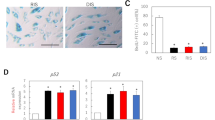
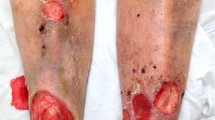
Abstract
Wound healing is a complex overlapping biological process that involves a sequence of events coordinated by various cells, proteins, growth factors, cytokines and signaling molecules. Recent evidence indicates that forkhead box O1 (FOXO1) transcription factors play an important role in organizing these events to stimulate wound healing. The ubiquitously expressed forkhead box, class O (FOXO) transcription factors act as cell signaling molecules in various transcriptional processes that are involved in diverse cellular activities, including cell death, cell differentiation, DNA repair, apoptosis, and oxidative stress in response to stimuli, and interact with numerous proteins. Due to the activation of FOXO targeted genes, FOXOs are involved in maintaining the balance between oxidative stress and antioxidants. In humans, different isoforms of FOXO namely FOXO1, FOXO3, FOXO4 and FOXO6 are present, however only FOXO1 and FOXO3 possess biological functions such as morphogenesis, maintenance and tissue regeneration. This might make FOXOs an important therapeutic target to enhance wound healing in diabetes, and to avoid over scarring. In spite of extensive literature, little is known regarding the role of FOXO and its relationship in wound healing. This review provides a summary of FOXO proteins and their biological role in wound healing and oxidative stress.





Similar content being viewed by others
References
Alblowi J, Kayal RA, Siqueria M (2009) High levels of tumor necrosis factor-α contribute to accelerated loss of cartilage in diabetic fracture healing. Am J Pathol 175:1574–1585
Alikhani M, Roy S, Graves DT (2010) FOXO1plays an essential role in apoptosis of retinal pericytes. Mol Vis 16:408–415
Anastasiou D, Poulogiannis G, Asara JM (2011) Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to cellular antioxidant responses. Science 334:1278–1283
Aoki M, Jiang H, Vogt PK (2004) Proteasomal degradation of the FoxO1 transcriptional regulator in cells transformed by the P3k and AKT oncoproteins. PNAS 101:13613–13617
Asmat U, Abad K, Ismail K (2016) Diabetes mellitus and oxidative stress-a concise review. Saudi Pharm J 24:547–553
Barthel A, Schmoll D, Unterman TG (2005) FOXO proteins in insulin action and metabolism. Trends Endocrinol Metab 16:183–189
Behl Y, Krothapalli P, Dessta T, Roy S, Graves DT (2009) FOXO1 plays an important role in enhanced microvascular cell apoptosis and microvascular cell loss in type 1 and type 2 diabetic rats. Diabetes 58:917–925
Birben E, Murat US, Sackesen C, Erzurum S, Kalayci O (2012) Oxidative stress and antioxidant defense. WAO Journal 5:9–19
Brown J, Wang H, Suttles J, Graves DT, Martin M (2011) Mammalian target of rapamycin complex 2 (mTORC2) negatively regulates toll-like receptor 4-mediated inflammatory response via FoxO1. The J Biol Chem 286(52):44295–44305
Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303:2011–2015
Calnan DR, Brunet A (2008) The FoxO code. Oncogene 27(16):2276–2288
Chong ZZ, Li F, Maiese K (2005) Oxidative stress in the brain: novel cellular targets that govern survival during neurodegenerative disease. Prog Neurobiol 75:207–246
Chong ZZ, Maiese K (2007) The Src homology 2 domain tyrosine phosphatases SHP-1 and SHP-2: diversified control of cell growth, inflammation, and injury. Histol Histopathol 22:1251–1267
Daitoku H, Sakamaki JI, Fukamizu A (2011) Regulation of FOXO transcription factors by acetylation and protein-protein interactions. Biochim Biophy Acta 1813:1954–1960
Dansen TB, Smits LMM, van Triest MH (2009) Redoxsensitive cysteines bridge p300/CBP-mediated acetylation and FoxO4 activity. Nat Chem Biol 5(9):664–672
Dharaneeswaran H, Ruhul A, Yuan L et al (2014) FOXO1-mediated activation of AKT plays a critical role in vascular homeostasis. Circ Res 115(2):238–251
Dijkers PF, Medema RH, Lammers JW, Koenderman L, Coffer PJ (2001) Expression of the pro-apoptotic Bcl-2 family member Bim is regulated by the forkhead transcription factor FKHR-L1. Curr Biol 10:1201–1204
