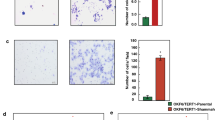
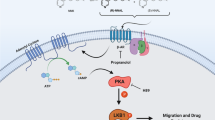
Abstract
Smoking is the leading cause of preventable death worldwide. Though cigarette smoke is an established cause of head and neck cancer (including oral cancer), molecular alterations associated with chronic cigarette smoke exposure are poorly studied. To understand the signaling alterations induced by chronic exposure to cigarette smoke, we developed a cell line model by exposing normal oral keratinocytes to cigarette smoke for a period of 12 months. Chronic exposure to cigarette smoke resulted in increased cellular proliferation and invasive ability of oral keratinocytes. Proteomic and phosphoproteomic analyses showed dysregulation of several proteins involved in cellular movement and cytoskeletal reorganization in smoke exposed cells. We observed overexpression and hyperphosphorylation of protein kinase N2 (PKN2) in smoke exposed cells as well as in a panel of head and neck cancer cell lines established from smokers. Silencing of PKN2 resulted in decreased colony formation, invasion and migration in both smoke exposed cells and head and neck cancer cell lines. Our results indicate that PKN2 plays an important role in oncogenic transformation of oral keratinocytes in response to cigarette smoke. The current study provides evidence that PKN2 can act as a potential therapeutic target in head and neck squamous cell carcinoma, especially in patients with a history of smoking.





Similar content being viewed by others
Abbreviations
- AGC:
-
Automatic Gain Control
- BCA:
-
Bicinchoninic acid
- EDTA:
-
Ethylenediaminetetraacetic acid
- EMT:
-
Epithelial–mesenchymal transition
- HCD:
-
Higher energy collisional dissociation
- HPLC:
-
High Pressure Liquid Chromatography
- SDS:
-
Sodium Dodecyl Sulfate
- TEABC:
-
Triethyl ammonium bicarbonate
- TMT:
-
Tandem Mass Tag
References
Amano M, Nakayama M, Kaibuchi K (2010) Rho-kinase/ROCK: a key regulator of the cytoskeleton and cell polarity. Cytoskeleton (Hoboken) 67:545–554
Chang SS, Jiang WW, Smith I, Glazer C, Sun WY, Mithani S, Califano JA (2010) Chronic cigarette smoke extract treatment selects for apoptotic dysfunction and mitochondrial mutations in minimally transformed oral keratinocytes. Int J Cancer 126:19–27
Chang X, Ravi R, Pham V, Bedi A, Chatterjee A, Sidransky D (2011) Adenylate kinase 3 sensitizes cells to cigarette smoke condensate vapor induced cisplatin resistance. PLoS One 6:e20806
Chatterjee A, Mambo E, Zhang Y, Deweese T, Sidransky D (2006) Targeting of mutant hogg1 in mammalian mitochondria and nucleus: effect on cellular survival upon oxidative stress. BMC Cancer 6:235
Chen J, Yan Y, Li J, Ma Q, Stoner GD, Ye J, Huang C (2005) Differential requirement of signal pathways for benzo[a]pyrene (B[a]P)-induced nitric oxide synthase (iNOS) in rat esophageal epithelial cells. Carcinogenesis 26:1035–1043
Chen RJ, Chang LW, Lin P, Wang YJ (2011) Epigenetic effects and molecular mechanisms of tumorigenesis induced by cigarette smoke: an overview. J Oncol 2011:654931
Cormier N, Yeo A, Fiorentino E, Paxson J (2015) Optimization of the wound scratch assay to detect changes in murine mesenchymal stromal cell migration after damage by soluble cigarette smoke extract. J Vis Exp e53414
Currier N, Solomon SE, Demicco EG, Chang DL, Farago M, Ying H, Dominguez I, Sonenshein GE, Cardiff RD, Xiao ZX, Sherr DH, Seldin DC (2005) Oncogenic signaling pathways activated in DMBA-induced mouse mammary tumors. Toxicol Pathol 33:726–737
