Abstract
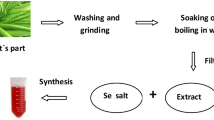
17.5 ± 2.41 mg/g and 15.2 ± 2.09 mg/g of total chlorophyll and chlorophyll a, respectively, were obtained from the marine microalga, Spirulina maxima under an optimal extraction condition of 70% ethanol at 65°C for 4 h associated with ultrasonic pretreatment at 40 kHz for 8 h. In comparison, 14.5 ± 1.36 mg/g and 7.1 ± 0.99 mg/g of total chlorophyll and chlorophyll a, respectively, were obtained from a conventional extraction process using 70% ethanol at 80°C for 12 h. The extract from the optimal conditions had the highest ratio of chlorophyll a to total chlorophyll, approximately 3:4, indicating that more intact chlorophyll a was obtained at low temperature. Moreover, the extract obtained using the optimal extraction condition showed substantial neuroprotective effects such as 92.78 ± 0.04% protection against glutamate-induced mouse hippocampal neuronal cells, compared to 81.64 ± 0.07% protection with the extract from the conventional extraction process. Compared to the control, the activities of two key enzymes related to glutathione synthesis, i.e., glutathione reductase and glutathione peroxidase, were also strongly increased, up to 90%, by the extracts from the optimal conditions. Interestingly, the addition of the same concentration of chlorophyll a as in the optimized extract had lower neuroprotective effects than did the extracts. This finding indicates that the extract likely exerted a synergistic effect, showing that the extract had better neuroprotective activity than the single component (chlorophyll a) alone. This work also confirmed the neuroprotective mechanism of the extract mainly due to its high antioxidant activity, allowing it to greatly decrease accumulation of ROS and Ca2+ within HT22 cells. The results of this work will have implications for expanding the use of a nonthermal ultrasonic process to extract heat-sensitive bioactive substances from natural resources.
Similar content being viewed by others
References
Citron, M. (2002) Alzheimer’s disease: treatments in discovery and development. Nat. Neurosci. 5: 1055–1057.
Bartus, R. T., R. L. Dean, D. Beer, and A. S. Lipa (1982) The cholinergic hypothesis of geriatric memory dysfunction. Scienc. 217: 408–417.
Dawson, G. R. and S. D. Iversen (1993) The effects of novel cholinesterase inhibitors and selective muscarinic receptor agonists in tests of reference and working memory. Behav. Brain Res. 57: 143–153.
Ushijima, M., N. Komoto, Y. Sugizono, I. Mizuno, M. Sumihiro, M. Ichikawa, M. Hayama, N. Kawahara, T. Nakane, O. Shirota, S. Sekita, and M. Kuroyanagi (2008) Triterpene glycosides from the roots of Codonopsis lanceolata. Chem. Pharm. Bull. 56: 308–314.
Li, J. P., Z. M. Liang, and Z. Yuan (2007) Triterpenoid saponins and anti–inflammatory activity of Codonopsis lanceolata. Pharmazi. 62: 463–466.
Wang, L., M. L. Xu, J. H. Hu, S. K. Rasmussen, and M. H. Wang (2011) Codonopsis lanceolata extract induces G0/G1 arrest and apoptosis in human colon tumor HT–29 cells–involvement of ROS generation and polyamine depletion. Food Chem. Toxicol. 49: 149–154.
Kay, R. A. (1991) Microalgae as food and supplement. Crit. Rev. Food Sci. Nutr. 30: 555–783.
Son, M. H., K. H. Park, A. R. Choi, G. Yoo, M. J. In, D. H. Kim, and H. J. Chae (2009) Investigation of biological activities of enzymatic hydrolysate of Spirulina. J. Korean Soc. Food Sci. Nutr. 38: 136–141.
Yang, H. N., E. H. Lee, and H. M. Kim (1997) Spirulina platensis inhibits anaphylactic reaction. Life Sci. 61: 1237–1244.
Bhat, S. R (2005) Chlorophyll: The wonder pigment. Sci. Rep. 42: 29–32.
Hosikian, A., S. Lim, R. Halim, and M. K. Danquah (2010) Chlorophyll extraction from microalgae: a review on the process engineering aspects. Int. J. Chem. Eng. 2010: 1–11.
Lee, Y. J., S. H. Kim, J. S. Kim, J. A. Han, H. J. Seo, H. J. Lim, and S. Y. Choi (2005) Studies on simultaneous determination of chlorophyll a and b, pheophorbide a, and ß–carotene in chlorella and Spirulina products. J. Food Hyg. Saf. 20: 141–146.
Gruskin, B. (1940) Chlorophyll—Its therapeutic place in acute and suppurative disease: preliminary report of clinical use and rationale. Am. J. Surg. 49: 49–55.
Mccarty, M. F. (2001) The chlorophyll metabolite phytanic acid is a natural rexinoid––potential for treatment and prevention of diabetes. Med. Hypothese. 56: 217–219.
Bechelli, J., M. Coppage, K. Rosell, and J. Liesveld (2011) Cytotoxicity of algae extracts on normal and malignant cells. Leuk. Res. Treatmen. 2011: 1–7.
Rehni, A. K., H. S. Pantlya, R. Shri, and M. Singh (2007) Effect of chlorophyll and aqueous extracts of Bacopa monniera and Valeriana wallichii on ischaemia and reperfusion–induced cerebral injury in mice. Indian J. Exp. Biol. 45: 764–769.
