Abstract
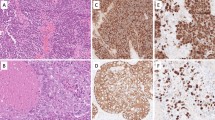
Mammary analogue secretory carcinoma (MASC) of the salivary glands is a recently described neoplasm of the salivary glands with a characteristic morphology complemented by a specific cytogenetic translocation and gene rearrangements. Although immunophenotypic and cytogenetic differences allow for a more reliable distinction, ultrastructural features can also provide important information about the relationship between MASC, classic acinic cell carcinoma (AciCC), and AciCC intercalated duct cell-predominant variant. Following approval from the hospital’s institutional review board, 7 cases of MASC, 8 cases of classic AciCC, and 4 cases of AciCC intercalated duct cell-predominant variant were retrieved from the pathology files of Massachusetts General Hospital from 2012 to 2015. Electron microscopy was performed using formalin-fixed, paraffin-embedded tissue. Ultrastructural features of all 19 neoplasms of the salivary glands were recorded. The predominant cell-types observed in MASC are those with intercalated/striated duct cell differentiation. These features include prominent invaginations of the cell surface studded with microvilli, and some intra- and intercellular lumina also with a microvillous surface. Classic AciCC dominant cell-type recapitulates acinar cell differentiation. These cells contain large intracytoplasmic zymogen-like granules. AciCC intercalated duct cell-predominant variant showed both cell populations in various proportions with the intercalated/striated duct cell type usually being the dominant one. MASC presents with distinctive ultrastructural features that allows its proper differentiation from classic AciCC. However, significant ultrastructural features overlaps between both AciCC intercalated duct cells-predominant and classic AciCC and MASC. These findings indicate a very close proximity between these tumors.



Similar content being viewed by others
References
Skálová A, Vanecek T, Sima R, Laco J, Weinreb I, Perez-Ordonez B, Starek I, Geierova M, Simpson RH, Passador-Santos F, Ryska A, Leivo I, Kinkor Z, Michal M. Mammary analogue secretory carcinoma of salivary glands, containing the ETV6-NTRK3 fusion gene: a hitherto undescribed salivary gland tumor entity. Am J Surg Pathol. 2010;34(5):599–608.
Drilon A, Li G, Dogan S, Gounder M, Shen R, Arcila M, Wang L, Hyman DM, Hechtman J, Wei G, Cam NR, Christiansen J, Luo D, Maneval EC, Bauer T, Patel M, Liu SV, Ou SH, Farago A, Shaw A, Shoemaker RF, Lim J, Hornby Z, Multani P, Ladanyi M, Berger M, Katabi N, Ghossein R, Ho AL. What hides behind the MASC: clinical response and acquired resistance to entrectinib after ETV6-NTRK3 identification in a mammary analogue secretory carcinoma (MASC). Ann Oncol. 2016;27(5):920–6.
Luk PP, Selinger CI, Eviston TJ, Lum T, Yu B, O’Toole SA, Clark JR, Gupta R. Mammary analogue secretory carcinoma: an evaluation of its clinicopathological and genetic characteristics. Pathology. 2015;47(7):659–66.
Skálová A, Vanecek T, Simpson RH, Laco J, Majewska H, Baneckova M, Steiner P, Michal M. Mammary Analogue Secretory Carcinoma of Salivary Glands: Molecular Analysis of 25 ETV6 Gene Rearranged Tumors With Lack of Detection of Classical ETV6-NTRK3 Fusion. Transcript by Standard RT-PCR: Report of 4 Cases Harboring ETV6-X Gene Fusion. Am J Surg Pathol. 2016;40(1):3–13.
Shah AA, Wenig BM, LeGallo RD, Mills SE, Stelow EB. Morphology in conjunction with immunohistochemistry is sufficient for the diagnosis of mammary analogue secretory carcinoma. Head Neck Pathol. 2015;9(1):85–95.
Patel KR, Solomon IH, El-Mofty SK, Lewis JS Jr, Chernock RD. Mammaglobin and S-100 immunoreactivity in salivary gland carcinomas other than mammary analogue secretory carcinoma. Hum Pathol. 2013;44(11):2501–8.
Skalova A. Mammary analogue secretory carcinoma of salivary gland origin: an update and expanded morphologic and immunohistochemical spectrum of recently described entity. Head Neck Pathol. 2013;7(Suppl 1):S30–6.
Pinto A, Nosé V, Rojas C, Fan YS, Gomez-Fernandez C. Searching for mammary analogue [corrected] secretory carcinoma of salivary gland among its mimics. Mod Pathol. 2014;27(1):30–7.
Bishop JA. Unmasking MASC: bringing to light the unique morphologic, immunohistochemical and genetic features of the newly recognized mammary analogue secretory carcinoma of salivary glands. Head Neck Pathol. 2013;7(1):35–9.
Chiosea SI, Griffith C, Assaad A, Seethala RR. Clinicopathological characterization of mammary analogue secretory carcinoma of salivary glands. Histopathology. 2012;61(3):387–94.
Ito Y, Ishibashi K, Masaki A, Fujii K, Fujiyoshi Y, Hattori H, Kawakita D, Matsumoto M, Miyabe S, Shimozato K, Nagao T, Inagaki H. Mammary analogue secretory carcinoma of salivary glands: a clinicopathologic and molecular study including 2 cases harboring ETV6-X fusion. Am J Surg Pathol. 2015;39(5):602–10.
Urano M, Nagao T, Miyabe S, Ishibashi K, Higuchi K, Kuroda M. Characterization of mammary analogue secretory carcinoma of the salivary gland: discrimination from its mimics by the presence of the ETV6-NTRK3 translocation and novel surrogate markers. Hum Pathol. 2015;46(1):94–103.
Griffith C, Seethala R, Chiosea SI. Mammary analogue secretory carcinoma: a new twist to the diagnostic dilemma of zymogen granule poor acinic cell carcinoma. Virchows Arch. 2011;459(1):117–8.
Resta L, Piscitelli D, Fiore MG, Sanguedolce F. Role of electron microscopy in diagnosis of parotid tumours. Acta Otorhinolaryngol Ital. 2005;25(3):150–2.
Dardick I, George D, Jeans MT, Wittkuhn JF, Skimming L, Rippstein P, van Nostrand AW. Ultrastructural morphology and cellular differentiation in acinic cell carcinoma. Oral Surg Oral Med Oral Pathol. 1987;63(3):325–34.
Lei Y, Chiosea SI. Re-evaluating historic cohort of salivary acinic cell carcinoma with new diagnostic tools. Head Neck Pathol. 2012;6(2):166–70.
Chaudhry AP, Cutler LS, Leifer C, Labay G, Satchidanand S, Yamane GM. Ultrastructural study of the histogenesis of salivary gland mucoepidermoid carcinoma. J Oral Pathol Med. 1989;18(7):400–9.
Chiosea SI, Griffith C, Assaad A, Seethala RR. The profile of acinic cell carcinoma after recognition of mammary analog secretory carcinoma. Am J Surg Pathol. 2012;36(3):343–50.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain ay studies with human participants performed by the authors.
IRB Approval
This Project was approved by the Massachusetts General Hospital Internal Review Board (Sadow, PI; 2015P001749).
Rights and permissions
About this article
Cite this article
Guilmette, J., Nielsen, G.P., Faquin, W.C. et al. Ultrastructural Characterization of Mammary Analogue Secretory Carcinoma of the Salivary Glands: A Distinct Entity from Acinic Cell Carcinoma?. Head and Neck Pathol 11, 419–426 (2017). https://doi.org/10.1007/s12105-017-0784-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-017-0784-3