effects. Upon activation, existing DREADDs and opsins allow for a range of modulatory effects on microglia, including depolarization, hyperpolarization, and GPCR signal transduction. Chemo- and optogenetic manipulations of microglia are able to inhibit nerve injury-induced neuropathic pain or directly trigger chronic “microgliogenic” pain. However, it is important to note the limitations of chemo- and optogenetic approaches, as both use artificially-engineered proteins activated by designed stimuli. With chronic DREADD approaches, it has been suggested that repeated administration of CNO may lead to clozapine accumulation, which may have side effects unrelated to DREADDs [113]. The optogenetic stimulation of microglia might be unnaturally strong. In addition, it should be noted that effects may vary depending on the type of opsin and the frequency/intensity of light stimulation.
Here, we highlight the future of investigations of microglia by applying chemo- and optogenetic tools. (1) Microglia play a central role in many pathophysiological processes, such as in epilepsy [114], stroke [115], neurodegeneration [116, 117], depressive-like behaviors [118], memory deficits [119], and autoimmune neurology [120]. These new microglial tools will help illuminate the microglial mechanisms of neurological disorders. (2) Supraspinal microglial activation is also implicated in chronic pain [121,122,123]. Future studies will apply microglial tools to study their function in pain sensation, aversion, and comorbidities during chronic pain conditions. (3) Given the heterogeneity of microglia in neuropathic pain [15, 16] and their beneficial role in resolving chronic pain [17], it is unknown whether this heterogeneity also occurs when manipulating microglia using chemo- or optogenetic tools. Future studies will apply microglial tools to harness the beneficial function of alternatively-activated microglia during chronic pain conditions. (4) The ability of chemo- and optogenetic tools to directly manipulate Ca2+ levels allows the investigation of the role of Ca2+ signaling in microglia. Interrogation of the downstream Gi and Gq signaling in microglia is also made possible. Thus, the recent advances in microglial chemo- and optogenetic manipulations highlight the importance and novelty of these emerging tools in studying the function of microglia in neurological diseases, particularly in chronic pain.
References
Colloca L, Ludman T, Bouhassira D, Baron R, Dickenson AH, Yarnitsky D. Neuropathic pain. Nat Rev Dis Primers 2017, 3: 17002.
Kuner R, Flor H. Structural plasticity and reorganisation in chronic pain. Nat Rev Neurosci 2017, 18: 113.
Scholz J, Woolf CJ. The neuropathic pain triad: Neurons, immune cells and glia. Nat Neurosci 2007, 10: 1361–1368.
Tsuda M, Inoue K, Salter MW. Neuropathic pain and spinal microglia: A big problem from molecules in “small” glia. Trends Neurosci 2005, 28: 101–107.
Zhuo M, Wu G, Wu LJ. Neuronal and microglial mechanisms of neuropathic pain. Mol Brain 2011, 4: 31.
Peng J, Gu N, Zhou L, Eyo BU, Murugan M, Gan WB, et al. Microglia and monocytes synergistically promote the transition from acute to chronic pain after nerve injury. Nat Commun 2016, 7: 12029.
Raghavendra V, Tanga F, DeLeo JA. Inhibition of microglial activation attenuates the development but not existing hypersensitivity in a rat model of neuropathy. J Pharmacol Exp Ther 2003, 306: 624–630.
Ledeboer A, Sloane EM, Milligan ED, Frank MG, Mahony JH, Maier SF, et al. Minocycline attenuates mechanical allodynia and proinflammatory cytokine expression in rat models of pain facilitation. Pain 2005, 115: 71–83.
Inoue K, Tsuda M. Microglia in neuropathic pain: Cellular and molecular mechanisms and therapeutic potential. Nat Rev Neurosci 2018, 19: 138–152.
Nimmerjahn A, Kirchhoff F, Helmchen F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 2005, 308: 1314–1318.
Davalos D, Grutzendler J, Yang G, Kim JV, Zuo Y, Jung S, et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci 2005, 8: 752–758.
Eyo UB, Wu LJ. Microglia: Lifelong patrolling immune cells of the brain. Prog Neurobiol 2019, 179: 101614.