Eijkelenboom A, Burgering BM (2013) FOXOs: signalling integrators for homeostasis maintenance. Nat Rev Mol Cell Biol 14:83–97
Eming SA, Martin P, Tomic-Canic M (2014) Wound repair and regeneration: mechanisms. Signaling and translation. Sci Transl Med 6:265–274
Engelman JA (2009) Targeting PI3K signaling in cancer: opportunities, challenges and limitations. Nat Reviews Can 9(8):550–562
Eric WF, Brosens JJ, Gomes AR, Koo CY (2013) Forkhead box proteins: tuning forks for transcriptional harmony. Nat Rew Can 13:482–495
Essers MA, Weijzen S, de Vries-Smits AM, Saarloos I, de Ruiter ND, Bos JL (2004) FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J 23:4802–4812
Giannakou ME, Partridge L (2004) The interaction between FOXO and SIRT1: tipping the balance towards survival. Trends Cell Biol 14:408–412
Gross DN, Wan M, Birnbaum MJ (2009) The role of FOXO in the regulation of metabolism. Curr Diab Rep 9:208–214
Guo S, Dipietro LA (2010) Factors affecting wound healing. J Dent Res 89:219–229
Hakuno F, Yamauchi Y, Kaneko G, Yoneyama Y (2011) Constitutive expression of insulin receptor substrate (IRS)-1 inhibits myogenic differentiation through nuclear exclusion of FOXO1 in L6 myoblasts. PLoS One 6:e25655
Hameedaldeen A, Liu J, Batres A, Graves GS, Graves DT (2014) FOXO1, TGF-beta regulation and wound healing. Int J Mol Sci 15:16257–16269
Ho KK, Myatt SS, Lam EW (2008) Many forks in the path: cycling with FOXO. Oncogene 27:2300–2311
Hosaka T, Biggs WH, Tieu D, Boyer AD, Varki NM, Cavenee WK, Arden KC (2004) Disruption of forkhead transcription factor (FOXO) family members in mice reveals their functional diversification. Proc Natl Acad Sci U S A 101:2975–2980
Hsu CP, Zhai P, Yamamoto T et al (2010) Silent information regulator 1 protects the heart from ischemia/reperfusion. Circulation 122(21):2170–2182
Huang H, Regan KM, Lou Z, Chen J, Tindall DJ (2006) CDK2-dependent phophorolation of FOXO1 as an apoptotic response to DNA damage. Science 314:294–297
Huang H, Tindall DJ (2007) Dynamic FOXO transcription factors. J Cell Sci 120:2479–2487
Jacobs FM, van der Heide LP, Wijchers JP, Burbach M, Hoekman MF, Smidt S (2003) FOXO6, a novel member of the FOXO class of transcription factors with distinct shuttling dynamic. J Biol Chem 278:35959–35967
Kayal RA, Siqueira M, Alblowi J et al (2010) TNF-a mediates diabetes-enhanced chondrocyte apoptosis during fracture healing and stimulates chondrocyte apoptosis through FOXO1. J Bone and Min Res 25:1604–1615
Kim DH, Perdomo G, Zhang T (2011) FoxO6 integrates insulin signaling with gluconeogenesis in the liver. Diabetes 60(11):2763–2774
Kobayashi Y, Furukawa-Hibi Y, Chen C (2005) SIRT1 is critical regulator of FOXO-mediated transcription in response to oxidative stress. Int J Mol Med 16(2):237–243
Kortylewski M, Feld F, Kruger KD, Bahrenberg G, Roth RA, Joost HG, Heinrich PC, Behrmann I, Barthel A (2003) Akt modulates STAT3-mediated gene expression through a FKHR (FOXO1a)-dependent mechanism. JBiol Chem 278:5242–5249
Lalmansingh AS, Karmakar S, Jin Y (2012a) Multiple modes of chromatin remodeling by Forkhead box proteins. Biochim Biophys Acta 181:707–715
Lalmansingh AS, Karmakar S, Jin Y, Nagaich AK (2012b) Multiple modes of chromatin remodeling by Forkhead box proteins. Biochim Biophys Acta: Gene Regul Mech 1819(7):707–715
Lam EW, Brosens JJ, Gomes AR (2013) Forkhead box proteins: tuning forks for transcriptional harmony. Nat Rev Cancer 13:482–495
Lee D, Goldberg AL (2013) SIRT1 protein, by blocking the activities of transcription factors FOXO1 and FOXO3, inhibits muscle atrophy and promotes muscle growth. J Biol Chem 288:30515–30526
Lehtinen MK, Yuan Z, Boag PR, Yang Y, Villen J, Becker E (2006) A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell 125:987–1001
Lima MH, Caricilli AM, deAbreu LL, Araujo EP, Pelegrinelli FF, Thirone AC, Tsukumo DM, Pessoa AF, dos Santos MF, de Moraes MA, Carvalheira JB, Velloso LA, Saad MJ (2012) Topical insulin accelerates wound healing in diabetes by enhancing the AKT and ERK pathways: a double-blind placebo-controlled clinical trial. PLoS One 7:e36974
