Abstract
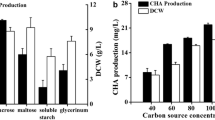
Fumigaclavine C (FC), which is produced by Aspergillus fumigatus, is a conidiation-associated ergot alkaloid with significant medical benefits. However, its application is restricted by low yields from submerged cultures. In this study, the technical feasibility of using molasses as a cost-effective ingredient for FC production in a two-stage culture of A. fumigatus was evaluated. The results indicated that molasses supplementation significantly enhanced FC accumulation by promoting conidiation and up-regulating hydroxymethylglutaryl-CoA reductase activity. Via the optimization of the two-stage process in the presence of molasses, FC production in shake flasks reached 226.9 mg/L, which was approximately three times that in the original medium (75.9 mg/L). The use of molasses as a cost-effective ingredient for FC fermentation was also successfully reproduced in a lab-scale bioreactor system in which the maximum FC production reached 215.0 mg/L. The FC production obtained in this study is the highest ever reported. This increased efficiency will enable large-scale production of FC and extend the application of molasses as a low-cost substrate for producing other conidiation-related secondary metabolites.
Similar content being viewed by others
References
Jakubczyk, D., J. Z. Cheng, and S. E. O’Connor (2014) Biosynthesis of the ergot alkaloids. Nat. Prod. Rep. 31: 1328–1338.
Wu, X. F., M. J. Fei, R. G. Shu, R. X. Tan, and Q. Xu (2005) Fumigaclavine C, an fungal metabolite, improves experimental colitis in mice via downregulating Th1 cytokine production and matrix metalloproteinase activity. Int. Immunopharmacol. 5: 1543–1553.
Zhao, Y., J.Y. Liu, J. Wang, L. Wang, H. Yin, R.X. Tan and Q. Xu (2004) Fumigaclavine C improves concanavalin A-induced liver injury in mice mainly via inhibiting TNF-alpha production and lymphocyte adhesion to extracellular matrices. J. Pharm. Pharmacol. 56: 775–782.
Du, R. H., E. G. Li, Y. Cao, Y. C. Song, and R. X. Tan (2011) Fumigaclavine C inhibits tumor necrosis factor á production via suppression of toll-like receptor 4 and nuclear factor êB activation in macrophages. Life Sci. 89: 235–240.
Li, Y. X., S. W. A. Himaya, P. Dewapriya, C. Zhang, and S. K. Kim (2013) Fumigaclavine C from a marine-derived fungus Aspergillus fumigatus induces apoptosis in MCF-7 breast cancer cells. Mar. Drug. 11: 5063–5086.
Du, R. H., S. Y. Qin, L. S. Shi, Z. Q. Zhou, X. Y. Zhu, J. Liu, R. X. Tan, and W. Cao (2014) Fumigaclavine C activates PPARγ pathway and attenuates atherogenesis in ApoE-deficient mice. Atherosclerosi. 234: 120–128.
Panaccione, D. G. and C. M. Coyle (2005) Abundant respirable ergot alkaloids from the common airborne fungus Aspergillus fumigatus. Appl. Environ. Microb. 71: 3106–3111.
Coyle, C. M., S. C. Kenaley, W. R. Rittenour, and D. G. Panaccione (2007) Association of ergot alkaloids with conidiation in Aspergillus fumigatus. Mycol. 99: 804–811.
Mulinti, P., N.A. Allen, C. M. Coyle, F. N. Gravelat, D. C. Sheppard, and D. G. Panaccione (2014) Accumulation of ergot alkaloids during conidiophore development in Aspergillus fumigatus. Curr. Microbiol. 68: 1–5.
Wu, Q., Y. Song, H. Xu, Y. Guo, J. Li, and R. X. Tan (2012) Medium optimization for enhanced co-production of two bioactive metabolites in the same fermentation by a statistical approach. J. Asian Nat. Prod. Res. 13: 37–41.
Zhu, Y. X., L. Y. Yao, R. H. Jiao, Y. H. Lu, and R. X. Tan (2014) Enhanced production of Fumigaclavine C in liquid culture of Aspergillus fumigatus under a two-stage process. Bioresour. Technol. 152: 162–168.
Yao, L.Y., Y. X. Zhu, R. H. Jiao, Y. H. Lu, and R. X. Tan (2014) Enhanced production of fumigaclavine C by ultrasound stimulation in a two-stage culture of Aspergillus fumigatus CY018. Bioresour. Technol. 159: 112–117.
Xu, K. and P. Xu (2014) Efficient production of l-lactic acid using co-feeding strategy based on cane molasses/glucose carbon sources. Bioresour. Technol. 153: 23–29.
Arshad, M., S. Ahmed, M. A. Zia, and M. I. Rajoka (2014) Kinetics and thermodynamics of ethanol production by Saccharomyces cerevisiae MLD10 using molasses. Appl. Biochem. Biotechnol. 172: 2455–2464.