Liu YU, Ying Y, Li Y, Eyo UB, Chen T, Zheng J, et al. Neuronal network activity controls microglial process surveillance in awake mice via norepinephrine signaling. Nat Neurosci 2019, 22: 1771–1781.
Inoue K, Tsuda M. Microglia and neuropathic pain. Glia 2009, 57: 1469–1479.
Sideris-Lampretsas G, Malcangio M. Microglial heterogeneity in chronic pain. Brain Behav Immun 2021, 96: 279–289.
Franco R, Fernández-Suárez D. Alternatively activated microglia and macrophages in the central nervous system. Prog Neurobiol 2015, 131: 65–86.
Kohno K, Shirasaka R, Yoshihara K, Mikuriya S, Tanaka K, Takanami K, et al. A spinal microglia population involved in remitting and relapsing neuropathic pain. Science 2022, 376: 86–90.
González JC, Egea J, Del Carmen GM, Fernandez-Gomez FJ, Sánchez-Prieto J, Gandía L, et al. Neuroprotectant minocycline depresses glutamatergic neurotransmission and Ca2+ signalling in hippocampal neurons. Eur J Neurosci 2007, 26: 2481–2495.
Song ZP, Xiong BR, Guan XH, Cao F, Manyande A, Zhou YQ, et al. Minocycline attenuates bone cancer pain in rats by inhibiting NF-κB in spinal astrocytes. Acta Pharmacol Sin 2016, 37: 753–762.
Szeto GL, Pomerantz JL, Graham DR, Clements JE. Minocycline suppresses activation of nuclear factor of activated T cells 1 (NFAT1) in human CD4+ T cells. J Biol Chem 2011, 286: 11275–11282.
Lee S, Shi XQ, Fan A, West B, Zhang J. Targeting macrophage and microglia activation with colony stimulating factor 1 receptor inhibitor is an effective strategy to treat injury-triggered neuropathic pain. Mol Pain 2018, 14: 1744806918764979.
Lei F, Cui N, Zhou C, Chodosh J, Vavvas DG, Paschalis EI. CSF1R inhibition by a small-molecule inhibitor is not microglia specific; affecting hematopoiesis and the function of macrophages. Proc Natl Acad Sci U S A 2020, 117: 23336–23338.
Gu N, Peng J, Murugan M, Wang X, Eyo UB, Sun D, et al. Spinal microgliosis due to resident microglial proliferation is required for pain hypersensitivity after peripheral nerve injury. Cell Rep 2016, 16: 605–614.
Tang Y, Liu L, Xu D, Zhang W, Zhang Y, Zhou J, et al. Interaction between astrocytic colony stimulating factor and its receptor on microglia mediates central sensitization and behavioral hypersensitivity in chronic post ischemic pain model. Brain Behav Immun 2018, 68: 248–260.
Zhou LJ, Peng J, Xu YN, Zeng WJ, Zhang J, Wei X, et al. Microglia are indispensable for synaptic plasticity in the spinal dorsal horn and chronic pain. Cell Rep 2019, 27: 3844-3859.e6.
Sawicki CM, Kim JK, Weber MD, Faw TD, McKim DB, Madalena KM, et al. Microglia promote increased pain behavior through enhanced inflammation in the spinal cord during repeated social defeat stress. J Neurosci 2019, 39: 1139–1149.
Green KN, Hume DA. On the utility of CSF1R inhibitors. Proc Natl Acad Sci U S A 2021, 118: e2019695118.
Ulmann L, Hatcher JP, Hughes JP, Chaumont S, Green PJ, Conquet F, et al. Up-regulation of P2X4 receptors in spinal microglia after peripheral nerve injury mediates BDNF release and neuropathic pain. J Neurosci 2008, 28: 11263–11268.
Chessell IP, Hatcher JP, Bountra C, Michel AD, Hughes JP, Green P, et al. Disruption of the P2X7 purinoceptor gene abolishes chronic inflammatory and neuropathic pain. Pain 2005, 114: 386–396.
Staniland AA, Clark AK, Wodarski R, Sasso O, Maione F, D’Acquisto F, et al. Reduced inflammatory and neuropathic pain and decreased spinal microglial response in fractalkine receptor (CX3CR1) knockout mice. J Neurochem 2010, 114: 1143–1157.
