Abstract
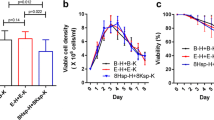
Cell cultures containing 0 ∼ 5 mM sodium butyrate (NaBu) and grown at 30 and 37°C were conducted to investigate the combined effect of NaBu and low temperature on the quantity and quality of an antibody production in CHO cells. Although NaBu addition decreased cell viability by apoptosis in a dose-dependent manner at both 30 and 37°C, the onset of significant apoptosis induced by NaBu was delayed by lowering culture temperature. The highest specific antibody productivity (q p) of 23.26 pg/cell/day was obtained in the culture containing 2 mM NaBu at 30°C; however, the highest antibody concentration of 167.84 mg/L was achieved in the culture containing 1 mM NaBu at 30°C, as the detrimental effect of further NaBu addition on cell growth compromised its beneficial effect on q p. Moreover, protein quality with respect to the total sialic acid content and Nglycolylneuraminic acid (Neu5Gc) level was evaluated. There were no apparent changes regarding the total sialic acid content of the antibody, but manipulation of cultures with NaBu treatment or (and) low culture temperature did decrease Neu5Gc levels by 5 ∼ 10%. Biological activity of the antibody was also assessed, and no obvious changes were observed. Collectively, the simultaneous application of NaBu and low culture temperature was an effective way to extend culture period and enhance final antibody concentration, without compromising the sialic acid content or biological activity.
Similar content being viewed by others
References
Wurm, F. M. (2004) Production of recombinant protein therapeutics in cultivated mammalian cells. Nat. Biotechnol. 22: 1393–1398.
Furukawa, K. and K. Ohsuye (1998) Effect of culture temperature on a recombinant CHO cell line producing a C-terminal α-amidating enzyme. Cytotechnol. 26: 153–164.
Fogolín, M. B., R. Wagner, M. Etcheverrigaray, and R. Kratje (2004) Impact of temperature reduction and expression of yeast pyruvate carboxylase on hGM-CSF-producing CHO cells. J. Biotechnol. 109: 179–191.
Yoon, S. K., J. Y. Song, and G. M. Lee (2003) Effect of low culture temperature on specific productivity, transcription level, and heterogeneity of erythropoietin in Chinese Hamster Ovary cells. Biotechnol. Bioeng. 82: 289–298.
Ahn, W. S., J. J. Jeon, Y. R. Jeong, S. J. Lee, and S. K. Yoon (2008) Effect of culture temperature on erythropoietin production and glycosylation in a perfusion culture of recombinant CHO cells. Biotechnol. Bioeng. 101: 1234–1244.
Virginie, M. B., S. Hisiger, C. Durand, M. Perrier, and M. Jolicoeur (2009) Na-butyrate sustains energetic states of metabolism and t-PA productivity of CHO cells. J. Biosci. Bioeng. 108: 160–167.
Mimura, Y., J. Lund, S. Church, S. Dong, J. Li, M. Goodall, and R. Jefferis (2001) Butyrate increases production of human chimeric IgG in CHO-K1 cells whilst maintaining function and glycoform profile. J. Immunol. Methods. 247: 205–216.
Yoon, S. K., J. K. Hong, and G. M. Lee (2004) Effect of simultaneous application of stressful culture conditions on specific productivity and heterogeneity of erythropoietin in Chinese Hamster Ovary cells. Biotechnol. Prog. 20: 1293–1296.
Sung, Y. H., Y. J. Song, S. W. Lim, J. Y. Chung, and G. M. Lee (2004) Effect of sodium butyrate on the production, heterogeneity and biological activity of human thrombopoietin by recombinant Chinese hamster ovary cells. J. Biotechnol. 112: 323–335.
Kim, N. S. and G. M. Lee (2000) Overexpression of bcl-2 inhibits sodium butyrate-induced apoptosis in Chinese hamster ovary cells resulting in enhanced humanized antibody production. Biotechnol. Bioeng. 71: 184–193.
Kim, N. S. and G. M. Lee (2001) Inhibition of sodium butyrateinduced apoptosis in recombinant Chinese Hamster Ovary cells by constitutively expressing antisense RNA of caspase-3. Biotechnol. Bioeng. 78: 217–228.
