Abstract
The use of sugarcane straw is a promising option to enhance bioenergy production. But the implications of straw removal for soil greenhouse gas (GHG) emission are not yet fully understood due to scarcity of studies under Brazilian conditions. Four field experiments were designed to assess soil N2O and CH4 emissions derived from nitrogen (N) fertilization under scenarios of sugarcane straw removal in São Paulo state, Brazil. Our focus was also to derive the direct N2O emission factor (EF) that represents the regional conditions of sugarcane production, taking into account the data obtained from this study and those from the literature. In each field study, four straw removal rates (no removal (NR); low removal (LR)—removal of 5 Mg ha−1; high removal (HR)—removal of 10 Mg ha−1; and total removal (TR)—removal of 15 Mg ha−1) were arranged in a randomized block design. This study shows a clear evidence that CH4 fluxes are very low for all assessed sites regardless of straw removal rates, indicating a predominance of CH4 consumption by the soil. Cumulative N2O emissions ranged from 0.20 to 4.09 kg ha−1 year−1 and were significantly affected by straw removal in two sites, indicating that straw removal reduces N2O emissions. The average direct N2O EFs obtained from this study and from the literature were 0.28, 0.44, 0.70, and 0.56% for TR, HR, LR, and NR treatments, respectively, which are consistently lower than the EF of 1% suggested by the IPCC. Based on our regional-specific EF (Tier 2), the direct N2O emissions derived from N fertilization under scenarios of straw removal showed a reduction of at least 50% in relation to IPCC approach. Our findings are a step forward in providing regional-specific data to reduce the high level of uncertainty concerning N2O emission assessments of sugarcane ethanol in Brazil, but further studies are needed to evaluate how straw removal for bioenergy production and the associated changes in soil organic carbon stocks affect the GHG balance of sugarcane.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Economic and population growth have driven the use of land and other natural resources to produce food, fiber, and energy [1] and, consequently, additional emission of greenhouse gases (GHG) requiring worldwide efforts to develop sustainable alternatives to energy sources [2]. Sugarcane ethanol is one of the most effective options to mitigate GHG emission when compared with other biofuel feedstocks [3].
Brazil is the largest sugarcane producer in the world, producing 28 billion liters of ethanol in a cultivated area of 8.8 million hectares in 2017/2018, and the state of São Paulo is responsible for 57% of the total cultivated area [4]. In the last few years, the harvest system of sugarcane has undergone profound changes associated with the prohibition of burning prior to harvest and the consequent adoption of the so-called green mechanized harvest without burning [5], in which a large amount of sugarcane straw is left in the field (10 to 20 Mg ha−1 of dry straw) [6]. Currently, around 96% of the sugarcane fields in São Paulo state are mechanically harvested without burning [4], and therefore, new possibilities of straw use arise with the growing demand for renewable energy sources. In Brazil, several companies are using sugarcane straw as a raw material for bioelectricity production and two mills are producing cellulosic (2G) ethanol in industrial scale [7].
However, the maintenance of straw on the soil surface provides numerous agronomic and environmental benefits, such as the maintenance of soil moisture, nutrient recycling, enhanced microbial community, lower water loss, protection against soil erosion, and increase in soil carbon stock, among others [3, 8, 9]. Conversely, the maintenance of large amount of straw in the field has drawbacks, such as increases in GHG emissions [7]. With a global warming potential (GWP) of 265 relative to CO2 [10], nitrous oxide (N2O) is among the main GHG emitted from soils under sugarcane production. Several studies have indicated that N2O emission in sugarcane fields is primarily affected by the application of synthetic N fertilizers [11,12,13]. Other studies have also shown that the application of N fertilizer associated with the presence of organic residues increased N2O emissions [11, 12], but the magnitude in which these emissions vary with different amounts of straw on soil surface remains poorly understood. The current IPCC [14] methodology establishes a default direct N2O emission factor (EF) of 1% of the N applied as fertilizers or crop residues, but this value may not represent regional-specific conditions of tropical soils in Brazil.
Nitrogen fertilization is a crucial practice to maximize sugarcane yields, being particularly important for highly weathered soils of tropical regions in Brazil [15]. But once N is applied to the soil, several processes occur before N absorption by sugarcane plants, including volatilization, lixiviation, and/or N2O emission [16]. The latter is formed in the soil mostly by the microbial processes of nitrification and denitrification [17, 18], which are highly influenced by moisture, temperature, and soil physical and chemical conditions. Methane (CH4) is another important GHG (GWP of 28 times higher than CO2) in agricultural environments [10], but so far little or no effect of straw maintenance on CH4 emissions has been observed in Brazilian sugarcane fields [11, 19], despite the higher soil moisture and organic C input caused by the straw mulching.
This study was based on the hypothesis that the maintenance of large amounts of straw on soil surface results in higher N2O and CH4 emissions, and the removal of this crop residue is a feasible strategy to mitigate such emissions. To test this hypothesis, four experimental fields were designed to quantify N2O and CH4 emissions under different straw removal strategies in São Paulo state, Brazil. The specific objective was to determine the direct N2O EFs from N fertilizer application in sugarcane areas under different amounts of straw. Our focus was also on deriving the direct N2O EFs that represent the regional-specific conditions of sugarcane production in São Paulo state, taking into account the data generated by this study and those obtained from published literature. Finally, this study aimed to estimate the direct N2O emissions (in kg CO2 eq ha−1) resulted from application of N fertilizer under different scenarios of straw removal by using the default N2O EF proposed by the IPCC (Tier 1), and the regional-specific N2O EFs (Tier 2) resulted from the entire dataset obtained in this study.
Material and Methods
Sites Description
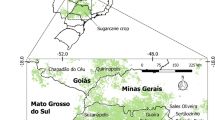
Four field experiments were carried out in commercial sugarcane farms located in Campinas (site 1), Paulínia (site 2), Quatá (site 3), and Sales Oliveira (site 4), in São Paulo state, southeast Brazil (Fig. 1). The experimental sites were strategically chosen to represent the sugarcane areas under different conditions of soil, weather, and harvesting season. Information on geographic coordinates, weather conditions, sugarcane variety, crop aging, harvesting period, and soil classification for each site is presented in Table 1. Sites 1 and 2 were evaluated during one sugarcane crop cycle (2016/2017), and sites 3 and 4 during two consecutive years (2016/2017 and 2017/2018). All sites have a history of sugarcane cultivation of over 20 years, and approximately 10 years of green mechanized harvesting without burning. The chemical and physical characterizations of soil attributes prior to establishing the experiments in each site are detailed in Table 2.
Geographic locations of the sites evaluated in this study and those obtained from the literature. Blue squares denote the experimental sites and red triangles show the sites where data were compiled from the literature. Sugarcane cultivation map (green circle) was obtained according to the updated data from the Canasat’s project [20]
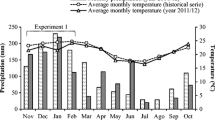
Weather data were recorded by automatic meteorological stations installed in the vicinity of the experimental sites, and the climatological water balance was calculated for each site according to methods proposed by Thornthwaite and Mather [24] (Fig. 2).
