Abstract
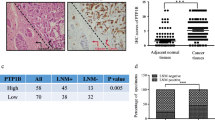
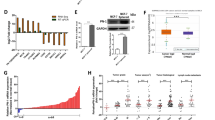
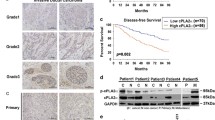
Invasive breast cancer is the most common type of malignancy in women worldwide. However, the mechanism responsible for breast cancer metastasis is still unclear and needs further illustration. It has been proven that matrix metallopeptidase 9 (MMP-9) promotes metastasis of the cancer cells. However, the interaction between mPRα and MMP-9 has not been studied. Therefore, in the present research, the effect of MMP-9 on the malignant progression of invasive breast cancer promoted by membrane progesterone receptorα (mPRα) was investigated. The results showed that the protein expression of mPRα, p-Akt and MMP-9 increased in the cancerous tissues compared to that of the noncancerous breast tissue. Furthermore, a positive correlation was found between mPRα and C-erbB-2, as well as the number of involved local lymph nodes. On the other hand, a negative correlation was observed between mPRα and estrogen receptors (ER) along with progesterone receptors (PR). Similarly, a positive association was found between MMP-9 and the number of involved local lymph nodes. Besides, the high expression of MMP-9 also had a positive correlation with the tumor size. However, the high level of MMP-9 had a negative correlation with ER and PR. In addition, there was a positive correlation between mPRα and p-Akt together with MMP-9. The results confirm that mPRα was a major marker of harmful prognosis and it promoted the expression of MMP-9 during invasion to the local lymph nodes through the pathway of PI3K/Akt. The present study provided a novel therapeutic strategy to inhibit breast cancer growth by preventing mPRα signaling pathway.


Similar content being viewed by others
References
Chambers AF, Naumov GN, Varghese HJ, Nadkarni KV, MacDonald IC, Groom AC (2001) Critical steps in hematogenous metastasis: an overview. Surg Oncol Clin N Am 10(2):243–255 vii
Basu A, Rowan BG (2005) Genes related to estrogen action in reproduction and breast cancer. Front Biosci : J Virtual Libr 10:2346–2372
Carnevale RP, Proietti CJ, Salatino M, Urtreger A, Peluffo G, Edwards DP, et al. (2007) Progestin effects on breast cancer cell proliferation, proteases activation, and in vivo development of metastatic phenotype all depend on progesterone receptor capacity to activate cytoplasmic signaling pathways. Mol Endocrinol 21(6):1335–1358. doi:10.1210/me.2006-0304
Karteris E, Zervou S, Pang Y, Dong J, Hillhouse EW, Randeva HS, et al. (2006) Progesterone signaling in human myometrium through two novel membrane G protein-coupled receptors: potential role in functional progesterone withdrawal at term. Mol Endocrinol 20(7):1519–1534. doi:10.1210/me.2005-0243
Dressing GE, Alyea R, Pang Y, Thomas P (2012) Membrane progesterone receptors (mPRs) mediate progestin induced antimorbidity in breast cancer cells and are expressed in human breast tumors. Horm Cancer 3(3):101–112. doi:10.1007/s12672-012-0106-x
Alizadeh AA, Ross DT, Perou CM, van de Rijn M (2001) Towards a novel classification of human malignancies based on gene expression patterns. J Pathol 195(1):41–52. doi:10.1002/path.889
Loya A, Guray M, Hennessy BT, Middleton LP, Buchholz TA, Valero V, et al. (2009) Prognostic significance of occult axillary lymph node metastases after chemotherapy-induced pathologic complete response of cytologically proven axillary lymph node metastases from breast cancer. Cancer 115(8):1605–1612. doi:10.1002/cncr.24173
Illemann M, Bird N, Majeed A, Sehested M, Laerum OD, Lund LR, et al. (2006) MMP-9 is differentially expressed in primary human colorectal adenocarcinomas and their metastases. Mol Cancer Res : MCR 4(5):293–302. doi:10.1158/1541-7786.MCR-06-0003