Dasgupta P, Rizwani W, Pillai S, Kinkade R, Kovacs M, Rastogi S, Banerjee S, Carless M, Kim E, Coppola D, Haura E, Chellappan S (2009) Nicotine induces cell proliferation, invasion and epithelial-mesenchymal transition in a variety of human cancer cell lines. Int J Cancer 124:36–45
Dong Y, Zhao Q, Ma X, Ma G, Liu C, Chen Z, Yu L, Liu X, Zhang Y, Shao S, Xiao J, Li J, Zhang W, Fu M, Dong L, Yang X, Guo X, Xue L, Fang F, Zhan Q, Zhang L (2015) Establishment of a new OSCC cell line derived from OLK and identification of malignant transformation-related proteins by differential proteomics approach. Sci Rep 5:12668
Dusek RL, Attardi LD (2011) Desmosomes: new perpetrators in tumour suppression. Nat Rev Cancer 11:317–323
Ezzati M, Henley SJ, Lopez AD, Thun MJ (2005) Role of smoking in global and regional cancer epidemiology: current patterns and data needs. Int J Cancer 116:963–971
Galoian K, Qureshi A, Wideroff G, Temple HT (2015) Restoration of desmosomal junction protein expression and inhibition of H3K9-specific histone demethylase activity by cytostatic proline-rich polypeptide-1 leads to suppression of tumorigenic potential in human chondrosarcoma cells. Mol Clin Oncol 3:171–178
Goel R, Muthusamy B, Pandey A, Prasad TS (2011) Human protein reference database and human proteinpedia as discovery resources for molecular biotechnology. Mol Biotechnol 48:87–95
Govindan R, Ding L, Griffith M, Subramanian J, Dees ND, Kanchi KL, Maher CA, Fulton R, Fulton L, Wallis J, Chen K, Walker J, McDonald S, Bose R, Ornitz D, Xiong D, You M, Dooling DJ, Watson M, Mardis ER, Wilson RK (2012) Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell 150:1121–1134
Gumus ZH, Du B, Kacker A, Boyle JO, Bocker JM, Mukherjee P, Subbaramaiah K, Dannenberg AJ, Weinstein H (2008) Effects of tobacco smoke on gene expression and cellular pathways in a cellular model of oral leukoplakia. Cancer Prev Res (Phila) 1:100–111
Hardonniere K, Saunier E, Lemarie A, Fernier M, Gallais I, Helies-Toussaint C, Mograbi B, Antonio S, Benit P, Rustin P, Janin M, Habarou F, Ottolenghi C, Lavault MT, Benelli C, Sergent O, Huc L, Bortoli S, Lagadic-Gossmann D (2016) The environmental carcinogen benzo[a]pyrene induces a Warburg-like metabolic reprogramming dependent on NHE1 and associated with cell survival. Sci Rep 6:30776
Hecht SS (1998) Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem Res Toxicol 11:559–603
Johnson JL, Najor NA, Green KJ (2014) Desmosomes: regulators of cellular signaling and adhesion in epidermal health and disease. Cold Spring Harb Perspect Med 4:a015297
Judd NP, Winkler AE, Murillo-Sauca O, Brotman JJ, Law JH, Lewis JS Jr, Dunn GP, Bui JD, Sunwoo JB, Uppaluri R (2012) ERK1/2 regulation of CD44 modulates oral cancer aggressiveness. Cancer Res 72:365–374
Julian L, Olson MF (2014) Rho-associated coiled-coil containing kinases (ROCK): structure, regulation, and functions. Small GTPases 5:e29846
Kelley LC, Hayes KE, Ammer AG, Martin KH, Weed SA (2010) Cortactin phosphorylated by ERK1/2 localizes to sites of dynamic actin regulation and is required for carcinoma lamellipodia persistence. PLoS One 5:e13847
Kim MS, Huang Y, Lee J, Zhong X, Jiang WW, Ratovitski EA, Sidransky D (2010) Cellular transformation by cigarette smoke extract involves alteration of glycolysis and mitochondrial function in esophageal epithelial cells. Int J Cancer 127:269–281