Haraguchi K, Kawamoto A, Isami K, Maeda S, Kusano A, Asakura K, et al. TRPM2 contributes to inflammatory and neuropathic pain through the aggravation of pronociceptive inflammatory responses in mice. J Neurosci 2012, 32: 3931–3941.
Gu N, Eyo UB, Murugan M, Peng J, Matta S, Dong H, et al. Microglial P2Y12 receptors regulate microglial activation and surveillance during neuropathic pain. Brain Behav Immun 2016, 55: 82–92.
Peng J, Yi MH, Jeong H, McEwan PP, Zheng J, Wu G, et al. The voltage-gated proton channel Hv1 promotes microglia-astrocyte communication and neuropathic pain after peripheral nerve injury. Mol Brain 2021, 14: 99.
Parkhurst CN, Yang G, Ninan I, Savas JN, Yates JR III, Lafaille JJ, et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 2013, 155: 1596–1609.
Buttgereit A, Lelios I, Yu X, Vrohlings M, Krakoski NR, Gautier EL, et al. Sall1 is a transcriptional regulator defining microglia identity and function. Nat Immunol 2016, 17: 1397–1406.
Kaiser T, Feng G. Tmem119-EGFP and Tmem119-CreERT2 transgenic mice for labeling and manipulating microglia. eNeuro 2019, 6: ENEURO.0448–18.2019.
Masuda T, Amann L, Sankowski R, Staszewski O, Lenz M, D’Errico P, et al. Novel Hexb-based tools for studying microglia in the CNS. Nat Immunol 2020, 21: 802–815.
McKinsey GL, Lizama CO, Keown-Lang AE, Niu A, Santander N, Larpthaveesarp A, et al. A new genetic strategy for targeting microglia in development and disease. eLife 2020, 9: e54590.
Armbruster BN, Li X, Pausch MH, Herlitze S, Roth BL. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc Natl Acad Sci U S A 2007, 104: 5163–5168.
Urban DJ, Roth BL. DREADDs (designer receptors exclusively activated by designer drugs): Chemogenetic tools with therapeutic utility. Annu Rev Pharmacol Toxicol 2015, 55: 399–417.
Krashes MJ, Koda S, Ye C, Rogan SC, Adams AC, Cusher DS, et al. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J Clin Invest 2011, 121: 1424–1428.
Garner AR, Rowland DC, Hwang SY, Baumgaertel K, Roth BL, Kentros C, et al. Generation of a synthetic memory trace. Science 2012, 335: 1513–1516.
Roth BL. DREADDs for neuroscientists. Neuron 2016, 89: 683–694.
Chai H, Diaz-Castro B, Shigetomi E, Monte E, Octeau JC, Yu X, et al. Neural circuit-specialized astrocytes: Transcriptomic, proteomic, morphological, and functional evidence. Neuron 2017, 95: 531-549.e9.
Adamsky A, Kol A, Kreisel T, Doron A, Ozeri-Engelhard N, Melcer T, et al. Astrocytic activation generates De novo neuronal potentiation and memory enhancement. Cell 2018, 174: 59-71.e14.
Nam MH, Han KS, Lee J, Won W, Koh W, Bae JY, et al. Activation of astrocytic μ-opioid receptor causes conditioned place preference. Cell Rep 2019, 28: 1154-1166.e5.
Kol A, Adamsky A, Groysman M, Kreisel T, London M, Goshen I. Astrocytes contribute to remote memory formation by modulating hippocampal–cortical communication during learning. Nat Neurosci 2020, 23: 1229–1239.
Kim JH, Rahman MH, Lee WH, Suk K. Chemogenetic stimulation of the G i pathway in astrocytes suppresses neuroinflammation. Pharmacol Res Perspect 2021, 9: e00822.
Xu Q, Ford NC, He S, Huang Q, Anderson M, Chen Z, et al. Astrocytes contribute to pain gating in the spinal cord. Sci Adv 2021, 7: eabi6287.
Lu J, Yang L, Xu Y, Ai L, Chen J, Xiong F, et al. The modulatory effect of motor cortex astrocytes on diabetic neuropathic pain. J Neurosci 2021, 41: 5287–5302.
