Abstract
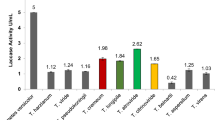
Qualitative screening of 295 fungi for laccases yielded 125 laccase positive ones, mostly basidiomycetes. Fifty of these were tested for laccase activity at pH 3.0, 4.5 and 6.0. Most showed maximum activity at pH 4.5, a few showed a broad activity range, two were optimal at pH 3.0 and only the mitosporic fungus Beltraniella sp. was best at pH 6. Most of the 25 fungi assayed at three different temperatures had an optimum at 45°C. The basidiomycete Auricularia sp. acted best at 30°C, while three others showed best activity at 60°C. This study shows the potential of screening diverse fungi for laccase with varying pH and temperature preferences for different applications.


Similar content being viewed by others
References
Baldrian P (2006) Fungal laccases-occurrence and properties. FEMS Microbiol Rev 30:215–242
Kunamneni A, Camarero S, Garcia-Burgos C, Plou F, Ballesteros A, Alcalde M (2008) Engineering and applications of fungal laccases for organic synthesis. Microb Cell Fact 7:32
Eriksson, K E L, Blanchette RA, Ander P (1990). Biodegradation of lignin. In: Timell T (ed) Microbial and enzymatic degradation of wood and wood components. Springer, Berlin, pp 225–334
D’Souza D, Tiwari R, Kumar Sah A, Raghukumar C (2006) Enhanced production of laccase by a marine fungus during treatment of colored effluents and synthetic dyes. Enzym Microb Technol 38:504–511
Niku-Paavola ML, Karhunen E, Salola P, Raunio V (1988) Ligninolytic enzymes of the white-rot fungus Phlebia radiata. Biochem J 254:877–884
Kiiskinen L, Ratto M, Kruus K (2004) Screening for novel laccase-producing microbes. J Appl Microbiol 97:640–646
Arora DS, Rampal P (2002) Laccase production by some Phlebia species. J Basic Microbiol 42:295–301
Shutova V, Revin V, Myakushina Yu (2008) The effect of copper ions on the production of laccase by the fungus Lentinus (Panus) tigrinus. Appl Biochem Microbiol 44(6):619–623
Ko EM, Leem YE, Choi HT (2001) Purification and characterization of laccase isozymes from the white-rot basidiomycete Ganoderma lucidum. Appl Microbiol Biotechnol 57(1–2):98–102
Maximo C, Pessoa Amorim M, Costa-Ferreira M (2003) Biotransformation of industrial reactive azo dyes by Geotrichum sp. CCMI 1019. Enzym Microb Technol 32(1):145–151
Bollag J, Leonowicz A (1984) Comparative studies of extracellular fungal laccases. Appl Environ Microbiol 48(4):849–854
Coll P, Fernandez-Abalos J, Villanueva J, Santamaria R, Perez P (1993) Purification and characterization of a phenoloxidase (laccase) from the lignin-degrading basidiomycete PM1 (CECT 2971). Appl Environ Microbiol 59(8):2607–2613
D’Souza-Ticlo D, Sharma D, Raghukumar C (2009) A thermostable metal-tolerant laccase with bioremediation potential from a marine-derived fungus. Mar Biotechnol 11(6):725–737
Perez J, Martinez J, Rubia T (1996) Purification and partial characterization of a laccase from the white rot fungus Phanerochaete flavido-alba. Appl Environ Microb 62(11):4263–4267
Acknowledgments
This work was funded by the Department of Biotechnology, Ministry of Science and Technology, Government of India, under its Small Business Innovation Research Initiative (SBIRI) Programme.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bodke, P.M., Senthilarasu, G. & Raghukumar, S. Screening Diverse Fungi for Laccases of Varying Properties. Indian J Microbiol 52, 247–250 (2012). https://doi.org/10.1007/s12088-011-0204-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-011-0204-4