Kim MS, Pinto SM, Getnet D, Nirujogi RS, Manda SS, Chaerkady R, Madugundu AK, Kelkar DS, Isserlin R, Jain S, Thomas JK, Muthusamy B, Leal-Rojas P, Kumar P, Sahasrabuddhe NA, Balakrishnan L, Advani J, George B, Renuse S, Selvan LD, Patil AH, Nanjappa V, Radhakrishnan A, Prasad S, Subbannayya T, Raju R, Kumar M, Sreenivasamurthy SK, Marimuthu A, Sathe GJ, Chavan S, Datta KK, Subbannayya Y, Sahu A, Yelamanchi SD, Jayaram S, Rajagopalan P, Sharma J, Murthy KR, Syed N, Goel R, Khan AA, Ahmad S, Dey G, Mudgal K, Chatterjee A, Huang TC, Zhong J, Wu X, Shaw PG, Freed D, Zahari MS, Mukherjee KK, Shankar S, Mahadevan A, Lam H, Mitchell CJ, Shankar SK, Satishchandra P, Schroeder JT, Sirdeshmukh R, Maitra A, Leach SD, Drake CG, Halushka MK, Prasad TS, Hruban RH, Kerr CL, Bader GD, Iacobuzio-Donahue CA, Gowda H, Pandey A (2014) A draft map of the human proteome. Nature 509:575–581
Lachmann S, Jevons A, De Rycker M, Casamassima A, Radtke S, Collazos A, Parker PJ (2011) Regulatory domain selectivity in the cell-type specific PKN-dependence of cell migration. PLoS One 6:e21732
Nanjappa V, Renuse S, Sathe GJ, Raja R, Syed N, Radhakrishnan A, Subbannayya T, Patil A, Marimuthu A, Sahasrabuddhe NA, Guerrero-Preston R, Somani BL, Nair B, Kundu GC, Prasad TK, Califano JA, Gowda H, Sidransky D, Pandey A, Chatterjee A (2015) Chronic exposure to chewing tobacco selects for overexpression of stearoyl-CoA desaturase in normal oral keratinocytes. Cancer Biol Ther 16:1593–1603
Olivera DS, Boggs SE, Beenhouwer C, Aden J, Knall C (2007) Cellular mechanisms of mainstream cigarette smoke-induced lung epithelial tight junction permeability changes in vitro. Inhal Toxicol 19:13–22
Parri M, Chiarugi P (2010) Rac and rho GTPases in cancer cell motility control. Cell Commun Signal 8:23
Pfeifer GP, Denissenko MF, Olivier M, Tretyakova N, Hecht SS, Hainaut P (2002) Tobacco smoke carcinogens, DNA damage and p53 mutations in smoking-associated cancers. Oncogene 21:7435–7451
Radhakrishnan A, Nanjappa V, Raja R, Sathe G, Chavan S, Nirujogi RS, Patil AH, Solanki H, Renuse S, Sahasrabuddhe NA, Mathur PP, Prasad TS, Kumar P, Califano JA, Sidransky D, Pandey A, Gowda H, Chatterjee A (2016) Dysregulation of splicing proteins in head and neck squamous cell carcinoma. Cancer Biol Ther 17:219–229
Raja R, Sahasrabuddhe NA, Radhakrishnan A, Syed N, Solanki HS, Puttamallesh VN, Balaji SA, Nanjappa V, Datta KK, Babu N, Renuse S, Patil AH, Izumchenko E, Prasad TS, Chang X, Rangarajan A, Sidransky D, Pandey A, Gowda H, Chatterjee A (2016) Chronic exposure to cigarette smoke leads to activation of p21 (RAC1)-activated kinase 6 (PAK6) in non-small cell lung cancer cells. Oncotarget 7:61229–61245
Rorth P (2009) Collective cell migration. Annu Rev Cell Dev Biol 25:407–429
Rosette C, Roth RB, Oeth P, Braun A, Kammerer S, Ekblom J, Denissenko MF (2005) Role of ICAM1 in invasion of human breast cancer cells. Carcinogenesis 26:943–950
Selvan LD, Renuse S, Kaviyil JE, Sharma J, Pinto SM, Yelamanchi SD, Puttamallesh VN, Ravikumar R, Pandey A, Prasad TS, Harsha HC (2014) Phosphoproteome of Cryptococcus neoformans. J Proteome 97:287–295
Spira A, Beane J, Shah V, Liu G, Schembri F, Yang X, Palma J, Brody JS (2004) Effects of cigarette smoke on the human airway epithelial cell transcriptome. Proc Natl Acad Sci U S A 101:10143–10148
