Abstract
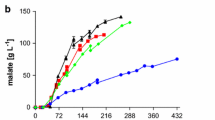
In an effort to improve TNFR-Fc production in GS-CHO cells, batch cultures were performed to investigate the effects of low culture temperature on TNFR-Fc production. It was observed that low culture temperatures resulted in cell cycle arrest in the G1 phase, led to suppressed cell growth, and prolonged the culture period. Although the highest TNFR-Fc concentration was achieved with a culture temperature of 32°C at 247.4 mg/L, the maximum q TNFR-Fc of 15.7 pg/cells/day was achieved at 30°C. Because the inhibition effect on cell growth at 30°C compromised its beneficial effects specifically to TNFR-Fc productivity, TNFR-Fc concentration at this temperature was not significantly increased. Furthermore, the increase in productivity of specific TNFR-Fc at low culture temperatures was also found to be due to an increase in the transcriptional level of the TNFR-Fc gene, determined by RT-PCR analysis. In addition, low culture temperatures had no significant effect on the degree of sialylation of TNFR-Fc. Taken together; a biphasic cultivation process developed in a fed-batch mode with a low temperature-production phase enhanced TNFR-Fc production by GS-CHO cells and therefore offers major potential for bioprocess optimization.
Similar content being viewed by others
References
Mohler, K. M., D. S. Torrance, C. A. Smith, R. G. Goodwin, K. E. Stremler, V. P. Fung, H. Madani, and M. B. Widmer (1993) Soluble tumor necrosis factor (TNF) receptors are effective therapeutic agents in lethal endotoxemia and function simultaneously as both TNF carriers and TNF antagonists. J. Immunol. 151: 1548–1561.
Ducharme, E. and J. M. Weinberg (2008) Etanercept. Expert. Opin. Biol. Ther. 8: 491–502.
Reuveny, S., D. Velez, J. D. Macmillan, and L. Miller (1986) Factors affecting cell growth and monoclonal antibody production in stirred reactors. J. Immunol. Methods. 86: 53–59.
Borys, M. C., D. I. H. Linzer, and E. T. Papoutsakis (1993) Culture pH affects expression rates and glycosylation of recombinant mouse placental lactogen proteins by Chinese hamster ovary (CHO) cells. Bio/Technol. 11: 720–724.
Masuda, S., S. K. Moon, T. Kambe, M. Nagao, and R. Sasaki (1999) A new biological strategy for high productivity of recombinant proteins in animal cells by the use of hypoxia-response enhancer. Biotechnol. Bioeng. 67: 157–164.
Furukawa, K. and K. Ohsuye (1998) Effect of culture temperature on a recombinant CHO cell line producing a C-terminal aamidating enzyme. Cytotechnol. 26: 153–164.
Fogolín, M. B., R. Wagner, M. Etcheverrigaray, and R. Kratje (2004) Impact of temperature reduction and expression of yeast pyruvate carboxylase on hGM-CSF-producing CHO cells. J. Biotechnol. 109: 179–191.
Chen, T., Y. Zhou, and W. S. Tan (2009) Effects of low temperature and lactate on osteogenic differentiation of human amniotic mesenchymal stem cells. Biotechnol. Bioprocess. Eng. 14: 708–715.
Yoon, S. K., S. H. Kim, and G. M. Lee (2003) Effect of low culture temperature on specific productivity and transcription level of anti-4-1BB antibody in recombinant Chinese hamster ovary cells. Biotechnol. Prog. 19: 1383–1386.
Ahn, W. S., J. J. Jeon, Y. R. Jeong, S. J. Lee, and S. K. Yoon (2008) Effect of culture temperature on erythropoietin production and glycosylation in a perfusion culture of recombinant CHO cells. Biotechnol. Bioeng. 101: 1234–1244.
Fussenegger, M., X. Mazur, and J. E. Bailey (1997) A novel cytostatic process enhances the productivity of Chinese hamster ovary cells. Biotechnol. Bioeng. 55: 927–939.
Renard, J. M., R. Spagnoli, C. Mazier, M. F. Salles, and E. Mandine (1988) Evidence that monoclonal antibody production kinetics is related to the integral of viable cells in batch systems. Biotechnol. Lett. 10: 91–96.
Gawlitzek, M., T. Ryll, J. Lofgren, and M. B. Sliwkowski (2000) Ammonium alters N-glycan structures of recombinant TNFR-IgG: Degradative versus biosynthetic mechanisms. Biotechnol. Bioeng. 68: 637–646.
Müthing, J., S. E. Kemminer, H. S. Conradt, D. Šagi, M. Nimtz, U. Kärst, and J. Peter-Kataliniæ (2003) Effects of buffering conditions and culture pH on production rates and glycosylation of clinical phase I anti-melanoma mouse IgG3 monoclonal antibody R24. Biotechnol. Bioeng. 83: 321–334.
Chuppa, S., Y. S. Tsai, S. Yoon, S. Shackleford, C. Rozales, R. Bhat, G. Tsay, C. Matanguihan, K. Konstantinov, and D. Naveh (1997) Fermentor temperature as a tool for control of high-density perfusion cultures of mammalian cells. Biotechnol. Bioeng. 55: 328–338.
Kaufmann, H., X. Mazur, M. Fussenegger, and J. E. Bailey (1999) Influence of low temperature on productivity, proteome and protein phosphorylation of CHO cells. Biotechnol. Bioeng. 63: 573–582.
Roessler, B., H. Luebben, and G. Kretzmer (1996) Temperature: A simple parameter for process optimization in fed-batch cultures of recombinant Chinese hamster ovary cells. Enz. Microb. Technol. 18: 423–427.
Jenkins, N. and A. Hovey (1993) Temperature control of growth and productivity in mutant Chinese hamster ovary cells synthesizing a recombinant protein. Biotechnol. Bioeng. 42: 1029–1036.
Kubbies, M. and H. Stockinger (1990) Cell cycle-dependent DHFR and t-PA production in cotransfected, MTX-amplified CHO cells revealed by dual-laser flow cytometry. Exp. Cell Res. 188: 267–271.
Mazur, X., M. Fussenegger, W. A. Renner, and J. E. Bailey (1998) Higher productivity of growth-arrested Chinese hamster ovary cells expressing the cyclin-dependent kinase inhibitor p27. Biotechnol Prog. 14: 705–713.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kou, TC., Fan, L., Zhou, Y. et al. Increasing the productivity of TNFR-Fc in GS-CHO cells at reduced culture temperatures. Biotechnol Bioproc E 16, 136–143 (2011). https://doi.org/10.1007/s12257-010-0157-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-010-0157-1