Abstract
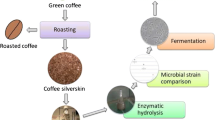
Coffee silverskin (CSS) is an agro-food waste obtained in large amount from the roasting phase of green coffee beans. Its potential use as biorefinery feedstock together with other agro-food wastes has been suggested, even though few experimental studies proved the production of bio-commodities at lab-scale from lignocellulosic sugars from CSS. The present work mainly aims at (i) the maximization of the sugar release by CSS pretreatment and hydrolysis; (ii) the fermentation of the CSS hydrolysate in order to produce butanol and succinic acid as bio-commodities having industrial interest. Alkali-pretreated CSS was enzymatically hydrolyzed in glass bottles on a rotary shaker. Biomass loading (5–12.5%w v−1) and cellulase concentration (1–80 FPU gcellulose−1) were the operating conditions investigated to maximize the sugar release. Biomass loading at 10%w v−1 gave the best results in terms of sugar concentration and sugar yield. Curve fitting of experimental data was performed: increasing of hydrolysis rate and sugar concentration was observed increasing cellulase concentration. Biomass loading at 10%w v−1 and enzyme loading at 80 FPU gcellulose−1 were applied to produce the hydrolysate to use as the medium for fermentation tests: the sugars from CSS were the only carbon source. The batch fermentations by Clostridium acetobutylicum DSM 792 and Actinobacillus succinogenes DSM 22257 were characterized in terms of metabolite and sugar conversion. Butanol (2.2 g L−1) and succinic acid (20.8 g L−1) were produced, respectively. The results highlight the possibility of using CSS to produce fermentable sugars, solvents, and biochemicals of industrial interest.






Similar content being viewed by others
Abbreviations
- ABE:
-
Acetone-butanol-ethanol
- AFWs:
-
Agro-food wastes
- AIL:
-
Acid-insoluble lignin
- ASL:
-
Acid-soluble lignin
- CSS:
-
Coffee silverskin
- SA:
-
Succinic acid
- YE:
-
Yeast extract
References
Fava F, Totaro G, Diels L, Reis M, Duarte J, Carioca OB, Poggi-Varaldo HM, Ferreira BS (2015) Biowaste biorefinery in Europe: opportunities and research & development needs. New Biotechnol 32:100–108. https://doi.org/10.1016/j.nbt.2013.11.003
Gallezot P (2007) Process options for converting renewable feedstocks to bioproducts. Green Chem 9:295. https://doi.org/10.1039/b615413a
Cherubini F (2010) The biorefinery concept : using biomass instead of oil for producing energy and chemicals. Energy Convers Manag 51:1412–1421. https://doi.org/10.1016/j.enconman.2010.01.015
Lee RA, Lavoie J-M (2013) From first- to third-generation biofuels: challenges of producing a commodity from a biomass of increasing complexity. Anim Front 3:6–11. https://doi.org/10.2527/af.2013-0010
Anwar Z, Gulfraz M, Irshad M (2014) Agro-industrial lignocellulosic biomass a key to unlock the future bio-energy: a brief review. J Radiat Res Appl Sci 7:163–173. https://doi.org/10.1016/j.jrras.2014.02.003
Sarkar N, Ghosh SK, Bannerjee S, Aikat K (2012) Bioethanol production from agricultural wastes: an overview. Renew Energy 37:19–27. https://doi.org/10.1016/j.renene.2011.06.045
Shi J, Liu J, Li M, Huang L, Chen L, Luo X (2018) Acid-free ethanol-water pretreatment with low ethanol concentration for robust enzymatic saccharification of cellulose in bamboo. Bioenergy Res 11:665–676. https://doi.org/10.1007/s12155-018-9928-x
Velmurugan R, Muthukumar K (2012) Ultrasound-assisted alkaline pretreatment of sugarcane bagasse for fermentable sugar production: optimization through response surface methodology. Bioresour Technol 112:293–299. https://doi.org/10.1016/j.biortech.2012.01.168
Russo ME, Niglio S, Procentese A, Marzocchella A (2018) Pretreatment of coffee silverskin with ultrasound and mild alkaline solutions for enhancement of sugar yield. New Biotechnol 44(Supplement):S38, ISSN 1871-6784. https://doi.org/10.1016/j.nbt.2018.05.1243