Kettenmann H, Hanisch UK, Noda M, Verkhratsky A. Physiology of microglia. Physiol Rev 2011, 91: 461–553.
Haynes SE, Hollopeter G, Yang G, Kurpius D, Dailey ME, Gan WB, et al. The P2Y12 receptor regulates microglial activation by extracellular nucleotides. Nat Neurosci 2006, 9: 1512–1519.
Kobayashi K, Yamanaka H, Fukuoka T, Dai Y, Obata K, Noguchi K. P2Y12 receptor upregulation in activated microglia is a gateway of p38 signaling and neuropathic pain. J Neurosci 2008, 28: 2892–2902.
Tozaki-Saitoh H, Tsuda M, Miyata H, Ueda K, Kohsaka S, Inoue K. P2Y12 receptors in spinal microglia are required for neuropathic pain after peripheral nerve injury. J Neurosci 2008, 28: 4949–4956.
Grace PM, Strand KA, Galer EL, Urban DJ, Wang X, Baratta MV, et al. Morphine paradoxically prolongs neuropathic pain in rats by amplifying spinal NLRP3 inflammasome activation. Proc Natl Acad Sci U S A 2016, 113: E3441–E3450.
Grace PM, Wang X, Strand KA, Baratta MV, Zhang Y, Galer EL, et al. DREADDed microglia in pain: Implications for spinal inflammatory signaling in male rats. Exp Neurol 2018, 304: 125–131.
Maes ME, Colombo G, Schulz R, Siegert S. Targeting microglia with lentivirus and AAV: Recent advances and remaining challenges. Neurosci Lett 2019, 707: 134310.
Hoffmann A, Kann O, Ohlemeyer C, Hanisch UK, Kettenmann H. Elevation of basal intracellular calcium as a central element in the activation of brain macrophages (microglia): Suppression of receptor-evoked calcium signaling and control of release function. J Neurosci 2003, 23: 4410–4419.
Umpierre AD, Bystrom LL, Ying Y, Liu YU, Worrell G, Wu LJ. Microglial calcium signaling is attuned to neuronal activity in awake mice. eLife 2020, 9: e56502.
Xiao X, Li J, McCown TJ, Samulski RJ. Gene transfer by adeno-associated virus vectors into the central nervous system. Exp Neurol 1997, 144: 113–124.
Saika F, Matsuzaki S, Kobayashi D, Ideguchi Y, Nakamura TY, Kishioka S, et al. Chemogenetic regulation of CX3CR1-expressing microglia using gi-DREADD exerts sex-dependent anti-allodynic effects in mouse models of neuropathic pain. Front Pharmacol 2020, 11: 925.
Saika F, Matsuzaki S, Kishioka S, Kiguchi N. Chemogenetic activation of CX3CR1-expressing spinal microglia using gq-DREADD elicits mechanical allodynia in male mice. Cells 2021, 10: 874.
Yi MH, Liu YU, Liu K, Chen T, Bosco DB, Zheng J, et al. Chemogenetic manipulation of microglia inhibits neuroinflammation and neuropathic pain in mice. Brain Behav Immun 2021, 92: 78–89.
Jung S, Aliberti J, Graemmel P, Sunshine MJ, Kreutzberg GW, Sher A, et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol Cell Biol 2000, 20: 4106–4114.
Binning W, Hogan-Cann AE, Yae Sakae D, Maksoud M, Ostapchenko V, Al-Onaizi M, et al. Chronic hM3Dq signaling in microglia ameliorates neuroinflammation in male mice. Brain Behav Immun 2020, 88: 791–801.
Klawonn AM, Fritz M, Castany S, Pignatelli M, Canal C, Similä F, et al. Microglial activation elicits a negative affective state through prostaglandin-mediated modulation of striatal neurons. Immunity 2021, 54: 225-234.e6.
Császár E, Lénárt N, Cserép C, Környei Z, Fekete R, Pósfai B, et al. Microglia modulate blood flow, neurovascular coupling, and hypoperfusion via purinergic actions. J Exp Med 2022, 219: e20211071.
Grace PM, Maier SF, Watkins LR. Opioid-induced central immune signaling: Implications for opioid analgesia. Headache 2015, 55: 475–489.