Experimental Design and Treatments
Experimental plots were arranged in a randomized block design with four treatments and four replicates for sites 1, 2, and 3 (16 plots in each location), and with three treatments and three replicates for site 4 (nine plots). Each plot was 9 m long by 9 m wide, comprising six rows of sugarcane at 1.5-m spacing. The treatments were established to evaluate the effects of same rates of straw removal under contrasting edaphoclimatic conditions. The average amount of straw produced in Brazilian sugarcane fields ranges from 10 to 20 Mg ha−1 [6, 25,26,27]; therefore, in this study, we established 15 Mg ha−1 of dry straw as the no removal (NR) treatment, in which all straw is maintained in the field. Subsequently, three straw removal rates were established: (i) removal of 5 Mg ha−1 (low removal (LR)); (ii) removal of 10 Mg ha−1 (high removal (HR)); and (iii) total removal (TR). In this study, four straw removal rates (NR, LR, HR, and TR) in sites 1, 2, and 3 and three (NR, HR, and TR) in site 4 were evaluated.
The amounts of straw on soil surface were quantified yearly after the harvesting of each sugarcane cycle using a metallic frame (0.25 m2) randomly thrown on the field ten times. The straw moisture measurement was performed directly in the field using the sensor AL-104 Agrologic® with E-831 Electrode coupling. After the quantification of the amount of straw produced annually in each site (on dry basis), the adjustment of the exact amount of straw in each experimental plot was performed manually using forage forks and rakes.
The fertilizer application in each site was performed approximately 30 days after harvest with 120 kg ha−1 year−1 of nitrogen and 120 kg ha−1 year−1 of potassium. The N fertilizer source was ammonium sulfate for sites 1 and 2, and ammonium nitrate for sites 3 and 4. The fertilizer was manually applied in bands 0.10 m apart from the sugarcane rows. During the experimental period, organic amendments such as vinasse and filter cake were not applied in the fields. Applications of fungicides, insecticides, and herbicides were uniform in all plots and conducted according to the management strategies as established by each sugarcane farm.
Measurements of N2O and CH4 Emissions
The sampling of CH4 and N2O was performed using static chambers installed in all experimental plots. In each plot, two chambers (0.30-m diameter × 0.22-m height) were used, one located in the fertilization band and the other in unfertilized band (interrow). In all sites, the chambers were installed 10 days prior to N fertilizer application with the goal of reducing soil disturbance effects. The bases of the chambers were buried into the soil at a 0.05-m depth. The amounts of straw and N fertilizer placed inside the chambers in the fertilized band were calculated considering the chamber diameter. Specifically, 5.4 g of N fertilizer was applied within each chamber, and the amount of straw was dependent on the treatment.
GHG samplings were carried out over the periods of 264 (site 1), 293 (site 2), 324 (site 3), and 332 (site 4) days in the 2016/2017 crop year, and of 279 (site 3) and 345 (site 4) days in the 2017/2018 crop year. Gas sampling was performed daily on the first five days after fertilization (DAF) and thereafter, three times per week up to 90 DAF. After this period, the evaluations were performed weekly, bi-weekly, and monthly as the emissions subsided. Forty milliliters of gas samples was collected using a BD nylon syringe (Becton Dickson Ind. Cirur. Ltda), after incubation of 0, 15, and 30 min. The samplings were always taken in the morning, between 8 and 10 a.m., in order to standardize the GHG sampling time. After that, the GHG samples were transferred to vacuum glass bottles. Simultaneously, soil moisture and temperature measurements were performed using the MPS 2 Decagon® sensor installed at a depth of 0.10 m.
All gas samples were analyzed in a Shimadzu® gas chromatograph (GC-2014) equipped with a packed column, an electron capture detector to analyze N2O, and a flame ionization detector to analyze CH4. The flux of each gas was calculated using the linear change in the concentration inside the chamber as a function of the incubation time (30 min). Atmospheric pressure, and chamber height and air temperature were measured concurrently with gas sampling to determine air chamber volume and calculate GHG emissions. The daily gas flux was calculated according to Eq. 1.
where F is the GHG (CH4 or N2O) flux (μg m−2 h−1), ΔC/Δt is the rate of change of the gas concentration inside the chamber during the incubation time (μmol mol−1 h−1), V is the chamber volume (L), Vm is the molar volume of the gas at the sampling temperature (L mol−1), m is the molecular mass of each GHG (N = 14, C = 12), and A is the area of soil covered by the chamber (m2). After this step, the fluxes of CH4 and N2O (in μg m−2 h−1) were converted and expressed in terms of grams per hectare per day.
Cumulative N2O and CH4 fluxes were calculated by linear interpolation between adjacent sampling dates [28]. We considered that the cumulative emissions by fertilized bands accounted for 20% of the total area and the interrow for 80%. The N2O emission factors were calculated based on the amounts of N applied as N fertilizer inside the chamber according to Eq. 2.
where \( {\mathrm{EF}}_{{\mathrm{N}}_2\mathrm{O}} \) is the emission factor (N2O-N as a percentage of N fertilizer applied); N2O-Ntreatment and N2O-Ncontrol are the cumulative N2O-N emissions from a specific treatment and control; and N fertilizer is the rate of N applied to the soil.
Literature Data
In parallel to the field experiments, we performed a literature review on the studies reporting N2O EFs derived from N fertilizers coupled with different amounts of straw maintained on soil surface of areas under sugarcane production in São Paulo state. We conducted a systematic search in the main databases (Web of Science, Scopus, and Google Scholar) to select publications presenting the following keywords: “sugarcane straw,” “sugarcane trash,” “sugarcane crop residues,” “N fertilization,” “N2O emissions,” “N2O emission factor,” “greenhouse gas emissions,” and “São Paulo state, Brazil.” After matching the keywords, our search yielded seven papers. To filter the results, we used additional criteria, such as the following: (i) only studies that evaluated N fertilizer and straw removal rates; (ii) studies performed under field conditions, excluding glasshouse and laboratory incubation trials. After that, only three papers were selected to compose our dataset. The results obtained in the literature were grouped with our field data to derive direct N2O EFs that represent the regional-specific conditions of sugarcane fields in São Paulo state, Brazil.
Calculation of Direct N2O Emissions from N Fertilizer and Straw
The calculation of the direct N2O emissions derived from N fertilizer plus straw was performed using the default N2O EF proposed by the IPCC [14] and the regional-specific N2O EF (Tier 2) obtained from the entire dataset of this study (e.g., our field studies plus literature review). To estimate the direct N2O emissions using the Tier 1 approach, the N2O EF of 1% was applied for the amount of N provided by the synthetic fertilizer and for the total amount of N added by sugarcane straw [14]. Thus, to estimate the straw N input, we assumed a N content in the straw of 4.9 g N kg−1 [6] and the respective amount of straw associated with each treatment. For Tier 2, we used the regional-specific N2O EFs derived from the application of N fertilizer in sugarcane areas coupled with different amounts of straw on the soil surface. Total N2O emissions were converted into CO2 equivalent using a global warming potential of 265 [10] and results were expressed in terms of kilogram CO2 eq per hectare.