Kim I, Han J (2012) Optimization of alkaline pretreatment conditions for enhancing glucose yield of rice straw by response surface methodology. Biomass Bioenergy 46:210–217. https://doi.org/10.1016/j.biombioe.2012.08.024
Volynets B, Ein-Mozaffari F, Dahman Y (2017) Biomass processing into ethanol: pretreatment, enzymatic hydrolysis, fermentation, rheology, and mixing. Green Process Synth 6:1–22
Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577. https://doi.org/10.1128/MMBR.66.3.506
Cara C, Moya M, Ballesteros I, Negro MJ, González A, Ruiz E (2007) Influence of solid loading on enzymatic hydrolysis of steam exploded or liquid hot water pretreated olive tree biomass. Process Biochem 42:1003–1009. https://doi.org/10.1016/j.procbio.2007.03.012
Nanda S, Golemi-Kotra D, McDermott JC et al (2017) Fermentative production of butanol: perspectives on synthetic biology. New Biotechnol 37:210–221. https://doi.org/10.1016/j.nbt.2017.02.006
Qureshi N, Liu S, Hughes S, Palmquist D, Dien B, Saha B (2016) Cellulosic butanol (ABE) biofuel production from sweet Sorghum bagasse (SSB): impact of hot water pretreatment and solid loadings on fermentation employing Clostridium beijerinckii P260. Bioenergy Res 9:1167–1179. https://doi.org/10.1007/s12155-016-9761-z
Sarchami T, Rehmann L (2015) Optimizing acid hydrolysis of Jerusalem artichoke-derived inulin for fermentative butanol production. Bioenergy Res 8:1148–1157. https://doi.org/10.1007/s12155-014-9568-8
Raganati F, Götz P, Olivieri G et al (2015) Butanol production from hexoses and pentoses by fermentation of Clostridium Acetobutylicum. Anaerobe 34:146–155. https://doi.org/10.3303/CET1227016
Raganati F, Procentese A, Montagnaro F, Olivieri G, Marzocchella A (2014) Butanol production from leftover beverages and sport drinks. BioEnergy Res 8:369–379. https://doi.org/10.1007/s12155-014-9531-8
Zheng P, Dong J-J, Sun Z-H, Ni Y, Fang L (2009) Fermentative production of succinic acid from straw hydrolysate by Actinobacillus succinogenes. Bioresour Technol 100:2425–2429. https://doi.org/10.1016/j.biortech.2008.11.043
Li Q, Yang M, Wang D, Li W, Wu Y, Zhang Y, Xing J, Su Z (2010) Efficient conversion of crop stalk wastes into succinic acid production by Actinobacillus succinogenes. Bioresour Technol 101:3292–3294. https://doi.org/10.1016/j.biortech.2009.12.064
Ferone M, Raganati F, Olivieri G, Salatino P, Marzocchella A (2017) Biosuccinic acid from lignocellulosic-based hexoses and pentoses by Actinobacillus succinogenes: characterization of the conversion process. Appl Biochem Biotechnol 183:1465–1477. https://doi.org/10.1007/s12010-017-2514-4
Procentese A, Raganati F, Olivieri G, Russo ME, de la Feld M, Marzocchella A (2019) Agro food wastes and innovative pretreatments to meet biofuel demand in Europe. Chem Eng Technol accepted article. https://doi.org/10.1002/ceat.201800459
Niglio S, Procentese A, Russo ME et al (2017) Ultrasound-assisted dilute acid pretreatment of coffee silverskin for biorefinery applications. Chem Eng Trans 57:109–114. https://doi.org/10.3303/CET1757019
Ballesteros LF, Teixeira JA, Mussatto SI (2014) Chemical, functional, and structural properties of spent coffee grounds and coffee silverskin. Food Bioprocess Technol 7:3493–3503. https://doi.org/10.1007/s11947-014-1349-z
ICO (2017/18) International coffee organization. http://www.ico.org. Accessed November 2018
Saenger M, Hartge EU, Werther J, Ogada T, Siagi Z (2001) Combustion of coffee husks. Renew Energy 23:103–121. https://doi.org/10.1016/S0960-1481(00)00106-3
Didanna HL (2014) A critical review on feed value of coffee waste for livestock feeding. World J Biol Biol Sci 2:72–86
Giacobbe S, Pezzella C, Lettera V, Sannia G, Piscitelli A (2018) Laccase pretreatment for agrofood wastes valorization. Bioresour Technol 265:59–65. https://doi.org/10.1016/j.biortech.2018.05.108
Mussatto SI, Machado EMS, Carneiro LM, Teixeira JA (2012) Sugars metabolism and ethanol production by different yeast strains from coffee industry wastes hydrolysates. Appl Energy 92:763–768. https://doi.org/10.1016/j.apenergy.2011.08.020