Mauermann E, Filitz J, Dolder P, Rentsch KM, Bandschapp O, Ruppen W. Does fentanyl lead to opioid-induced hyperalgesia in healthy volunteers? : A double-blind, randomized, crossover trial. Anesthesiology 2016, 124: 453–463.
Rowbotham MC, Wallace M. Evolution of analgesic tolerance and opioid-induced hyperalgesia over 6 months: Double-blind randomized trial incorporating experimental pain models. J Pain 2020, 21: 1031–1046.
Hayashi Y, Kawaji K, Sun L, Zhang X, Koyano K, Yokoyama T, et al. Microglial Ca2+-activated K+ channels are possible molecular targets for the analgesic effects of S-ketamine on neuropathic pain. J Neurosci 2011, 31: 17370–17382.
Zhu H, Aryal DK, Olsen RH, Urban DJ, Swearingen A, Forbes S, et al. Cre-dependent DREADD (designer receptors exclusively activated by designer drugs) mice. Genesis 2016, 54: 439–446.
Masuda T, Tsuda M, Yoshinaga R, Tozaki-Saitoh H, Ozato K, Tamura T, et al. IRF8 is a critical transcription factor for transforming microglia into a reactive phenotype. Cell Rep 2012, 1: 334–340.
Ji RR, Xu ZZ, Gao YJ. Emerging targets in neuroinflammation-driven chronic pain. Nat Rev Drug Discov 2014, 13: 533–548.
Clark AK, Gruber-Schoffnegger D, Drdla-Schutting R, Gerhold KJ, Malcangio M, Sandkühler J. Selective activation of microglia facilitates synaptic strength. J Neurosci 2015, 35: 4552–4570.
Sorge RE, Mapplebeck JCS, Rosen S, Beggs S, Taves S, Alexander JK, et al. Different immune cells mediate mechanical pain hypersensitivity in male and female mice. Nat Neurosci 2015, 18: 1081–1083.
Sorge RE, Lacroix-Fralish ML, Tuttle AH, Sotocinal SG, Austin JS, Ritchie J, et al. Spinal cord Toll-like receptor 4 mediates inflammatory and neuropathic hypersensitivity in male but not female mice. J Neurosci 2011, 31: 15450–15454.
Taves S, Berta T, Liu DL, Gan S, Chen G, Kim YH, et al. Spinal inhibition of p38 MAP kinase reduces inflammatory and neuropathic pain in male but not female mice: Sex-dependent microglial signaling in the spinal cord. Brain Behav Immun 2016, 55: 70–81.
Deisseroth K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat Neurosci 2015, 18: 1213–1225.
Govorunova EG, Sineshchekov OA, Li H, Spudich JL. Microbial rhodopsins: Diversity, mechanisms, and optogenetic applications. Annu Rev Biochem 2017, 86: 845–872.
Mirzayi P, Shobeiri P, Kalantari A, Perry G, Rezaei N. Optogenetics: Implications for Alzheimer’s disease research and therapy. Mol Brain 2022, 15: 20.
Sun Y, Li M, Cao S, Xu Y, Wu P, Xu S, et al. Optogenetics for understanding and treating brain injury: Advances in the field and future prospects. Int J Mol Sci 1800, 2022: 23.
Guo F, Du Y, Qu FH, Lin SD, Chen Z, Zhang SH. Dissecting the neural circuitry for pain modulation and chronic pain: Insights from optogenetics. Neurosci Bull 2022, 38: 440–452.
Gourine AV, Kasymov V, Marina N, Tang F, Figueiredo MF, Lane S, et al. Astrocytes control breathing through pH-dependent release of ATP. Science 2010, 329: 571–575.
Diaz Verdugo C, Myren-Svelstad S, Aydin E, van Hoeymissen E, Deneubourg C, Vanderhaeghe S, et al. Glia-neuron interactions underlie state transitions to generalized seizures. Nat Commun 2019, 10: 3830.
Nam Y, Kim JH, Kim JH, Jha MK, Jung JY, Lee MG, et al. Reversible induction of pain hypersensitivity following optogenetic stimulation of spinal astrocytes. Cell Rep 2016, 17: 3049–3061.