Gompel A (2012) Micronized progesterone and its impact on the endometrium and breast vs. progestogens. Climacteric : J Int Menopause Soc 15(Suppl 1):18–25. doi:10.3109/13697137.2012.669584
L’Hermite M (2013) HRT optimization, using transdermal estradiol plus micronized progesterone, a safer HRT. Climacteric : J Int Menopause Soc. 16(Suppl 1):44–53. doi:10.3109/13697137.2013.808563
Kuhl H, Schneider HP (2013) Progesterone–promoter or inhibitor of breast cancer. Climacteric : J Int Menopause Soc 16(Suppl 1):54–68. doi:10.3109/13697137.2013.768806
Zhu Y, Rice CD, Pang Y, Pace M, Thomas P (2003) Cloning, expression, and characterization of a membrane progestin receptor and evidence it is an intermediary in meiotic maturation of fish oocytes. Proc Natl Acad Sci U S A 100(5):2231–2236. doi:10.1073/pnas.0336132100
Zhu Y, Bond J, Thomas P (2003) Identification, classification, and partial characterization of genes in humans and other vertebrates homologous to a fish membrane progestin receptor. Proc Natl Acad Sci U S A 100(5):2237–2242. doi:10.1073/pnas.0436133100
Thomas P, Pang Y, Dong J, Groenen P, Kelder J, de Vlieg J, et al. (2007) Steroid and G protein binding characteristics of the seatrout and human progestin membrane receptor alpha subtypes and their evolutionary origins. Endocrinology 148(2):705–718. doi:10.1210/en.2006-0974
Lyons TJ, Villa NY, Regalla LM, Kupchak BR, Vagstad A, Eide DJ (2004) Metalloregulation of yeast membrane steroid receptor homologs. Proc Natl Acad Sci U S A 101(15):5506–5511. doi:10.1073/pnas.0306324101
van Beijnum JR, Petersen K, Griffioen AW (2009) Tumor endothelium is characterized by a matrix remodeling signature. Front Biosci. 1:216–225
Youssef NS, Hakim SA (2014) Association of fascin and matrix metalloproteinase-9 expression with poor prognostic parameters in breast carcinoma of egyptian women. Diagn Pathol. 9:136. doi:10.1186/1746-1596-9-136
Xue B, Huang W, Yuan X, Xu B, Lou Y, Zhou Q, et al. (2015) YSY01A, a novel proteasome inhibitor, induces cell cycle arrest on G2 phase in MCF-7 cells via ERalpha and PI3K/Akt pathways. J Cancer 6(4):319–326. doi:10.7150/jca.10733
Nagaraj G, Ma C (2015) Revisiting the estrogen receptor pathway and its role in endocrine therapy for postmenopausal women with estrogen receptor-positive metastatic breast cancer. Breast Cancer Res Treat. doi:10.1007/s10549-015-3316-4
Paez J, Sellers WR (2003) PI3K/PTEN/AKT pathway. A critical mediator of oncogenic signaling. Cancer Treat Res 115:145–167
Pu P, Kang C, Li J, Jiang H (2004) Antisense and dominant-negative AKT2 cDNA inhibits glioma cell invasion. Tumour Biol : J Int Soc Oncodevelopmental Biol Med 25(4):172–178. doi:10.1159/000081099
Authors’ Contributions
Zhishuang Li and ChunyanZhang carried out the tissue microarray. Limin Sun and Yan Wang carried out the immunohistochemistry. Xiaojuan Wu, Peng Su and Xiaowang participated in the design of the study and performed the statistical analysis. Rong Ma and Peng Gao conceived of the study, and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The procedures in this study were in accordance with the ethical standards of the responsible committee of the Shandong University on human experimentation.
Conflict of Interest
The experiments abide by the current laws of China and there were no conflict of interest among the authors.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81402181).
Rights and permissions
About this article
Cite this article
Wu, X., Sun, L., Wang, X. et al. Breast Cancer Invasion and Metastasis by mPRα Through the PI3K/Akt Signaling Pathway. Pathol. Oncol. Res. 22, 471–476 (2016). https://doi.org/10.1007/s12253-015-0023-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-015-0023-8