Mussatto SI, Ballesteros LF, Martins S, Maltos DAF, Aguilar CN, Teixeira JA (2013) Maximization of fructooligosaccharides and β-fructofuranosidase production by Aspergillus japonicus under solid-state fermentation conditions. Food Bioprocess Technol 6:2128–2134. https://doi.org/10.1007/s11947-012-0873-y
Procentese A, Raganati F, Navarini L et al (2018) Coffee silverskin as a renewable resource to produce butanol and isopropanol. Chem Eng Trans 64. https://doi.org/10.3303/CET1864024
Sluiter A, Hames B, Ruiz R, et al (2012) Determination of structural carbohydrates and lignin in biomass. Laboratory Analytical Procedure (LAP); NREL/TP-510-42618
Adney B, Baker J (2008) Measurement of cellulase activities. Laboratory Analytical Procedure (LAP), NREL, TP 510 42628
Ghose TK (1987) Measurement of cellulase activities. Pure Appl Chem 59:257–268. https://doi.org/10.1351/pac198759020257
Monschein M, Reisinger C, Nidetzky B (2013) Enzymatic hydrolysis of microcrystalline cellulose and pretreated wheat straw: a detailed comparison using convenient kinetic analysis. Bioresour Technol 128:679–687. https://doi.org/10.1016/j.biortech.2012.10.129
Procentese A, Raganati F, Olivieri G, Elena Russo M, Marzocchella A (2017) Pre-treatment and enzymatic hydrolysis of lettuce residues as feedstock for bio-butanol production. Biomass Bioenergy 96:172–179. https://doi.org/10.1016/j.biombioe.2016.11.015
Wood IP, Elliston A, Ryden P, Bancroft I, Roberts IN, Waldron KW (2012) Rapid quantification of reducing sugars in biomass hydrolysates: improving the speed and precision of the dinitrosalicylic acid assay. Biomass Bioenergy 44:117–121. https://doi.org/10.1016/j.biombioe.2012.05.003
Montavon P, Kukic KR, Bortlik K (2007) A simple method to measure effective catalase activities: optimization , validation , and application in green coffee. 360:207–215. https://doi.org/10.1016/j.ab.2006.10.035
Kumar P, Barrett DM, Delwiche MJ, Stroeve P (2009) Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind Eng Chem Res 48:3713–3729. https://doi.org/10.1021/ie801542g
Ioelovich M, Morag E (2012) Study of enzymatic hydrolysis of pretreated biomass at increased solids loading. BioResources 7:4672–4682. https://doi.org/10.15376/biores.7.4.4672-4682
Xue Y, Jameel H, Phillips R, Chang HM (2012) Split addition of enzymes in enzymatic hydrolysis at high solids concentration to increase sugar concentration for bioethanol production. J Ind Eng Chem 18:707–714. https://doi.org/10.1016/j.jiec.2011.11.132
Zhu S, Wu Y, Yu Z, Liao J, Zhang Y (2005) Pretreatment by microwave / alkali of rice straw and its enzymic hydrolysis. Process Biochem 40:3082–3086. https://doi.org/10.1016/j.procbio.2005.03.016
Mussatto SI, Dragone G, Fernandes M, Milagres AMF, Roberto IC (2008) The effect of agitation speed, enzyme loading and substrate concentration on enzymatic hydrolysis of cellulose from brewer’s spent grain. Cellulose 15:711–721. https://doi.org/10.1007/s10570-008-9215-7
Pan X (2005) Strategies to enhance the enzymatic hydrolysis of pretreated softwood with high residual lignin content. Appl Biochem Biotechnol 121:1069–1079. https://doi.org/10.1385/ABAB:124:1-3:1069
Yáñez R, Alonso JL, Parajó JC (2006) Enzymatic saccharification of hydrogen peroxide-treated solids from hydrothermal processing of rice husks. Process Biochem 41:1244–1252. https://doi.org/10.1016/j.procbio.2005.12.020
Preeti VE, Sandhya SV, Kuttiraja M, Sindhu R, Vani S, Kumar SR, Pandey A, Binod P (2012) An evaluation of chemical pretreatment methods for improving enzymatic saccharification of chili postharvest residue. Appl Biochem Biotechnol 167:1489–1500. https://doi.org/10.1007/s12010-012-9591-1
Ludwig D, Michael B, Hirth T, et al (2014) High solids enzymatic hydrolysis of pretreated lignocellulosic materials with a powerful stirrer concept. pp 1699–1713. https://doi.org/10.1007/s12010-013-0607-2