Yi MH, Liu YU, Umpierre AD, Chen T, Ying Y, Zheng J, et al. Optogenetic activation of spinal microglia triggers chronic pain in mice. PLoS Biol 2021, 19: e3001154.
Hulsmans M, Clauss S, Xiao L, Aguirre AD, King KR, Hanley A, et al. Macrophages facilitate electrical conduction in the heart. Cell 2017, 169: 510-522.e20.
Izquierdo P, Attwell D, Madry C. Ion channels and receptors as determinants of microglial function. Trends Neurosci 2019, 42: 278–292.
Nguyen HM, di Lucente J, Chen YJ, Cui Y, Ibrahim RH, Pennington MW, et al. Biophysical basis for Kv1.3 regulation of membrane potential changes induced by P2X4-mediated calcium entry in microglia. Glia 2020, 68: 2377–2394.
Madry C, Kyrargyri V, Arancibia-Cárcamo IL, Jolivet R, Kohsaka S, Bryan RM, et al. Microglial ramification, surveillance, and interleukin-1β release are regulated by the two-pore domain K+ channel THIK-1. Neuron 2018, 97: 299-312.e6.
Wu LJ, Wu G, Sharif MR, Baker A, Jia Y, Fahey FH, et al. The voltage-gated proton channel Hv1 enhances brain damage from ischemic stroke. Nat Neurosci 2012, 15: 565–573.
Wu LJ, Sweet TB, Clapham DE. International Union of Basic and Clinical Pharmacology. LXXVI. Current progress in the mammalian TRP ion channel family. Pharmacol Rev 2010, 62: 381–404.
Burma NE, Bonin RP, Leduc-Pessah H, Baimel C, Cairncross ZF, Mousseau M, et al. Blocking microglial pannexin-1 channels alleviates morphine withdrawal in rodents. Nat Med 2017, 23: 355–360.
Mousseau M, Burma NE, Lee KY, Leduc-Pessah H, Kwok CHT, Reid AR, et al. Microglial pannexin-1 channel activation is a spinal determinant of joint pain. Sci Adv 2018, 4: eaas9846.
Calovi S, Mut-Arbona P, Sperlágh B. Microglia and the purinergic signaling system. Neuroscience 2019, 405: 137–147.
Nörenberg W, Gebicke-Haerter PJ, Illes P. Inflammatory stimuli induce a new K+ outward current in cultured rat microglia. Neurosci Lett 1992, 147: 171–174.
Wu LJ, Vadakkan KI, Zhuo M. ATP-induced chemotaxis of microglial processes requires P2Y receptor-activated initiation of outward potassium currents. Glia 2007, 55: 810–821.
Swiatkowski P, Murugan M, Eyo UB, Wang Y, Rangaraju S, Oh SB, et al. Activation of microglial P2Y12 receptor is required for outward potassium currents in response to neuronal injury. Neuroscience 2016, 318: 22–33.
Laprell L, Schulze C, Brehme ML, Oertner TG. The role of microglia membrane potential in chemotaxis. J Neuroinflammation 2021, 18: 33.
Tanaka KF, Matsui K, Sasaki T, Sano H, Sugio S, Fan K, et al. Expanding the repertoire of optogenetically targeted cells with an enhanced gene expression system. Cell Rep 2012, 2: 397–406.
Lin JY, Knutsen PM, Muller A, Kleinfeld D, Tsien RY. ReaChR: A red-shifted variant of channelrhodopsin enables deep transcranial optogenetic excitation. Nat Neurosci 2013, 16: 1499–1508.
Eyo UB, Peng J, Swiatkowski P, Mukherjee A, Bispo A, Wu LJ. Neuronal hyperactivity recruits microglial processes via neuronal NMDA receptors and microglial P2Y12 receptors after status epilepticus. J Neurosci 2014, 34: 10528–10540.
Cheng KP, Kiernan EA, Eliceiri KW, Williams JC, Watters JJ. Blue light modulates murine microglial gene expression in the absence of optogenetic protein expression. Sci Rep 2016, 6: 21172.
Umpierre AD, Wu LJ. How microglia sense and regulate neuronal activity. Glia 2021, 69: 1637–1653.