Hosikian, A., S. Lim, R. Halim, and M. K. Danquah (2010) Chlorophyll extraction from microalgae: A review on the process engineering aspects. Int. J. Chem. Eng. 2010: 1–11.
Chung, K. W., W. I. Kim, I. K. Hong, and K. A. Park (2000) Ultrasonic energy effects on squalene extraction from amaranth seed. Appl. Chem. 4: 149–152.
Oh, S. H., J. H. Ahn, D. H. Kang, and H. Y. Lee (2011) The effect of ultrasonificated extracts of Spirulina maxima on the Anticancer activity,” Marine Biotechnol. 13: 205–214.
Lee, D. H. and J. H. Hong (2015) Antioxidant activities of chlorella extracts and physicochemical characteristics of spraydried chlorella powders. Korean J. Food Preserv. 22: 591–597.
Petrovic, S. M., S. R. Savic, D. Z. Markovic, and Ž. B. Petronijevic (2014) In vitro studies of temperature and pH influence on chlorophyll degradation by horseradish peroxidase: Spectroscopic and HPLC studies. Hem. Ind. 68: 233–239.
Weon, J. B., B. Lee, B. R. Yun, J. Lee, H. Y. Lee, D. S. Park, H. C. Chung, J. Y. Chung, and C. J. Ma (2013) Memory enhancing effect of codonopsis lanceolata by high hydrostatic pressure process and fermentation. Korean J. Pharmacogn. 44: 41–46.
Lee, J., J. B. Weon, and C. J. Ma (2014) Neuroprotective Activity of Phytosterols Isolated from Artemisia apiacea. Korean J. Pharmacogn. 45: 214–219.
Lee, J. B., J. H. Ha, C. J. Song, S. D. Jung, H. B. Park, J. S. Kim, H. M. Sung, and M. J. Chang (2000) Effect of repeated stress on the antioxidant activities of brain. Korean Circ. J. 30: 67–72.
Lee, M. R., S. H. Moon, A. R. Choi, S. C. Lee, K. H. Ahn, and H. R. Park (2011) Neuroprotective effects of extracts from diospyros kaki L. peel. Korean J. Food Cookery Sci. 27: 67–73.
Satoh, T., Y. Enokido, H. Aoshima, Y. Uchiyama, and H. Hatanaka (1997) Changes in mitochondrial membrane potential during oxidative stress–induced apoptosis in PC12 cells. J. Neurosci. Res. 50: 413–420.
Hernadez, A. C., I. Nieves, M. Meckes, G. Chamorro, and B. L. Barron (2002) Antiviral activity of Spirulina maxima against Herpes simplex virus type 2. Antiviral Res. 56: 297–285.
Coyle, J. T. and P. Puttfarcken (1993) Oxidative stress, glutamate, and neurodegenerative disorders. Scienc. 262: 689–695.
Ha, J. S. and S. S. Park (2006) Glutamate–induced oxidative stress, but not cell death, is largely dependent upon extracellular calcium in mouse neuronal HT22 cells. Neurosci. Lett. 393: 165–169.
Ahmed, I., A. John, C. Vijayasarathy, M. A. Robin, and H. Raza (2002) Differential modulation of growth and glutathione metabolism in cultured rat astrocytes by 4–hydroxynonenal and green tea polyphenol, epigallocatechin–3–gallate. Neurotoxicolog. 23: 289–300.
Armstrong, J. S. and D. P. Jones (2002) Glutathione depletion enforces the mitochondrial permeability transition and causes cell death in Bcl–2 overexpressing HL60 cells. FASEB J. 16: 1263–1265.
Ansari, M. A., H. M. Abdul, G. Joshi, W. O. Opii, and D. A. Butterfield (2009) Protective effect of quercetin in primary neurons against Abeta (1–42): relevance to Alzheimer’s disease. J. Nutr. Biochem. 20: 269–75.
Miyamoto, M., T. H. Murphy, R. L. Schnaar, and J. T. Coyle (1989) Antioxidants protect against glutamate–induced cytotoxicity in a neuronal cell line. J. Pharmacol. Exp. Ther. 250: 1132–1140.
Albrecht, P. J., J. P. Dahl, O. K. Stoltzfus, R. Levenson, and S. W. Levison (2002) Ciliary neurotrophic factor activates spinal cord astrocytes, stimulating their production and release of fibroblast growth factor–2, to increase motor neuron survival. Exp. Neurol. 173: 46–62.
Bando, Y., T. Katayama, K. Kasai, M. Taniguchi, M. Tamatani, and M. Tohyama (2003) GRP94 (94 kDa glucose–regulated protein) suppresses ischemic neuronal cell death against ischemia/ reperfusion injury. Eur. J. Neurosci. 18: 829–840.
Acknowledgment
This work was supported by a research grant from the Marine Biotechnology Program by the Ministry of Oceans and Fisheries (PM59530).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, W.Y., Kang, D.H. & Lee, H.Y. Enhancement of Neuroprotective Effects of Spirulina maxima by a Low-temperature Extraction Process with Ultrasonic Pretreatment. Biotechnol Bioproc E 23, 415–423 (2018). https://doi.org/10.1007/s12257-018-0066-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-018-0066-2