Terán-hilares R, Reséndiz AL, Martínez RT, et al (2016) Bioresource Technology Successive pretreatment and enzymatic saccharification of sugarcane bagasse in a packed bed flow-through column reactor aiming to support biorefineries. 203:42–49. https://doi.org/10.1016/j.biortech.2015.12.026
Bansal P, Hall M, Realff MJ, Lee JH, Bommarius AS (2009) Modeling cellulase kinetics on lignocellulosic substrates. Biotechnol Adv 27:833–848. https://doi.org/10.1016/j.biotechadv.2009.06.005
Jeoh T, Cardona MJ, Karuna N, Mudinoor AR, Nill J (2017) Mechanistic kinetic models of enzymatic cellulose hydrolysis—a review. Biotechnol Bioeng 114:1369–1385
Klein-Marcuschamer D, Oleskowicz-Popiel P, Simmons BA, Blanch HW (2012) The challenge of enzyme cost in the production of lignocellulosic biofuels. Biotechnol Bioeng 109:1083–1087. https://doi.org/10.1002/bit.24370
Liu G, Zhang J, Bao J (2016) Cost evaluation of cellulase enzyme for industrial-scale cellulosic ethanol production based on rigorous Aspen Plus modeling. Bioprocess Biosyst Eng 39:133–140. https://doi.org/10.1007/s00449-015-1497-1
Millat T, Janssen H, Bahl H, et al (2011) The pH-induced metabolic shift from acidogenesis to solventogenesis in Clostridium acetobutylicum – from experiments to models. Beilstein-Institut, pp 33–54
Procentese A, Raganati F, Olivieri G, Russo ME, Marzocchella A (2019) Combined antioxidant-biofuel production from coffee silverskin. Appl Microbiol Biotechnol 103:1021–1029. https://doi.org/10.1007/s00253-018-9530-3
Gao K, Boiano S, Marzocchella A, Rehmann L (2014) Cellulosic butanol production from alkali-pretreated switchgrass (Panicum virgatum) and phragmites (Phragmites australis). Bioresour Technol 174:176–181. https://doi.org/10.1016/j.biortech.2014.09.152
Ezeji TC, Blaschek HP (2008) Fermentation of dried distillers’ grains and solubles (DDGS) hydrolysates to solvents and value-added products by solventogenic clostridia. Bioresour Technol 99:5232–5242. https://doi.org/10.1016/j.biortech.2007.09.032
Gao K, Rehmann L (2014) ABE fermentation from enzymatic hydrolysate of NaOH-pretreated corncobs. Biomass Bioenergy 66:110–115. https://doi.org/10.1016/j.biombioe.2014.03.002
Linggang S, Phang LY, Wasoh H (2013) Acetone – butanol – ethanol production by Clostridium acetobutylicum ATCC 824 using sago pith residues hydrolysate. Bioenergy Res 6:321–328. https://doi.org/10.1007/s12155-012-9260-9
Valsero MH, Cambronero JG, García AIP, Antolínez RD (2018) Biobutanol production from coffee silverskin. Microb Cell Factories 17:1–9. https://doi.org/10.1186/s12934-018-1002-z
Raganati F, Procentese A, Olivieri G et al (2016) Butanol production by Clostridium acetobutylicum in a series of packed bed biofilm reactors. Chem Eng Sci 152:678–688. https://doi.org/10.1007/s10295-010-0707-8
McKinlay JB, Laivenieks M, Schindler BD et al (2010) A genomic perspective on the potential of Actinobacillus succinogenes for industrial succinate production. BMC Genomics 11:680. https://doi.org/10.1186/1471-2164-11-680
Jiang M, Xu R, Xi Y-L, Zhang JH, Dai WY, Wan YJ, Chen KQ, Wei P (2013) Succinic acid production from cellobiose by Actinobacillus succinogenes. Bioresour Technol 135:469–474. https://doi.org/10.1016/j.biortech.2012.10.019
Funding
The present work has been funded by the European Union’s Horizon 2020 research and innovation program, project no. 654623 “Waste2Fuels - Sustainable production of next generation biofuels from waste streams.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 329 kb)
Rights and permissions
About this article
Cite this article
Niglio, S., Procentese, A., Russo, M.E. et al. Investigation of Enzymatic Hydrolysis of Coffee Silverskin Aimed at the Production of Butanol and Succinic Acid by Fermentative Processes. Bioenerg. Res. 12, 312–324 (2019). https://doi.org/10.1007/s12155-019-09969-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-019-09969-6