Sung, Y. H., S. J. Hwang, and G. M. Lee (2005) Influence of down-regulation of caspase-3 by siRNAs on sodium-butyrateinduced apoptotic cell death of Chinese hamster ovary cells producing thrombopoietin. Metab. Eng. 7: 457–466.
Sung, Y. H., J. S. Lee, S. H. Park, J. Koo, and G. M. Lee (2007) Influence of co-down-regulation of caspase-3 and caspase-7 by siRNAs on sodium butyrate-induced apoptotic cell death of Chinese hamster ovary cells producing thrombopoietin. Metab. Eng. 9: 452–464.
Oh, H. K., M. K. So, J. Yang, H. C. Yoon, J. S. Ahn, J. M. Lee, J. T. Kim, J. U. Yoo, and T. H. Byun (2005) Effects of N-Acetylcysteine on butyrate-treated Chinese Hamster Ovary cells to improve the production of recombinant human interferon-β-1a. Biotechnol. Prog. 21: 1154–1164.
Chuppa, S., Y. S. Tsai, S. Yoon, S. Shackleford, C. Rozales, R. Bhat, G. Tsay, C. Matanguihan, K. Konstantinov, and D. Naveh (1997) Fermentor temperature as a tool for control of high-density perfusion cultures of mammalian cells. Biotechnol. Bioeng. 55: 328–338.
Kaufmann, H., X. Mazur, M. Fussenegger, and J. E. Bailey (1999) Influence of low temperature on productivity, proteome and protein phosphorylation of CHO cells. Biotechnol. Bioeng. 63: 573–582.
Kim, N. S., K. H. Chang, B. S. Chung, S. H. Kim, J. H. Kim, and G. M. Lee (2003) Characterization of humanized antibody produced by apoptosis-resistant CHO cells under sodium butyrateinduced condition. J. Microbiol. Biotechnol. 13: 926–936.
Sethuraman, N. and T. A. Stadheim (2006) Challenges in therapeutic glycoprotein production. Curr. Opin. Biotechnol. 17: 341–346.
Lamari, F. N. and N. K. Karamnnos (2002) Separation methods for sialic acids and critical evaluation of their biologic relevance. J. Chrom. B. 781: 3–19.
Irie, A., S. Koyama, Y. Kozutsumi, T. Kawasaki, and A. Suzuki (1998) The molecular basis for the absence of N-glycolylneuraminic acid in humans. J. Biol. Chem. 273: 15866–15871.
Varki, A. (2001) N-glycolylneuraminic acid deficiency in humans. Biochimie. 83: 615–622.
Lipscomb, M. L., L. A. Palomares, V. Hernandez, O. T. Ramirez, and D. S. Kompala (2005) Effect of production method and gene amplification on the glycosylation pattern of a secreted reporter protein in CHO cells. Biotechnol. Prog. 21: 40–49.
Zanghi, J. A., A. E. Schmelzer, T. P. Mendoza, R. H. Knop, and W. M. Miller (1999) Bicarbonate concentration and osmolality are key determinants in the inhibition of CHO cell polysialylation under elevated pCO2 or pH. Biotechnol. Bioeng. 65: 182–191.
Renard, J. M., R. Spagnoli, C. Mazier, M. F. Salles, and E. Mandine (1988) Evidence that monoclonal antibody production kinetics is related to the integral of viable cells in batch systems. Biotechnol. Lett. 10: 91–96.
Gawlitzek, M., T. Ryll, J. Lofgren, and M. B. Sliwkowski (2000) Ammonium alters N-glycan structures of recombinant TNFRIgG: Degradative versus biosynthetic mechanisms. Biotechnol. Bioeng. 68: 637–646.
Anumula, K. R. (1995) Rapid quantitative determination of sialic acids in glycoproteins by high-performance liquid chromatography with a sensitive fluorescence detection. Anal. Biochem. 230: 24–30.
