Abstract
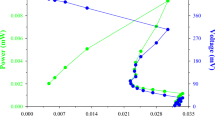
This study deals with production and characterization of biosurfactant from renewable resources by Pseudomonas aeruginosa. Biosurfactant production was carried out in 3L fermentor using waste motor lubricant oil and peanut oil cake. Maximum biomass (11.6 mg/ml) and biosurfactant production (8.6 mg/ml) occurred with peanut oil cake at 120 and 132 h respectively. Characterization of the biosurfactant revealed that, it is a lipopeptide with chemical composition of protein (50.2%) and lipid (49.8%). The biosurfactant (1 mg/ml) was able to emulsify waste motor lubricant oil, crude oil, peanut oil, kerosene, diesel, xylene, naphthalene and anthracene, comparatively the emulsification activity was higher than the activity found with Triton X-100 (1 mg/ml). Results obtained in the present study showed the possibility of biosurfactant production using renewable, relatively inexpensive and easily available resources. Emulsification activity found with the biosurfactant against different hydrocarbons showed its possible application in bioremediation of environments polluted with various hydrocarbons.



Similar content being viewed by others
References
Rosenberg E (1986) Microbial biosurfactants. Crit Rev Biotechnol 3:109–132
Mercade ME, Manresa MA (1994) The use of agroindustrial byproducts for biosurfactant production. J Am Oil Chem Soc 71:61–64
Makkar RS, Cameotra SS (1999) Biosurfactant production by microorganisms on unconventional carbon sources. J Surfactants Deterg 2:237–242
Makkar RS, Cameotra SS (1997) Utilization of molasses for biosurfactant production by two Bacillus strains at thermophilic conditions. J Am Oil Chem Soc 74:887–889
Sheppard JD, Mulligan CN (1987) The production of surfactin by Bacillus subtilis grown on peat hydrolysate. Appl Microbiol Biotechnol 27:110–116
Fox SL, Bala GA (2000) Production of surfactant from Bacillus subtilis ATCC 21332 using potato substrates. Bioresour Technol 75:235–240
Besson F, Michel G (1992) Biosynthesis of iturin and surfactin by Bacillus subtilis. Evidence for amino acid activating enzymes. Biotechnol Lett 14:1013–1018
Cooper DG, Goldenberg BG (1987) Surface-active agents from two Bacillus species. Appl Environ Microbiol 53:224–229
Buchanan RE, Gibbons NE, Cowan ST, Holt TG, Liston J, Murry RG, Niven CF, Ravin AW, RY Stainer (1974) Bergey’s manual of determinative bacteriology. Williams and Wilkins Co., Baltimore, 1246 p
Thavasi R (2006) Biosurfactants from marine hydrocarbonoclastic bacteria and their application in marine oil pollution abatement. Ph.D. Thesis, Annamalai University, Chidambaram, 162 p
Li Z-Y, Lang S, Wagner F, Witte L, Wary V (1984) Formation and identification of interfacial-active glycolipids from resting microbial cells. Appl Environ Microbiol 48:610–617
Rosenberg E, Zuckerberg A, Rubinovitz C, Gutnick DL (1979) Emulsifier of Arthrobacter RAG-I: isolation and emulsifying properties. Appl Environ Microbiol 37:402–408
Intergovernmental Oceanographic Commission Manuals and Guide No. 11 (1982) The determination of petroleum hydrocarbons in sediments. UNESCO, Paris, 38 p
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Folch JM, Lees M, Stanly HS (1956) A simple method for the isolation and quantification of total lipids from animal tissues. J Biol Chem 226:497–509
Mercade ME, Manresa MA, Robert M, Espuny MJ, de Andres C, Guinea J (1993) Olive oil mill effluent (OOME). New substrate for biosurfactant production. Bioresour Technol 43:1–6
Deshpande M, Daniels L (1995) Evaluation of sophorolipid biosurfactant production by Candida bombicola using animal fat. Bioresour Technol 54:143–150
Benincasa M, Contiero J, Manresa MA, Moreaes IO (2002) Rhamnolipid production by Pseudomonas aeruginosa LBI growing on soap stock as the sole carbon source. J Food Eng 54:283–288
Nitschke M, Pastore GM (2004) Biosurfactant production by Bacillus subtilis using cassava processing effluent. Appl Biochem Biotechnol 112:163–172
Haba E, Espuny MJ, Busquets M, Manresa A (2000) Screening and production of rhamnolipids by Pseudomonas aeruginosa NCBI 40044 from waste frying oils. J Appl Microbiol 88:379–387
Goldman S, Shabtai Y, Rubinovitz C, Rosenberg E, Gutnick DL (1982) Emulsan in Acinetobacter calcoaceticus RAG-I: distribution of cell-free and cell associated cross reacting materials. Appl Environ Microbiol 44:165–170
Lee K, Tremblay GH, Cobanli SE (1995) Bioremediation of oiled beach sediments: assessment of inorganic and organic fertilizers. In: Proceedings of the 1995 oil spill conference. American Petroleum Institute, Washington, pp 107–113
Bragg JR, Prince E, Harner J, Atlas RM (1994) Effectiveness of bioremediation for the Exxon Valdez oil spill. Nature (London) 368:413–418
Fayad NM, Overton E (1995) A unique biodegradation pattern of the oil spilled during the 1991. Gulf War Mar Pollut Bull 30:239–246
Marty P, Martin Y (1996) Seed and feed strategy against oil spill in a marine environment: laboratory and simulated outdoor experiments with selected natural bacterial strains. J Mar Biotechnol 4:155–158
Kim S-J, Sohn JH, Sim DS, Kwon KK, Kim TH (1998) The effects of bioremediation on the oil degradation in oil polluted environments. In: Le Gal Y, Halvorson HO (eds) New developments in marine biotechnology. Plenum Press, New York, pp 181–188
Atlas RM (1981) Microbial degradation of petroleum hydrocarbons: an environmental perspective. Microbiol Rev 45:180–209
Kuiper I, Lagendijk EL, Pickford R, Derrick JP, Lamers GEM, Thomas-Oates JE, Lugtenberg BJJ, Bloemberg GV (2004) Characterization of two Pseudomonas putida lipopeptide biosurfactants, putisolvin I and II. which inhibit biofilm formation and break down existing biofilms. Mol Microbiol 51:97–113
Kalinovskaya N, Kuznetsova T, Ya R, Yu M, Mil’grom E, Willis R, Wood A, Kurtz H, Carabedian C, Murphy P, Elyakov G (1995) Surfactin-like structures of five cyclic despsipeptises from the marine isolates of Bacillus pumilus Russ. Chem Bull (Engl Trans) 44:951
Acknowledgment
The authors are thankful to the authorities of Annamalai University for providing the facilities and Department of Ocean Development and Council of Scientific and Industrial Research, Government of India for their financial support to carry out the work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thavasi, R., Subramanyam Nambaru, V.R.M., Jayalakshmi, S. et al. Biosurfactant Production by Pseudomonas aeruginosa from Renewable Resources. Indian J Microbiol 51, 30–36 (2011). https://doi.org/10.1007/s12088-011-0076-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-011-0076-7