Statistical Analysis
Analysis of variance (ANOVA) was performed to assess differences in CH4 and N2O cumulative emissions among treatments, and Tukey’s test (p value < 0.05) was used to compare means. The ANOVA and Tukey’s test were performed using the Sisvar® software [29]. Pearson’s correlation analysis was carried out in the R software with the package “PerformanceAnalytics” [30].
Results
Methane Emissions
The daily CH4 fluxes observed in all sites varied from negative to positive values. In general, the fluxes varied from − 10 to 10 g ha−1 day−1 for sites 1, 2, and 3 and from − 20 to 10 g ha−1 day−1 for site 4 (Fig. 3A–D). The cumulative CH4 fluxes were low for all sites, and in most cases, it indicated a modest consumption of CH4 by the soil (Fig. 3). In site 1, CH4 consumption was observed for all treatments with significant differences (p < 0.05) among them (Fig. 3A). In site 2, CH4 consumption (− 0.27 kg ha−1 year−1) was observed only in HR treatment, and low CH4 emissions were observed in other treatments (Fig. 3B). No significant differences among treatments were observed in sites 3 and 4 in both years of evaluation (Fig. 3C, D).
Daily (g ha−1 day−1) and cumulative (kg ha−1 year−1) emissions of CH4 in areas under different scenarios of sugarcane straw removal, São Paulo state, Brazil. Straw removal rates: total removal (TR), high removal (HR), low removal (LR), and no removal (NR). Panels A, B, C, and D represent the CH4 emissions at site 1, site 2, site 3, and site 4, respectively. Cumulative emission values followed by the same letters do not differ significantly by Tukey’s test (p < 0.05) and “ns” means no significant difference across treatments
Nitrous Oxide Emissions
Higher N2O emissions were observed after N fertilizer application following rainfall periods, with significant variations among sites (Fig. 4). In site 1, straw removal and N fertilization were performed in the dry season and, thus, the peaks of N2O emission started 70 DAF simultaneously with rainfall events. The daily emissions ranged from 0.9 to 45.6, from 1.2 to 52.9, from 1.5 to 39.5, and from 1.0 to 68.3 g N2O ha−1 day−1 for treatments TR, HR, LR, and NR, respectively (Fig. 4A). The cumulative N2O emissions varied significantly across treatments (p < 0.05), and values of 1.08, 1.90, 1.84, and 2.41 kg ha−1 year−1 were observed for TR, HR, LR, and NR, respectively (Fig. 4A).
Daily (g ha−1 day−1) and cumulative (kg ha−1 year−1) emissions of N2O in areas under different scenarios of sugarcane straw removal, São Paulo state, Brazil. Straw removal rates: total removal (TR), high removal (HR), low removal (LR), and no removal (NR). Panels A, B, C, and D represent the CH4 emissions at site 1, site 2, site 3, and site 4, respectively. Cumulative emission values followed by the same letters do not differ significantly by Tukey’s test (p < 0.05) and “ns” means no significant difference across treatments
For sites 2, 3, and 4, peaks of N2O emission were observed soon after N fertilizer application (Fig. 4B–D). Increases in N2O emissions were observed after N fertilization in sites 2 and 3, but the magnitude of changes was clearly site-specific. Peaks of N2O were observed at 30 DAF, with emissions of 118, 85, and 7 g N2O-N ha−1 day−1 for sites 2, 3, and 4 in the first-year evaluation, respectively (Fig. 4B–D). In the second year, site 3 presented N2O emissions 60% lower compared with those in the first year, while the opposite was observed in site 4, where N2O emissions were twice as high as those observed in the first year (Fig. 4C, D).
The cumulative N2O emissions in site 2 were significantly different among treatments, with values of 1.64, 2.10, 4.09, and 2.80 kg ha−1 year−1 for TR, HR, LR, and NR, respectively (Fig. 4B). In site 3, no differences between treatments were observed in both years, and the cumulative N2O emissions ranged from 1.31 to 1.56 kg ha−1 and from 0.97 to 1.35 kg ha−1 year−1 in the first and second years, respectively (Fig. 4C). In site 4, the lower daily and cumulative N2O emissions were observed for all treatments in both years of evaluation, with cumulative emissions ranging from 0.20 to 0.27 kg ha−1 and from 0.24 to 0.38 kg ha−1 year−1 in the first and second years, respectively.
Correlation Between CH4 and N2O Emissions and Soil Attributes
Significant Pearson correlation between CH4 and soil attributes was observed only in site 4 (Table 3). Methane emission was directly correlated with NO3− content (p < 0.05) and soil moisture (p < 0.001) and inversely correlated with soil temperature (p < 0.001). In most sites, N2O emissions were directly correlated with soil temperature, moisture, and NH4+ content (p < 0.01, p < 0.001). Inverse correlations were also observed between N2O emissions and soil NH4+ content in the sites 1 and 2 (Table 3).
N2O Emission Factors
The direct N2O EFs from N fertilizer application were highly variable across sites, ranging from 0.05 to 1.44% of the N applied (Table 4). In site 1, the EFs were significantly increased with the maintenance of straw on the soil surface. Similarly, the N2O EFs in site 2 increased with large amount of straw on the soil surface (p < 0.05), with the highest N2O EF occurring in LR treatment. No differences between treatments were observed in site 3 in both years of evaluation, and higher values of N2O EF (from 0.42 to 0.69%) were observed in the first year in comparison with the second year (from 0.23 to 0.38%). Site 4 presented lower N2O EF in relation to the other sites, and differences across treatments were observed only in the second year.
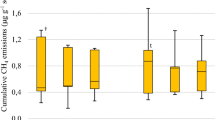
By grouping the N2O EF values found in this study with those summarized from the literature (Table 4), we observed decrease in N2O EFs as a function of straw removal. Averaged N2O EFs of 0.28, 0.44, 0.70, and 0.56% were observed in the following straw removal rates: TR, HR, LR, and NR, respectively (Fig. 5). There was clear evidence of lower N2O EF with lower amount of straw on soil, despite the high variability of the data indicated by the boxplots.
Direct N2O emission factors from the application of N fertilizers in soils under different straw removal rates in São Paulo state, Brazil. Straw removal rates: total removal (TR), high removal (HR), low removal (LR), and no removal (NR). n, number of observations. Internal asterisks and lines represent the mean and median, respectively. Black dash line indicates the IPCC default value
Direct N2O Emissions from N Fertilizer plus Straw Removal
Using the Tier 1 approach, the direct N2O emissions from N fertilizer and straw N (expressed in CO2 eq) from sugarcane soils were 500, 602, 704, and 806 kg CO2 eq ha−1 for TR, HR, LR, and NR, respectively (Fig. 6). Conversely, using the regional-specific EF data (Tier 2), the direct N2O emissions from N fertilizer plus straw were 139, 218, 351, and 280 kg CO2 eq ha−1 for TR, HR, LR, and NR, respectively.