Lin K, Chen L, Abdel Khalek W, Ward JL, Yang H, Chabi B, Wrutniak-Cabello C, Tong Q (2014) Regulation of skeletal muscle oxidative capacity and muscle mass by SIRT3. PLoS One 9:e85636
Lu Q, Zhai Y, Cheng Q (2013) The Akt-FOXO3a-manganese superoxide dismutase pathway is involved in the regulation of oxidative stress in diabetic nephropathy. Exp Physiol 98:934–945
Rached M-T, Kode A, Xu L et al (2010) FoxO1 is a positive regulator of bone formation by favoring protein synthesis and resistance to oxidative stress in osteoblasts. Cell Metab 11(2):147–160
Maiese K (2015) FOXO proteins in the nervous system. Anal Cell Pathol 56
Maiese K, Hou J, Chong ZZ, Shang YC (2009) Erythropoietin, Forkhead proteins and oxidative injury: biomarkers and biology. Sci World J 9:1072–1104
Maiese K, Morhan SD, Chong ZZ (2007) Oxidative stress biology and cell injury during type 1 and type 2 diabetes mellitus. Curr Neurovasc Res 4:63–71
Martins VL, Caley M, O’Toole EA (2013) Matrix metalloproteinases and epidermal wound repair. Cell Tissue Res 351:255–268
Milan G, Romanello V, Pescatore F, Armani A, Paik JH (2015) Regulation of autophagy and the ubiquitin-proteasome system by the FOXO transcriptional network during muscle atrophy. Nat Commun 6:6670–6683
Mori R, Tanaka K, Kerckhove M, Kashiyama MO (2014) Reduced FOXO1 expression accelerates skin wound healing and attenuates scarring. Am J Pathol 184:2465–2479
OS I, Zhang W, Wasserman DH, Liew CW, Liu J, Paik J (2015) FOXO1 integrates direct and indirect effects of insulin on hepatic glucose production and glucose utilization. Nature Comm 6:70–79
Paap RH, Oosterbroek S, Wagemans CM, Oerthel LV et al (2016) FOXO6 affects Plxna4 mediated neuronal migration during mouse cortical development. PNAS 113(45):23–32
Papanicolaou KN, Izumiya Y, Walsh K (2008) Forkhead transcription factor and cardiovascular biology. Circ Res 102:16–31
Ponugoti B, Dong G, Graves DT (2012) Role of forkhead transcription factors in diabetes-induced oxidative stress. Exp Diabetes Res 51:93–97
Ponugoti B, Xu F, Zhang C, Tian C, Pacios S, Graves DT (2013) FOXO1 promotes wound healing through the up-regulation of TGF-beta1 and prevention of oxidative stress. J Cell Biol 203:327–343
Putker M, Madl T, Vos HR (2013) Redox-dependent control of FOXO/DAF-16 by transportin-1. Mol Cell 49(4):730–742
Ramaswamy S, Nakamura N, Sansal I, Bergeron L, Sellers WR (2002) A novel mechanism of gene regulation and tumor suppression by the transcription factor FKHR. Cancer Cell 2(1):81–91.
Renault VM, Rafalski VA, Morgan AA (2009) FOXO3 regulates neural stem cell homeostasis. Cell Stem Cell 5:527–539
Roupe KM, Alberius P, Schmidtchen A, Sorensen OE (2010) Gene expression demonstrates increased resilience toward harmful inflammatory stimuli in the proliferating epidermis of human skin wounds. Exp Dermatol 9:e329–e332
Roupe S, Veerla A, Olson J, Stone EL, Sorensen OE, Hedrick SM, Nizet V (2014) Transcription factor binding site analysis identifies FOXO transcription factors as regulators of the cutaneous wound healing process. PLoS One 9:e89274
Salathia NS, Shi J, Zhang J, Glynne RJ (2013) An in vivo screen of secreted proteins identifies adiponectin as a regulator of murine cutaneous wound healing. J Invest Dermatol 133:812–821
Salih DA, Rashid AJ, Colas D (2012) FOXO6 regulates memory consolidation and synaptic function. Gen Devel 26:2780–2801
Sangodkar J, Dhawan NS, Melville H, Singh VJ, Yuan E, Rana H (2012) Targeting the FOXO1/KLF6 axis regulates EGFR signaling and treatment response. J Clin Invest 122:2637–2651
Schafer M, Werner S (2008) Oxidative stress in normal and impaired wound repair, Pharmacol. Res 58(2):165–171. https://doi.org/10.1016/j.phrs.2008.06.004
Serravallo M, Jagdeo J, Glick SA, Siegel DM, Brody NL (2013) Sirtuins in dermatology: applications for future research and therapeutics. Arch Dermatol Res 305:269–282
Shaklai G, Shefer N, Stern K (2015) Glucose-dependent FOXO1 switch in healing wounds: a shred of hope for diabetic ulcers? Diabetes 64:6–8
Shaw TS, Martin P (2009) Wound repair at a glance. J Cell Sci 122:3209–3213