Wagner, A., A. Marc, J. M. Engasser, and A. Einsele (1992) The use of lactate dehydrogenase (LDH) release kinetics for the evaluation of death and growth of mammalian cells in perfusion reactors. Biotechnol. Bioeng. 39: 320–326.
Sunley, K. and M. Butler (2010) Strategies for the enhancement of recombinant protein production from mammalian cells by growth arrest. Biotechnol. Adv. 28: 385–394.
Hendrick, V., P. Winnepenninckx, C. Abdelkafi, O. Vandeputte, M. Cherlet, T. Marique, G. Renemann, A. Loa, G. Kretzmer, and J. Werenne (2001) Increased productivity of recombinant tissular plasminogen activator (t-PA) by butyrate and shift of temperature: A cell cycle phases analysis. Cytotechnol. 36: 71–83.
Jiang, Z. and S. T. Sharfstein (2008) Sodium butyrate stimulates monoclonal antibody over-expression in CHO cells improving gene accessibility. Biotechnol. Bioeng. 100: 189–194.
Yee, J. C., M. L. Gatti, R. J. Philp, M. Yap, and W. S. Hu (2008) Genomic and proteomic exploration of CHO and Hybridoma cells under sodium butyrate treatment. Biotechnol. Bioeng. 99: 1186–1204.
Moore, A., J. Mercer, G. Dutina, C. J. Donahue, K. D. Bauer, J. P. Mather, T. Etcheverry, and T. Ryll (1997) Effects of temperature shift on cell cycle, apoptosis and nucleotide pools in CHO cell batch cultures. Cytotechnol. 23:47–54.
Takeuchi, M., N. Inoue, T. W. Strickland, M. Kubota, M. Wada, R. Shimizu, S. Hoshi, H. Kozutsumi, S. Takeuchi, and A. Kobata (1989) Relationship between sugar chain structure and biological activity of recombinant human erythropoietin produced in Chinese hamster ovary cells. Proc. Natl. Acad. Sci. USA. 86: 7819–7822.
Yamaguchi. K., K. Akai, G. Kawanishi, M. Ueda, S. Masuda, and R. Sasaki (1991) Effect of site-directed removal of N-glycosylation sites in human erythropoietin on its production and biological properties. J. Biol. Chem. 266: 20434–20439.
Malykh, Y. N., L. Shaw, and R. Schauer (1998) The role of CMPN-acetylneuraminic acid hydroxylase in determining the level of N-glycolylneuraminic acid in porcine tissues. Glycoconj. J. 15: 885–893.
Tangvoranuntakul, P., P. Gagneux, S. Diaz, M. Bardor, A. Varki, and E. Muchmore (2003) Human uptake and incorporation of an immunogenic nonhuman dietary sialic acid. Proc. Natl. Acad. Sci. USA. 100: 12045–12050.
Borys, M. C., N. G. Dalal, N. R. Abu-Absi, S. F. Khattak, Y. Jing, Z. H. Xing, and Z. J. Li (2010) Effects of culture conditions on N-Glycolylneuraminic acid (Neu5Gc) content of a recombinant fusion protein produced in CHO cells. Biotechnol. Bioeng. 105: 1048–1057.
Shaw, L. and R. Schauer (1988) The biosynthesis of N-glycoloylneuraminic acid occurs by hydroxylation of the CMP-glycoside of N-acetylneuraminic acid. Biol. Chem. Hoppe Seyler 369: 477–486.
Shaw, L. and R. Schauer (1989) Detection of CMP-N-acetylneuraminic acid hydroxylase activity in fractionated mouse liver. Biochem. J. 263: 355–363.
Grampp, G. E., T. K. Blumen, K. Kelly, P. Derby, L. A. Sleeman, and D. Hettwer (1994) Environmental control of sialic acid composition in glycoproteins secreted by mammalian cells. Presented at the Cell Culture Engineering IV meeting. San Diego, CA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, F., Kou, T., Fan, L. et al. The combined effect of sodium butyrate and low culture temperature on the production, sialylation, and biological activity of an antibody produced in CHO cells. Biotechnol Bioproc E 16, 1157–1165 (2011). https://doi.org/10.1007/s12257-011-0069-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-011-0069-8