Direct N2O emissions (kg CO2 eq ha−1) due to synthetic N fertilizer and straw in scenarios of sugarcane straw removal, São Paulo state, Brazil. Straw removal rates: total removal (TR), high removal (HR), low removal (LR), and no removal (NR). Annual N fertilizer rate, 120 kg ha−1. N content in the straw, 4.9 g kg−1 [6]. Tier 1 - IPCC approach; Tier 2 - Regional-specific approach based on data from this study
Discussion
Implications of Straw Removal for Soil CH4 Emissions
Sugarcane ethanol has a high potential to mitigate GHG emissions from fossil fuels and other bioenergy crops [3], but the effective mitigation potential is highly influenced by management practices adopted within the sugarcane production chain. Few studies have presented data on CH4 emissions from sugarcane management practices in Brazil, and most of them indicate low emissions or consumption of CH4 from sugarcane soils [11, 19, 32, 33]. These studies indicate that site-specific conditions including soil type, climate conditions, tillage, irrigation, and straw mulching can determine whether the soil is a sink or a source of CH4.
No clear effect of straw removal on CH4 fluxes was observed in all locations evaluated in this study. Considering that sugarcane in Brazil is predominantly cultivated in deep, well-drained, and highly weathered soils [3], the combination of temperature, water content, and available C provided by crop residues seems to favor the methanotrophic bacteria activity and increase CH4 oxidation [34], resulting in low emissions or even consumption of CH4. Additionally, the maintenance of straw improves soil physical conditions [8], which probably decreases the microsites of anaerobiosis, offsetting the effect of the high organic C input, thus reducing CH4 emissions in sugarcane fields.
In our study, the highest CH4 uptake over 2 years was observed in site 4 (Fig. 3d), which may be attributed to the combined effect of lower soil bulk density and higher macroporosity (Table 2), indicating an inverse correlation between soil compaction and CH4 consumption. Site 4 is a well-drained Oxisol with high clay content (710 g kg−1) and high macroporosity (Table 2), which provides good drainage and tends to promote CH4 oxidation [33, 35], thereby making this soil a natural sink of CH4. Recent results indicate consistent CH4 consumption in areas under lower soil compaction [36]. Aligned with these findings, the CH4 consumption in site 4 was inversely correlated with temperature and directly with soil moisture (Table 3), which is consistent with findings that the straw mulching increases soil moisture and reduces soil temperature [37]. The mechanism of CH4 consumption in Brazilian sugarcane soils is likely to be associated with the “low-affinity oxidation,” in which CH4 is produced in microsites of anaerobiosis, diffused into a more aerated region, and then converted into CO2 by methanotrophic bacteria [38].
Some studies pointed out that clayey soils emitted less CH4 than sandy soils [39,40,41], especially because clay particles favor the trapping of CH4 bubbles in soils, decreasing such emissions [39]. In this way, the highest CH4 uptake was observed in the site with higher clay content (Fig. 3D). However, site-specific conditions may prevail because sites 1 and 2 also present high clay content but showed a different pattern of CH4 emissions. In addition, substantial variability of CH4 fluxes (i.e., from emissions to consumption of CH4) is quite common in sugarcane cultivated soils, and other environmental factors that regulate CH4 dynamics could make data interpretation difficult. Notwithstanding, our data indicate that sugarcane soils, regardless of straw removal rates, act predominantly as a net sink for CH4, in line with other studies conducted in sugarcane fields in Brazil [11, 19, 32, 33].
Implications of Straw Removal for Soil N2O Emissions
Despite being highly variable, the daily N2O emissions (ranging from 0.2 to 4.1 kg N2O-N ha−1 year−1) observed in this study are of the same magnitude as those reported in other studies conducted in sugarcane in Brazil [11, 12, 17, 18, 31]. The N2O emissions induced by N fertilizer plus straw removal in sugarcane fields were clearly site-specific (Fig. 4). In agreement with previous studies [11, 12, 18, 42, 43], our findings confirmed a distinct pattern of N2O emissions when N fertilizer is applied during the dry and the wet seasons, with higher N2O emissions in the wet season (site 2). Enhanced N2O emissions occurred when N fertilizer was applied in the rainy season (Fig. 4B), probably due to the increase in moisture condition and fast organic C mineralization, thus favoring the soil microbial activity [44].
Sandy soils are commonly seen as well-drained, it can produce lower N2O emissions than clay soils [45]. However, in this study, the coarse-textured soil (site 3) showed lower soil macroporosity (Table 2) and higher N2O emissions relative to a clay-textured soil (site 4). The coarse-textured soil (site 3) was also compacted (Table 1) and exhibited low water infiltration rates (data not shown). Soils on sites 1, 2, and 4 presented similar clay content (> 60%) but exhibited distinct bulk density and macro- and microporosity distribution. Increased proportions of micropores with consequent reduction in macropores in sites 1 and 2 indicate that a higher tension is required for soil drainage and air diffusion [46], allowing anaerobic microsite formation where nitrifier-denitrification and denitrification can occur [47, 48]. Our results did not provide a clear association between clay content and N2O emissions, suggesting that other factors including soil physical quality and/or local soil microbial community can determine the magnitude of such emissions.
Averaged N2O emissions in site 4 were approximately 85% and 90% lower than those observed in sites 1 and 2, respectively. As already discussed for the CH4 emission data, the low N2O emissions in site 4 are likely to be associated with low soil bulk density and high macroporosity (Table 2), since this area had been manually harvested without machinery traffic in the last 4 years. Other studies have indicated high N2O emissions in soils under low macroporosity and high soil bulk density [36, 46]. Tullberg et al. [36] reported that the adoption of controlled traffic could reduce soil N2O emissions by 30%, since this practice reduces soil trampling on sugarcane rows and improves soil physical conditions. In a comprehensive review about the effects of soil structure on N2O emissions, Ball et al. [49] concluded that the N2O production is mainly derived from “hot spots” activity and, therefore, mitigation strategies that increase soil porosity and the interaction between soil physical and biological processes should be a key priority. Additionally, soil pH may have had a dominant role in the lower N2O emissions from site 4. Wang et al. [50] reported that N2O emission induced by N fertilizer application is negatively correlated with soil pH, indicating that more acidic soils (like sites 1 and 2) can release significantly high N2O rates.
We observed increased soil temperature with straw removal from all sites, which may have caused the significant correlations between temperature and N2O emissions in sites 1 and 3 (Table 3). Similarly, other studies concluded that the N2O production, regardless of the pathway (nitrification or denitrification), is enhanced with increased soil temperature [51, 52]. Straw mulching also acts as a physical barrier and preserves soil moisture by avoiding direct exposure of soil to solar radiation while reducing the amplitude of soil temperature along the day [7]. By isolating the effects of straw N input and soil moisture, Fracetto et al. [52] concluded that the indirect effect mulching on soil moisture rather than straw N input is the main factor contributing to increases in N2O emission from sugarcane fields where straw was preserved. Similarly, Vargas et al. [35] observed twofold higher N2O emission under straw retention, indicating that soil moisture accelerates crop residue decomposition by microbial activity and, consequently, enhances N2O production. Besides soil attributes, sugarcane straw quality can affect N2O emissions in sugarcane soils. Sugarcane straw has a slow decomposition rate because of the high C/N ratio (100:1) and the high concentrations of lignin and polyphenols [6], which in turn affect N availability (immobilization of soil N), thus leading to lower N2O emissions.