Shibata S, Tada Y, Asano Y, Hau CS, Kato T, Saeki H, Yamauchi T, Kubota N, Kadowaki T, Sato S (2012) Adiponectin regulates cutaneous wound healing by promoting keratinocyte proliferation and migration via the ERK signaling pathway. J Immunol 189:3231–3241
Siqueira MF, Li J, Chehab L, Desta T, Chino T, Krothpali N, Behl Y, Alikhani M, Yang J, Braasch C, Graves DT (2010) Impaired wound healing in mouse models of diabetes is mediated by TNF-alpha dysregulation and associated with enhanced activation of forkhead box O1 (FOXO1). Diabetologia 53:378–388
Slomka M, Zieminska E, Lazarewicz J (2008) Nicotinamide and 1-methylnicotinamide reduce homocysteine neurotoxicity in primary cultures of rat cerebellar granule cells. Acta Neurobiol Exp 68:1–9
Stadeli R, Hoffmans R, Basler K (2006) Transcription under the ¨ control of nuclear arm/훽-catenin. Curr Biol 16(10):378–385
Storz P (2011) Forkhead homeobox type O transcription factors in the responses to oxidative stress. Antioxid Redox Signal 14:593–605
Tang ED, Nunez G, Barr FG, Guan KL (1999) Negative regulation of the forkhead transcription factor FKHR by Akt. J Biol Chem 274:16741–16746
Tikhanovich I, Cox J, Weinman SA (2013) Forkhead box class O transcription factors in liver function and disease. J Gastroenterol Hepatol 28:125–131
Urbanek P, Klotz LO (2016) Posttranscriptional regulation of FOXO expression: microRNAs and beyond. Br J Pharmacol. https://doi.org/10.1111/bph.13471
Van Der Vos KE, Coffer PJ (2011) The extending network of FOXO transcriptional target genes. Antioxid Redox Signal 14:579–592
Wang MC, Bohmann D, Jasper H (2005) JNK extends life span and limits growth by antagonizing cellular and organism wide responses to insulin signaling. Cell 121:115–125
Wang Y, Zhou Y, Graves DT (2014) FOXO transcription factors: their clinical significance and regulation. BioMed res. Int. 1-13
Wolfgang L, Fernandex-Marcos PJ (2017) FOXO transcription factors at the interface of metabolism and cancer. Int J Can 15:2379–2391
Woods YL, Rena G, Morrice N, Barthel A, Becker W, Guo S, Unterman TG, Cohen P (2001) The kinase DYRK1A phosphorylates the transcription factor FKHR at Ser329 in vitro a novel in vivo phosphorylation site. Biochem J 355:597–607
Xie Q, Chen J, Yuan Z (2012) Post-translational regulation of FOXO. Acta Biochim Biophys Sin 44(11):897–901
Xu F, Othman B, Lim J, Batres A, Ponugoti B, Zhang C, Yi L, Liu J, Tian C (2015) FOXO1 inhibits diabetic mucosal wound healing but enhances healing of normoglycemic wounds. Diabetes 64:243–256
Yamamura Y, Wei LL, Inoue KI, Ida H, Ito Y (2006) RUNX3 cooperates with FoxO3a to induce apoptosis in gastric cancer cells. J Biol Chem 281(8):5267–5276
Zhang C, Ponugoti B, Tian C, Xu F, Tarapore R, Batres A, Alsadun S, Lim J, Dong G, Graves DT (2015) FOXO1 differentially regulates both normal and diabetic wound healing. J Cell Biol 209:289–303
Zou ZQ, Xu J, Li L, Han YS (2015) Down-regulation of SENCR promotes smooth muscle cells proliferation and migration in db/db mice through up-regulation of FOXO1 and TRPC6. Biomed Pharmacother 74:35–41
Acknowledgements
This work was supported by the South African Research Chairs Initiative of the Department of Science and Technology and National Research Foundation of South Africa (Grant No 98337); the University of Johannesburg; the CSIR National Laser Centre Laser Rental Pool Program; and the National Research Foundation of South Africa. Sathish Sundar Dhilip Kumar is supported by funding from the Claude Leon Foundation, South Africa. Funding sources had no involvement in study design, data collection, analysis and interpretation, writing of the report, and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors confirm that this article content has no conflict of interest.
Rights and permissions
About this article
Cite this article
Rajendran, N.K., Dhilip Kumar, S.S., Houreld, N.N. et al. Understanding the perspectives of forkhead transcription factors in delayed wound healing. J. Cell Commun. Signal. 13, 151–162 (2019). https://doi.org/10.1007/s12079-018-0484-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-018-0484-0