Abstract
Objectives
MicroRNA-200 family (miR-200f) has been consistently reported to be deregulated and modulate the metastatic process in multiple cancers. In the present study, we detected the expression of miR-200f in breast cancer (BC) tissue and explored its relationships with clinicopathological characteristics, especially with lymph node metastasis.
Methods
Expression levels of miR-200a, miR-200b, miR-200c, miR-141, and miR-429 in 99 pairs of BC tissues and adjacent normal tissues were measured by real-time quantitative PCR. The correlation between miR-200f level and multiple clinicopathological factors was then examined by Mann–Whitney test, ANOVA, and operating characteristic (ROC) analysis.
Results
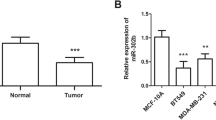
All members of the miR-200f were down-regulated in BC tissue compared with that in normal adjacent tissue; miR-200a, miR-200b, and miR-200c were highly decreased (p < 0.05), while the differences of miR-141 and miR-429 between patients and the control group were not statistically significant. Furthermore, all five members were found to be distinctly decreased with the incidence of lymph node metastasis (p < 0.05); When the patients were divided into three groups according to the number of lymph node metastasis (0; 1–3; ≥4), a gradual decrease of miR-200f expression was observed with the increasing number of lymph node metastasis; ROC revealed that miR-200b can differentiate patients with lymph node metastasis from those without lymph node metastasis.
Conclusion
These observations imply that the down-regulation of miR-200f in human BC is associated with an invasive phenotype, and miR-200b may be useful to estimate the likelihood of the presence of pathologically positive lymph nodes.


Similar content being viewed by others
References
Redig AJ, McAllister SS. Breast cancer as a systemic disease: a view of metastasis. J Intern Med. 2013;274(2):113–26.
Wang L, Wang J. MicroRNA-mediated breast cancer metastasis: from primary site to distant organs. Oncogene. 2012;31(20):2499–511.
Huber MA, Kraut N, Beug H. Molecular requirements for epithelial–mesenchymal transition during tumor progression. Curr Opin Cell Biol. 2005;17(5):548–58.
Shi M, Liu D, Duan H, Shen B, Guo N. Metastasis-related miRNAs, active players in breast cancer invasion, and metastasis. Cancer Metastasis Rev. 2010;29:785–99.
Bouyssou JM, Manier S, Huynh D, Issa S, Roccaro AM, Ghobrial IM. Regulation of microRNAs in cancer metastasis. Biochim Biophys Acta. 2014;45(2):255–65.
Hurst DR, Edmonds MD, Welch DR. Metastamir: the field of metastasis-regulatory. microRNA is spreading. Cancer Res. 2009;69(19):7495–8.
Park SM, Gaur AB, Lengyel E, Peter ME. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008;22:894–907.
Korpal M, Lee ES, Hu G, Kang Y. The miR-200 family inhibits epithelial–mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem. 2008;283:14910–4.
Liu YN, Yin JJ, Abou-Kheir W, Hynes PG, Casey OM, Fang L, et al. MiR-1 and miR-200 inhibit EMT via slug-dependent and tumorigenesis via slug-independent mechanisms. Oncogene. 2013;32:296–306.
Choi PS, Zakhary L, Choi WY, Caron S, Alvarez-Saavedra E, Miska EA, et al. Members of the miRNA-200 family regulate olfactory neurogenesis. Neuron. 2008;57:41–55.
Baffa R, Fassan M, Volinia S, O’Hara B, Liu CG, Palazzo JP, et al. MicroRNA expression profiling of human metastatic cancers identifies cancer gene targets. J Pathol. 2009;219:214.
Tuomarila M, Luostari K, Soini Y, Kataja V, Kosma VM, Mannermaa A. Overexpression of microRNA-200c predicts poor outcome in patients with PR-negative breast cancer. PLoS One. 2014;9(10):e109508.
Yu SJ, Hu JY, Kuang XY, Luo JM, Hou YF, Di GH, et al. MicroRNA-200a promotes anoikis resistance and metastasis by targeting YAP1 in human breast cancer. Clin Cancer Res. 2013;19(6):1389–99.
Zhang HF, Xu LY, Li EM. A family of pleiotropically acting microRNAs in cancer progression, miR-200: potential cancer therapeutictargets. Curr Pharm Des. 2014;20(11):1896–903.
Du Y, Xu Y, Ding L, Yao H, Yu H, Zhou T, et al. Down-regulation of miR-141 in gastric cancer and its involvement in cell growth. J Gastroenterol. 2009;44(6):556–61.
Paterson EL, Kazenwadel J, Bert AG, Khew-Goodall Y, Ruszkiewicz A, Goodall GJ. Down-regulation of the miRNA-200 family at the invasive front of colorectal cancers with degraded basement membrane indicates EMT is involved in cancer progression. Neoplasia. 2013;15(2):180–91.
Braun J, Hoang-Vu C, Dralle H, Hüttelmaier S. Downregulation of microRNAs directs the EMT and invasive potential of anaplastic thyroid carcinomas. Oncogene. 2010;29(29):4237–44.
Wiklund ED, Bramsen JB, Hulf T, Dyrskjøt L, Ramanathan R, Hansen TB, et al. Coordinated epigenetic repression of the miR-200 family and miR-205 in invasive bladder cancer. Int J Cancer. 2011;128(6):1327–34.
Murakami Y, Yasuda T, Saigo K, Urashima T, Toyoda H, Okanoue T, et al. Comprehensive analysis of microRNA expression patterns in hepatocellular carcinoma and non-tumorous tissues. Oncogene. 2006;25:2537–45.
Jang K, Ahn H, Sim J, Han H, Abdul R, Paik SS, et al. Loss of microRNA-200a expression correlates with tumor progression in breast cancer. Transl Res. 2014;163(3):242–51.
Ye F, Tang H, Liu Q, Xie X, Wu M, Liu X, et al. MiR-200b as a prognostic factor in breast cancer targets multiple members of RAB family. J Transl Med. 2014;12:17.
Berber U, Yilmaz I, Narli G, Haholu A, Kucukodaci Z, Demirel D. MiR-205 and miR-200c: predictive micro RNAs for lymph node metastasis in triple negative breast cancer. J Breast Cancer. 2014;17(2):143–8.
Weidner N, Cady B, Goodson WH 3rd. Pathologic prognostic factors for patients with breast carcinoma. Which factors are important. Surg Oncol Clin N Am. 1997;6(3):415–62.
Vinh-Hung V, Cserni G, Burzykowski T. Effect of the number of uninvolved nodes on survival in early breast cancer. Oncol Rep. 2003;10(2):363–8.
Hilsenbeck SG, Ravdin PM, de Moor CA, Chamness GC, Osborne CK, Clark GM. Time-dependence of hazard ratios for prognostic factors in primary breast cancer. Breast Cancer Res Treat. 1998;52:227–37.
Truong PT, Vinh-Hung V, Cserni G, Woodward WA, Tai P, Vlastos G, et al. The number of positive nodes and the ratio of positive to excised nodes are significant predictors of survival in women with micrometastatic node-positive breast cancer. Eur J Cancer. 2008;44(12):1670–7.
Acknowledgments
This work was supported in part by the National Natural Science Foundation of China (Grant Number: 81172513, 81071028).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Feng Xu and Hua He contributed equally to this article.
Rights and permissions
About this article
Cite this article
Xu, F., He, H., Huang, W. et al. Decreased expression of MicroRNA-200 family in human breast cancer is associated with lymph node metastasis. Clin Transl Oncol 18, 283–288 (2016). https://doi.org/10.1007/s12094-015-1364-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1364-1