Lower N2O emissions induced by straw removal on sites 1 and 2 are consistent with previous studies conducted in sugarcane fields in Brazil [11, 12, 19, 35, 53]. But this does not apply to all situations [31, 42] as observed in sites 3 and 4 (Fig. 4), where straw removal did not show significant influence on N2O emission. The high variability of N2O emissions observed in this study as well as those reported in the literature are associated with many factors, such as the large variations in edaphoclimatic conditions, long periods of sugarcane harvesting (extending from March to November), and soil microbiome heterogeneity, making it difficult to determine the overall effects of straw removal on N2O emissions [19, 42, 54].
The direct N2O EFs from N fertilizer plus straw in sugarcane soils are quite variable and are, on average, below the 1% value proposed by the IPCC [14]. This was confirmed in this study with an averaged N2O EF of 0.47% (including this study and literature data), which has also significantly changed in scenarios of straw removal (Fig. 5). The direct N2O EFs were 72%, 56%, 30%, and 44% lower than those proposed by the IPCC [14] for TR, HR, LR, and NR scenarios, respectively. Our findings indicate that straw removal could be considered a feasible strategy to reduce the direct N2O EFs from N fertilizers in sugarcane fields on a short-term basis. However, it is important to mention that this study evaluated only the implications of straw removal on N2O emissions and did not account for changes in SOC stocks. Several studies have indicated that straw is an important source of C to the soil [25, 55,56,57] and the indiscriminate removal of this crop residue depletes SOC stocks in sugarcane fields [58, 59]. Aligned with these results, previous studies on GHG balance of sugarcane ethanol have shown that SOC stock change is an important component of the overall GHG balance in sugarcane fields [5, 60, 61]. Based on that, we advocate that further studies should quantify the net GHG emission balance in scenarios of straw removal by taking into account the emissions of CH4 and N2O, and the changes in SOC stocks induced by straw removal for bioenergy production.
Additionally, we did not analyze the drawbacks of straw removal on soil quality [62] and nutrient recycling [63]. Nutrient recycling is one of the main reasons for maintaining straw in the field, and the indiscriminate removal should intensify the use of synthetic N fertilizers as well as that of other nutrients. Although several studies have indicated that only a small proportion (ranging from 3 to 30%) of the N from straw is mineralized over 1 year [64,65,66,67,68], suggesting that the maintenance of straw in the field every year is beneficial for improving the soil N budget in the long term [69]. Indeed, Trivelin et al. [63] used a modeling approach to show that maintaining straw in sugarcane fields over a 40-year period would lead to a reduction of N fertilizer inputs equivalent to 40 kg ha−1 year−1. Therefore, we suggest that additional studies should also include the effects of straw removal on N fertilizers requirements in the long-term, as well as the associated potential for reducing soil N2O emissions.
Estimation of N2O Emissions from N Fertilizer in Different Scenarios of Straw Removal
Since measured data on N2O emissions from sugarcane fields have not often been available to represent the Brazilian edaphoclimatic conditions, studies on life cycle assessment of sugarcane ethanol have usually relied on IPCC default data (e.g., recommended when regional-specific data are unavailable) to assess the GHG emissions associated with sugarcane cultivation [61, 70]. However, this approach involves many uncertainties and could not represent the prevailing conditions on a regional scale. Our findings indicated that regardless of treatments and/or sites, the average direct N2O EF value is consistently lower than the IPCC default value (Fig. 5).
By using a default N2O EF (1%) proposed by the IPCC [14], studies of life cycle assessment show that the N2O emissions derived from synthetic N fertilizers (40%) and sugarcane straw (9%) may represent around 49% of the total GHG emissions associated with sugarcane production [71]. However, the dataset presented herein indicates that the average N2O EF associated with N fertilization under scenarios of straw removal (ranging from 0.28 to 0.70%) is consistently lower compared with that of the IPCC Tier 1 approach (Fig. 5). Therefore, the share of N fertilizers plus straw in the overall GHG emissions associated with sugarcane production could be strongly reduced by applying the regional-specific EF obtained in this study. Large discrepancy of site-specific data in relation to IPCC default EF (1%) has also been reported in a global meta-analysis of N2O EF from agricultural soils, indicating an average N2O EF of 0.59 ± 0.27% for crop residues plus N fertilizer [72], which is of the same magnitude of those observed in this study. Based on these evidences, our results suggest that the use of regional-specific data (Tier 2) may represent an average reduction of 63% (ranging from 50 to 72%) in the direct N2O emissions from N fertilization in scenarios of straw removal (Fig. 6). This will improve the already good sustainability indicators of bioenergy derived from sugarcane [3].
Our findings are a step forward in providing regional-specific data to reduce the high level of uncertainty concerning N2O emissions assessments of sugarcane ethanol. However, it is important to highlight that these findings were obtained in short-term studies and may be associated with specific weather conditions. We advocate that long-term studies, as well as the use of biogeochemical models, should be encouraged in Brazil in order to obtain more accurate data on N2O emissions in sugarcane fields. Certainly, LCA users will be able to estimate GHG emissions of sugarcane ethanol using regional-specific N2O EF, thus better representing the prevalent edaphoclimatic conditions of sugarcane production in São Paulo state, Brazil. Furthermore, the data presented in this study may have practical relevance to help the sugarcane industry to make informed decisions on straw management as a key strategy of GHG mitigation and financial gains. To encourage the adoption of more sustainable management practices, the Brazilian government recently launched RenovaBio programme [73] that establishes mechanisms of financial compensation for bioenergy with low GHG emissions.
Conclusion
This study provides information to guide future inventories of the N2O emissions from sugarcane ethanol and how the straw management can affect such emissions on a short-term basis. Our findings suggest that GHG emissions are highly variable due to soil type, soil physical and chemical conditions, N fertilization season, and crop year, among others. Overall, this study shows clear evidence that CH4 fluxes are very low for all assessed sites regardless of straw removal rates, indicating a predominance of CH4 consumption by the soil. The N2O emissions induced by straw removal in sugarcane fields were clearly site-specific and, on average, straw removal can be used as a management target to reduce N2O emissions from sugarcane fields. Averaged regional N2O EFs of 0.28%, 0.44%, 0.70%, and 0.56% were observed for TR, HR, LR, and NR scenarios, respectively. This study indicates that the use of default N2O EF proposed by the IPCC may overestimate the direct N2O emissions from N fertilizer plus straw in the most intensively cultivated sugarcane region in southern Brazil and suggests that the use of the regional-specific N2O EF data can reduce the high levels of uncertainties concerning the GHG emissions of sugarcane bio-based products.