Pozner A, Xu B, Palumbos S, Gee JM, Tvrdik P, Capecchi MR. Intracellular calcium dynamics in cortical microglia responding to focal laser injury in the PC: : G5-tdT reporter mouse. Front Mol Neurosci 2015, 8: 12.
Liu L, Kearns KN, Eli I, Sharifi KA, Soldozy S, Carlson EW, et al. Microglial calcium waves during the hyperacute phase of ischemic stroke. Stroke 2021, 52: 274–283.
Brawek B, Schwendele B, Riester K, Kohsaka S, Lerdkrai C, Liang Y, et al. Impairment of in vivo calcium signaling in amyloid plaque-associated microglia. Acta Neuropathol 2014, 127: 495–505.
Nagel G, Szellas T, Huhn W, Kateriya S, Adeishvili N, Berthold P, et al. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci U S A 2003, 100: 13940–13945.
Eyo UB, Gu N, De S, Dong H, Richardson JR, Wu LJ. Modulation of microglial process convergence toward neuronal dendrites by extracellular calcium. J Neurosci 2015, 35: 2417–2422.
Eyo UB, Peng J, Murugan M, Mo M, Lalani A, Xie P, et al. Regulation of physical microglia-neuron interactions by fractalkine signaling after status epilepticus. eNeuro 2017, 3: ENEURO.0209-16.2016.
Fyfe I. Microglial optogenetics triggers chronic pain in mice. Nat Rev Neurol 2021, 17: 262.
Claes M, De Groef L, Moons L. The DREADDful hurdles and opportunities of the chronic chemogenetic toolbox. Cells 2022, 11: 1110.
Eyo UB, Murugan M, Wu LJ. Microglia-neuron communication in epilepsy. Glia 2017, 65: 5–18.
Qin C, Zhou LQ, Ma XT, Hu ZW, Yang S, Chen M, et al. Dual functions of microglia in ischemic stroke. Neurosci Bull 2019, 35: 921–933.
Xie M, Zhao S, Bosco DB, Nguyen A, Wu LJ. Microglial TREM2 in amyotrophic lateral sclerosis. Dev Neurobiol 2022, 82: 125–137.
Chen Y, Colonna M. Microglia in Alzheimer’s disease at single-cell level. Are there common patterns in humans and mice? J Exp Med 2021, 218: e20202717.
Bassett B, Subramaniyam S, Fan Y, Varney S, Pan H, Carneiro AMD, et al. Minocycline alleviates depression-like symptoms by rescuing decrease in neurogenesis in dorsal hippocampus via blocking microglia activation/phagocytosis. Brain Behav Immun 2021, 91: 519–530.
Cornell J, Salinas S, Huang HY, Zhou M. Microglia regulation of synaptic plasticity and learning and memory. Neural Regen Res 2022, 17: 705–716.
Chen T, Bosco DB, Ying Y, Tian DS, Wu LJ. The emerging role of microglia in neuromyelitis optica. Front Immunol 2021, 12: 616301.
Miyamoto K, Kume K, Ohsawa M. Role of microglia in mechanical allodynia in the anterior cingulate cortex. J Pharmacol Sci 2017, 134: 158–165.
Liu Y, Zhou LJ, Wang J, Li D, Ren WJ, Peng J, et al. TNF-α differentially regulates synaptic plasticity in the Hippocampus and spinal cord by microglia-dependent mechanisms after peripheral nerve injury. J Neurosci 2017, 37: 871–881.
Taylor AMW, Mehrabani S, Liu S, Taylor AJ, Cahill CM. Topography of microglial activation in sensory-and affect-related brain regions in chronic pain. J Neurosci Res 2017, 95: 1330–1335.
Acknowledgements
This review was supported by the National Institutes of Health (R01NS088627 and R01NS110825). We thank Dr. Anthony Umpierre (Mayo Clinic) for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Parusel, S., Yi, MH., Hunt, C.L. et al. Chemogenetic and Optogenetic Manipulations of Microglia in Chronic Pain. Neurosci. Bull. 39, 368–378 (2023). https://doi.org/10.1007/s12264-022-00937-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-022-00937-3