Cazetta, M. L., M. A. P. C. Celligoi, J. B. Buzato, and I. S. Scarmino (2007) Fermentation of molasses by Zymomonas mobilis: Effects of temperature and sugar concentration on ethanol production. Bioresour. Technol. 98: 2824–2828.
Wee, Y. J., J. N. Kim, J. S. Yun, and H. W. Ryu (2004) Utilization of sugar molasses for economical L(+)-lactic acid production by batch fermentation of Enterococcus faecalis. Enz. Microb. Technol. 35: 568–573.
Wang, J. L., X. H. Wen, and D. Zhou (2000) Production of citric acid from molasses integrated with in-situ product separation by ion-exchange resin adsorption. Bioresour. Technol. 75: 231–234.
Lee, S. M., B. R. Oh, J. M. Park, A. Yu, S. Y. Heo, W. K. Hong, J. W. Seo, and C. H. Kim (2014) Optimized production of 2,3-butanediol by a lactate dehydrogenase-deficient mutant of Klebsiella pneumoniae. Biotechnol. Bioproc. Eng. 18: 1210–1215.
Park, K. M., M. W. Song, and J. H. Lee (2008) Production of carotenoids by ß-ionone-resistant mutant of Xanthophyllomyces dendrorhous using various carbon sources. Biotechnol. Bioproc. Eng. 13: 197–203.
López, J., J. M. Naranjo, J. Higuita, M. Cubitto, C. Cardona, and M. Villar (2012) Biosynthesis of PHB from a new isolated Bacillus megaterium strain: Outlook on future developments with endospore forming bacteria. Biotechnol. Bioproc. Eng. 17: 250–258.
Lee, Y., M. J. Lee, Y. E. Choi, G. T. Chun, and Y. S. Jeong (2015) Optimization of cultivation medium and fermentation parameters for lincomycin production by Streptomyces lincolnensis. Biotechnol. Bioproc. Eng. 19: 1014–1021.
Chang, P. K. and S. S. T. Hua (2007) Molasses supplementation promotes conidiation but suppresses aflatoxin production by small sclerotial Aspergillus flavus. Lett. Appl. Microbiol. 44: 131–137.
Blankenship, J. D., M. J. Spiering, H. H. Wilkinson, F. F. Fannin, L. P. Bush, and C. L. Schardl (2001) Production of loline alkaloids by the grass endophyte, Neotyphodium uncinatum, in defined media. Phytochem. 58: 395–401.
Boichenko, L.V., N. F. Zelenkova, M. U. Arinbasarov, and R. T. A. Reshetilova (2003) Optimization of conditions for storage and cultivation of the fungus Claviceps sp., a producer of the ergot alkaloid agroclavine. Appl. Biochem. Microbiol. 39: 335–340.
Shasaltaneh, M. D., J. Fooladi, and S. Z. Moosavi-Nejad (2010) L-tryptophan production by Escherichia coli in the presence of Iranian cane molasses. J. Paramed. Sci. 1: 19–25.
Wallwey, C. and S. M. Li (2011) Ergot alkaloids: structure diversity, biosynthetic gene clusters and functional proof of biosynthetic genes. Nat. Prod. Rep. 28: 496–510.
Liu, J., J. Huang, Y. Jiang, and F. Chen (2012) Molasses-based growth and production of oil and astaxanthin by Chlorella zofingiensis. Bioresour. Technol. 107: 393–398.
Hatano, K. I., S. Kikuchi, Y. Nakamura, H. Sakamoto, M. Takigami, and Y. Kojima (2009) Novel strategy using an adsorbentcolumn chromatography for effective ethanol production from sugarcane or sugar beet molasses. Bioresour. Technol. 100: 4697–4703.
Xia, J., Z. Xu, H. Xu, J. Liang, S. Li, and X. Feng (2014) Economical production of poly(ε-l-lysine) and poly(l-diaminopropionic acid) using cane molasses and hydrolysate of streptomyces cells by Streptomyces albulus PD-1. Bioresour. Technol. 164: 241–247.
Liu, Y. P., P. Zheng, Z. H. Sun, Y. Ni, J. J. Dong, and L. L. Zhu (2008) Economical succinic acid production from cane molasses by Actinobacillus succinogenes. Bioresour. Technol. 99: 1736–1742.
Wang, G. and J. D. Keasling (2002) Amplification of HMG-CoA reductase production enhances carotenoid accumulation in Neurospora crassa. Metab. Eng. 201: 193–201.
Adams, T. H., J. K. Wieser, and J. H. Yu (1998) Asexual sporulation in Aspergillus nidulans. Microbiol. Mol. Biol. R. 62: 35–54.
Morton, A. G. (1961) The induction of sporulation in mould fungi. Proc. R. Soc. Lond. 153: 548–569.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhu, YX., Hu, WW., Yao, LY. et al. Improvement of fumigaclavine C production in a two-stage culture of Aspergillus fumigatus with molasses as a cost-effective ingredient. Biotechnol Bioproc E 20, 1106–1113 (2015). https://doi.org/10.1007/s12257-015-0193-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-015-0193-y