References
Tilman D, Socolow R, Foley JA, Hill J, Larson E, Lynd L, Pacala S, Reilly J, Searchinger T, Somerville C, Willians R (2009) Beneficial biofuels-the food, energy, and environment trilemma. Science 325:270–271. https://doi.org/10.1126/science.1177970
Goldemberg J (2007) Ethanol for a sustainable energy future. Science 315:808–8010. https://doi.org/10.1126/science.1137013
de Oliveira Bordonal R, Carvalho JLN, Lal R, Figueiredo EB, de Oliveira BG, La Scala N Jr (2018) Sustainability of sugarcane production in Brazil. A review. Agron Sustain Dev 38:13. https://doi.org/10.1007/s13593-018-0490-x
CONAB (2018) Acompanhamento da safra brasileira de cana-de-açúcar. In: Companhia Nacional de Abastecimento (Ed.), Quarto Levantamento - Safra 2018/19, 75p. https://www.conab.gov.br/info-agro/safras/cana/boletim-da-safra-de-cana-de-acucar. Accessed 01 May 2019
de Oliveira Bordonal R, de Figueiredo EB, Aguiar DA, Adami DA, Rudorff BFT, La Scala N Jr (2013) Greenhouse gas mitigation potential from green harvested sugarcane scenarios in São Paulo State, Brazil. Biomass Bioenergy 59:195–207. https://doi.org/10.1016/j.biombioe.2013.08.040
Menandro LMS, Cantarella H, Franco HCJ, Franco HCJ, Kolln OT, Pimenta MTB, Sanches GM, Rabelo SC, Carvalho JLN (2017) Comprehensive assessment of sugarcane straw: implications for biomass and bioenergy production. Biofuels Bioprod Biorefin 11:488–504. https://doi.org/10.1002/bbb.1760
Carvalho JLN, Nogueirol RC, Menandro LMS, de Oliveira Bordonal R, Borges CD, Cantarella H, Franco HCJ (2017) Agronomic and environmental implications of sugarcane straw removal: a major review. GCB Bioenergy 9:1181–1195. https://doi.org/10.1111/gcbb.12410
Castioni GA, Cherubin MR, Menandro LMS, Sanches GM, de Oliveira Bordonal R, Barbosa LC, Franco HCJ, Carvalho JLN (2018) Soil physical quality response to sugarcane straw removal in Brazil: a multi-approach assessment. Soil Tillage Res 184:301–309. https://doi.org/10.1016/j.still.2018.08.007
Corrêa STR, Barbosa LC, Menandro LMS, Scarpare FV, Reichardt K, de Moraes LO, Hernandes TAD, Franco HCJ, Carvalho JLN (2019) Straw removal effects on soil water dynamics, soil temperature, and sugarcane yield in south-central Brazil. Bioenergy Res. https://doi.org/10.1007/s12155-019-09981-w
IPCC (2013) In: Stocker TF, Qin D, Plattner GK, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge
do Carmo JB, Filoso S, Zotelli LC, de Sousa Neto R, Pitombo LM, Duarte-Neto PJ, Andrade CA, GJC G, Rossetto R, Cantarella H, Neto AE, Martinelli LA (2013) Infield greenhouse gas emissions from sugarcane soils in Brazil: effects from synthetic and organic fertilizer application and crop trash accumulation. GCB Bioenergy 5:267–280. https://doi.org/10.1111/j.1757-1707.2012.01199.x
Pitombo LM, do Carmo JB, de Hollander M, Rossetto R, Lopez MV, Cantarella H, Kuramae E (2016) Exploring soil microbial 16S rRNA sequence data to increase carbon yield and nitrogen efficiency of a bioenergy crop. GCB Bioenergy 8:867–879. https://doi.org/10.1111/gcbb.12284
Lourenço KS, Dimitrov MR, Pijl A, Soares JR, Carmo JB, van Veen JA, Cantarella H, Kuramae E (2018) Strategies to mitigate the nitrous oxide emissions from nitrogen fertilizer applied with organic fertilizers in sugarcane. Sci Total Environ 650:1476–1486. https://doi.org/10.1016/j.scitotenv.2018.09.037
IPCC (2006) In: Eggleston HS, Miwa K, Srivastava N, Tanabe K (eds) Guidelines for National Greenhouse Gas Inventories – a primer, prepared by the National Greenhouse Gas Inventories Programme. IGES, Hayama
Cantarella H (2007) Nitrogênio. In: Novaes RF, Hugo AV, Barros AV, Cantarutti RB (eds) Fertilidade do Solo. Sociedade Brasileira de Ciências do Solo, Viçosa, pp 375–470
Otto R, Castro SAQ, Mariano E, Castro SAQ, Mariano E, Castro SGQ, Franco HCJ, Trivelin PCO (2016) Nitrogen use efficiency for sugarcane-biofuel production: what is next? Bioenergy Research 9:1272–1289. https://doi.org/10.1007/s12155-016-9763-x
Lourenço KS, Dimitrov MR, Pijl A, Soares JR, Carmo JB, van Veen JA, Cantarella H, Kuramae EE (2018) Dominance of bacterial ammonium oxidizers and fungal denitrifiers in the complex nitrogen cycle pathways related to nitrous oxide emission. GCB Bioenergy:1–16. https://doi.org/10.1111/gcbb.12519
Soares JR, Cassman NA, Kielak AM, Pijl A, Carmo JB, Lourenço KS, Laanbroek HJ, Cantarella H, Kuramae E (2016) Nitrous oxide emission related to ammonia-oxidizing bacteria and mitigation options from N fertilization in a tropical soil. Sci Rep 6:30349. https://doi.org/10.1038/srep30349
Vasconcelos ALS, Cherubin MR, Feigl BJ, Cherubin MR, Feigl BJ, Cerri CEP, Gmach MR, Siqueira Neto M (2018) Greenhouse gas emission responses to sugarcane straw removal. Biomass Bioenergy 113:15–21. https://doi.org/10.1016/j.biombioe.2018.03.002
Canasat (2019) Sugarcane crop monitoring in Brazil by Earth observing satellite images. National Institute for Space Research (NIPE). http://www.dsr.inpe.br/laf/canasat/en/. Accessed 15 Apr 2019
Soil Survey Staff (2014) Keys to soil taxonomy, 12th edn. United States Department of Agriculture-Natural Resources Conservation Service, Washington, DC.
van Raij B, Andrade JC, Cantarella H, Quaggio JA (2001) Análise química para avaliação da fertilidade de solos tropicais, 1st edn. Instituto Agronômico, Campinas
Camargo OA, Moniz AC, Jorge JA, Valadares JMAS (2009) Métodos de análise química mineralógica e física de solos do Instituto Agronômico de Campinas. Instituto Agronômico, Campinas
Thornthwaite CW, Mather JR (1955) The water balance. In: Centeron NJ (ed) Publications in Climatology, vol VIII. Drexel Institute of Technology, Laboratory of Climatology, Philadelphia, p 104
Carvalho JLN, Otto R, Junqueira Franco HC, Ocheuze Trivelin PC (2013) Input of sugarcane post-harvest residues into the soil. Sci Agric 70:336–344. https://doi.org/10.1590/S0103-90162013000500008
Franco HCJ, Pimenta MTB, Carvalho JLN, Magalhães PSG, Rossell CEV, Braunbeck AO, Vitti AC, Kölln OT, Rossi Neto J (2013) Assessment of sugarcane trash for agronomic and energy purposes in Brazil. Sci Agric 70:305–312. https://doi.org/10.1590/S0103-90162013000500004
de Landell MGA, Scarpari MS, Xavier MA et al (2013) Residual biomass potential of commercial and pre-commercial sugarcane cultivars. Sci Agric 70:299–304. https://doi.org/10.1590/s0103-90162013000500003
Whittaker WJ, Robinson G (1967) The calculus of observations: an introduction to numerical analysis, 4th edn. Dover, New York
Ferreira DF (2000) In: Universidade de São (ed) Análises estatísticas por meio do Sisvar para Windows 4.0. Reunião anual da região brasileira da sociedade internacional de biometria [In Portuguese], São Carlos, pp 255–258
R Development Core Team (2015) R: a Language and Environment for Statistical Computing, R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org. Accessed 15 Jan 2019
Pitombo LM, Cantarella H, Packer APC, Ramos NP, Carmo JB (2017) Straw preservation reduced total N2O emissions from a sugarcane field. Soil Use Manag. https://doi.org/10.1111/sum.12384
de Oliveira BG, Carvalho JLN, Cerri CEP, Cerri CC, Feigl BJ (2013) Soil greenhouse gas fluxes from vinasse application in Brazilian sugarcane areas. Geoderma 200–201:77–84. https://doi.org/10.1016/j.geoderma.2013.02.005
Paredes DDS, Alves BJRR, Dos Santos MA, Bolonhezi D, Sant’Anna SAC, Urquiaga S, Lima MA, Boddey RM (2015) Nitrous oxide and methane fluxes following ammonium sulfate and vinasse application on sugar cane soil. Environ Sci Technol 49:11209–11217. https://doi.org/10.1021/acs.est.5b01504
Serrano-Silva N, Sarria-Guzmán L, Dendooven L, Luna-Guido (2014) Methanogenesis and methanotrophy in soil: a review. Pedosphere 24:291–307. https://doi.org/10.1016/S1002-0160(14)60016-3
Vargas VP, Cantarella H, Martins AA, Soares JR, Carmo JB, Andrade CA (2014) Sugarcane crop residue increases N2O and CO2 emissions under high soil moisture conditions. Sugar Tech 16:174–179. https://doi.org/10.1007/s12355-013-0271-4
Tullberg J, Antille DL, Bluett C, Eberhard J, Scheer C (2018) Controlled traffic farming effects on soil emissions of nitrous oxide and methane. Soil Tillage Res 176:18–25. https://doi.org/10.1016/j.still.2017.09.014
Gonzaga LC, Carvalho JLN, de Oliveira BG, Soares JR, Cantarella H (2018) Crop residue removal and nitrification inhibitor application as strategies to mitigate N2O emissions in sugarcane fields. Biomass Bioenergy 119:206–216. https://doi.org/10.1016/j.biombioe.2018.09.015
Moreira FMDS, Siqueira JO (2006) Microbiologia e bioquímica do solo, 2nd edn. Universidade Federal de Lavras, Lavras
Le Mer J, Roger P (2001) Production, oxidation, emission and consumption of methane by soils: a review. Eur J Soil Biol 37:25–50. https://doi.org/10.1016/S1164-5563(01)01067-6
Dorr H, Katruff L, Levin I (1993) Soil texture parameterization of the methane uptake in aerated soils. Chemosphere 26:697–713
Jarecki MK, Parkin TB, Chan ASK, Hatfield JL, Jones R (2008) Greenhouse gas emissions from two soils receiving nitrogen fertilizer and swine manure slurry. J Environ Qual 37:1432. https://doi.org/10.2134/jeq2007.0427
Siqueira Neto M, Galdos MV, Feigl BJ, Cerri CEP, Cerri CC (2016) Direct N2O emission factors for synthetic N-fertilizer and organic residues applied on sugarcane for bioethanol production in Central-Southern Brazil. GCB Bioenergy 8:269–280. https://doi.org/10.1111/gcbb.12251
Soares JR, Cantarella H, Vargas VP, Carmo JB, Martins AA, Sousa RM, Andrade CA (2015) Enhanced-efficiency fertilizers in nitrous oxide emissions from urea applied to sugarcane. J Environ Qual 44:423–430. https://doi.org/10.2134/jeq2014.02.0096
Samad MS, Bakken LR, Nadeem S, Clough TJ, Klein CAM, Richards KG, Lanigan GJ, Morales SE (2016) High-resolution denitrification kinetics in pasture soils link N2O emissions to pH, and denitrification to C mineralization. PLoS One 11:1–12. https://doi.org/10.1371/journal.pone.0151713
van Groenigen JW, Kasper GJ, Velthof GL, van Dasselaar AP, Kuikman PJ (2004) Nitrous oxide emissions from silage maize fields under different mineral nitrogen fertilizer and slurry applications. Plant Soil 263:101–111. https://doi.org/10.1023/B:PLSO.0000047729.43185.46
Harrison-Kirk T, Thomas SM, Clough TJ, Beare MH, van Weerden TW, Meenken ED (2015) Compaction influences N2O and N2 emissions from 15N-labeled synthetic urine in wet soils during successive saturation/drainage cycles. Soil Biol Biochem 88:178–188. https://doi.org/10.1016/j.soilbio.2015.05.022
Berisso FE, Schjønning P, Keller T, Lamandé M, Jonge LW, Iversen BV, Arvdisson J, Forkman J (2012) Persistent effects of subsoil compaction on pore size distribution and gas transport in a loamy soil. Soil Tillage Res 122:42–51. https://doi.org/10.1016/j.still.2012.02.005
Lourenço KS, Cassman NA, Pijl AS, van Veen JA, Cantarella H, Kuramae E (2018) Nitrosospira sp. govern nitrous oxide emissions in a tropical soil amended with residues of bioenergy crop. Front Microbiol:9. https://doi.org/10.3389/fmicb.2018.00674
Ball BC (2013) Soil structure and greenhouse gas emissions: a synthesis of 20 years of experimentation. Eur J Soil Sci 64:357–373. https://doi.org/10.1111/ejss.12013
Signor D, Cerri CEP, Conant R (2013) N2O emissions due to nitrogen fertilizer applications in two regions of sugarcane cultivation in Brazil. Environ Res Lett 8:015013. https://doi.org/10.1088/1748-9326/8/1/015013
Reeves S, Wang W, Salter B, Halpin N (2016) Quantifying nitrous oxide emissions from sugarcane cropping systems: optimum sampling time and frequency. Atmos Environ 136:123–133. https://doi.org/10.1016/j.atmosenv.2016.04.008
Fracetto FJC, Fracetto GGM, Bertini SCB, Bertini SCB, Cerri CC, Feigl BJ, Siqueira Neto M (2017) Effect of agricultural management on N2O emissions in the Brazilian sugarcane yield. Soil Biol Biochem 109:205–213. https://doi.org/10.1016/j.soilbio.2017.02.004
Wang WJ, Reeves SH, Salter B, Moody PW, Dalal RC (2016) Effects of urea formulations, application rates and crop residue retention on N2O emissions from sugarcane fields in Australia. Agric Ecosyst Environ 216:137–146. https://doi.org/10.1016/j.agee.2015.09.035
Malhi SS, Lemke R (2007) Tillage, crop residue and N fertilizer effects on crop yield, nutrient uptake, soil quality and nitrous oxide gas emissions in a second 4-yr rotation cycle. Soil Tillage Res 96:269–283. https://doi.org/10.1016/j.still.2007.06.011
Cerri CC, Galdos MV, Maia SMF, Bernoux M, Feigl BJ, Powlson D, Cerri CEP (2011) Effect of sugarcane harvesting systems on soil carbon stocks in Brazil: an examination of existing data. Eur J Soil Sci 62:23–28. https://doi.org/10.1111/j.1365-2389.2010.01315.x
Thorburn PJ, Meier EA, Collins K, Robertson FA (2012) Changes in soil carbon sequestration, fractionation and soil fertility in response to sugarcane residue retention are site-specific. Soil Tillage Res 120:99–111. https://doi.org/10.1016/j.still.2011.11.009
Carvalho JLN, Hudiburg TW, Franco HCJ, DeLucia EH (2017) Contribution of above- and belowground bioenergy crop residues to soil carbon. GCB Bioenergy 9:1333–1343. https://doi.org/10.1111/gcbb.12411
de Oliveira Bordonal R, Menandro LMS, Barbosa LC, Rattan L, Milori DMBP, Kölln OT, Franco HCJ, Carvalho JLN (2018) Sugarcane yield and soil carbon response to straw removal in south-central Brazil. Geoderma 328:79–90. https://doi.org/10.1016/j.geoderma.2018.05.003
Tenelli S, de Oliveira Bordonal R, Barbosa LC, Carvalho JLN (2019) Can reduced tillage sustain sugarcane yield and soil carbon if straw is removed? Bioenergy Research. https://doi.org/10.1007/s12155-019-09996-3
Galdos MV, Cerri CC, Cerri CEP, Paustian K, van Antwepen R (2010) Simulation of sugarcane residue decomposition and aboveground growth. Plant Soil 326:243–259. https://doi.org/10.1007/s11104-009-0004-3
de Figueiredo EB, La Scala N (2011) Greenhouse gas balance due to the conversion of sugarcane areas from burned to green harvest in Brazil. Agric Ecosyst Environ 141:77–85. https://doi.org/10.1016/j.agee.2011.02.014
Cherubin MR, da Oliveira DMS, Feigl BJ et al (2018) Crop residue harvest for bioenergy production and its implications on soil functioning and plant growth: a review. Sci Agric 75:255–272. https://doi.org/10.1590/1678-992x-2016-0459
Trivelin PCO, Franco HCJ, Otto R, Ferreira DA, Vitti AC, Fortes C, Faroni CE, Oliveira ECA, Cantarella H (2013) Impact of sugarcane trash on fertilizer requirements for São Paulo, Brazil. Sci Agric 70:345–352. https://doi.org/10.1590/S0103-90162013000500009
de Oliveira MW, Trivelin PCO, Kingston G, Barbosa MHP, Vitti AC (2002) Decomposition and release of nutrients from sugarcane trash in two agricultural environments in Brazil. Proceedings of the Australian Society of Sugar Cane Technology 24:1–10
Basanta MV, Dourado-Neto D, Reichardt K, Bacchi OOS, Oliveira JCM, Trivelin PCO, Timm LC, Tominaga TT, Correchel V, Cássaro FAM, Pires LF, de Macedo JR (2003) Management effects on nitrogen recovery in a sugarcane crop grown in Brazil. Geoderma 116:235–248. https://doi.org/10.1016/S0016-7061(03)00103-4
Gava GJC, Trivelin PCO, Vitti AC, de Oliveira MW (2005) Urea and sugarcane straw nitrogen balance in a soil-sugarcane crop system. Pesq Agrop Brasileira 40:689–695
Robertson FA, Thorburn PJ (2007) Decomposition of sugarcane harvest residue in different climatic zones. Aust J Soil Res 45:1–11. https://doi.org/10.1071/SR06079
Ferreira DA, Franco HCJ, Otto R, Vitti AC, Fortes C, Faroni CE, Garside AL, Trivelin PCO (2016) Contribution of N from green harvest residues for sugarcane nutrition in Brazil. GCB Bioenergy 8:859–866. https://doi.org/10.1111/gcbb.12292
Dourado-Neto D, Powlson D, Bakar RA et al (2010) Multiseason recoveries of organic and inorganic nitrogen-15 in tropical cropping systems. Soil Sci Soc Am J 74:139. https://doi.org/10.2136/sssaj2009.0192
Cavalett O, Chagas MF, Seabra JEA, Bonomi A (2013) Comparative LCA of ethanol versus gasoline in Brazil using different LCIA methods. Int J Life Cycle Assess 18:647–658. https://doi.org/10.1007/s11367-012-0465-0
Chagas MF, Bordonal RO, Cavalett O, Carvalho JLN, Bonomi A, La Scala JN (2016) Environmental and economic impacts of different sugarcane production systems in the ethanol biorefinery. Biofuels Bioprod Biorefin 10:89–106. https://doi.org/10.1002/bbb.1623
Charles A, Rochette P, Whalen JK, Angers DA, Chantigny MH, Bertrand N (2017) Global nitrous oxide emission factors from agricultural soils after addition of organic amendments: a meta-analysis. Agric Ecosyst Environ 236:88–98. https://doi.org/10.1016/j.agee.2016.11.021
MME (2017) Ministry of Mines and Energy. Renova Bio Programme. http://www.mme.gov.br/web/guest/secretarias/petroleo-gas-natural-e-combustiveis-renovaveis/programas/renovabio/principal. Accessed 26 Jul 2017
Acknowledgments
LCG and LCZ are grateful to the Brazilian Federal Agency for Support and Evaluation of Graduate Education – (CAPES) for providing doctoral fellowships. BGO thanks the São Paulo Research Foundation – FAPESP for providing the research grant (no. 2017/02299-4). HC is supported by grants from National Council for Scientific and Technological Development - CNPq (310478/2017-0) and FAPESP (2014/20593-9 and 2018/20793-9). We would like to thank LNBR’s group of technicians for all the support provided for field and laboratory activities, and the mills management for the experimental sites and the logistics support during the fieldwork.
Funding
This research was financially supported by the Sugarcane Renewable Electricity project — SUCRE/PNUD (grant number BRA/10/G31).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Gonzaga, L.C., Zotelli, L.d., de Castro, S.G.Q. et al. Implications of Sugarcane Straw Removal for Soil Greenhouse Gas Emissions in São Paulo State, Brazil. Bioenerg. Res. 12, 843–857 (2019). https://doi.org/10.1007/s12155-019-10006-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-019-10006-9