Sun W, Chang SS, Fu Y, Liu Y, Califano JA (2011) Chronic CSE treatment induces the growth of normal oral keratinocytes via PDK2 upregulation, increased glycolysis and HIF1alpha stabilization. PLoS One 6:e16207
Tegtmeyer N, Wittelsberger R, Hartig R, Wessler S, Martinez-Quiles N, Backert S (2011) Serine phosphorylation of cortactin controls focal adhesion kinase activity and cell scattering induced by helicobacter pylori. Cell Host Microbe 9:520–531
Vaz M, Hwang SY, Kagiampakis I, Phallen J, Patil A, O'Hagan HM, Murphy L, Zahnow CA, Gabrielson E, Velculescu VE, Easwaran HP, Baylin SB (2017) Chronic cigarette smoke-induced epigenomic changes precede sensitization of bronchial epithelial cells to single-step transformation by KRAS mutations. Cancer Cell 32(360–376):e366
Vigil D, Kim TY, Plachco A, Garton AJ, Castaldo L, Pachter JA, Dong H, Chen X, Tokar B, Campbell SL, Der CJ (2012) ROCK1 and ROCK2 are required for non-small cell lung cancer anchorage-independent growth and invasion. Cancer Res 72:5338–5347
Villa-Moruzzi E (2007) Targeting of FAK Ser910 by ERK5 and PP1delta in non-stimulated and phorbol ester-stimulated cells. Biochem J 408:7–18
Vincent S, Settleman J (1997) The PRK2 kinase is a potential effector target of both rho and Rac GTPases and regulates actin cytoskeletal organization. Mol Cell Biol 17:2247–2256
Vizcaino JA, Cote RG, Csordas A, Dianes JA, Fabregat A, Foster JM, Griss J, Alpi E, Birim M, Contell J, O'Kelly G, Schoenegger A, Ovelleiro D, Perez-Riverol Y, Reisinger F, Rios D, Wang R, Hermjakob H (2013) The PRoteomics IDEntifications (PRIDE) database and associated tools: status in 2013. Nucleic Acids Res 41:D1063–D1069
Walker JM (1994) The bicinchoninic acid (BCA) assay for protein quantitation. Methods Mol Biol 32:5–8
Wang Y, Wu J, Wang Z (2006) Akt binds to and phosphorylates phospholipase C-gamma1 in response to epidermal growth factor. Mol Biol Cell 17:2267–2277
Yang S, Long M, Tachado SD, Seng S (2015) Cigarette smoke modulates PC3 prostate cancer cell migration by altering adhesion molecules and the extracellular matrix. Mol Med Rep 12:6990–6996
Yuan X, Zhang X, Zhang W, Liang W, Zhang P, Shi H, Zhang B, Shao M, Yan Y, Qian H, Xu W (2016) SALL4 promotes gastric cancer progression through activating CD44 expression. Oncogene 5:e268
Zhang L, Gallup M, Zlock L, Finkbeiner WE, McNamara NA (2013) Rac1 and Cdc42 differentially modulate cigarette smoke-induced airway cell migration through p120-catenin-dependent and -independent pathways. Am J Pathol 182:1986–1995
Acknowledgements
We thank the Department of Biotechnology (DBT), Government of India for research support to the Institute of Bioinformatics (IOB), Bangalore. Pavithra Rajagopalan is a recipient of Senior Research Fellowship from the Council of Scientific and Industrial Research (CSIR), New Delhi, India. Kiran K. Mangalaparthi is a recipient of Senior Research Fellowship from University Grants Commission (UGC), New Delhi, India. Harsha Gowda is a Wellcome Trust/DBT India Alliance Intermediate Career Fellow. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
AC and HG participated in study conception and study design. PR and VN were involved in cell culture and performed all assays and experiments. APJ and KKM carried out fractionation and mass spectrometric analysis of samples. PR and KP prepared the manuscript and manuscript figures. PR, KP and AHP were involved in data analyses and interpretation. AC, HG, DS, JAC, PPM, TSKP and BN edited, critically read and revised the manuscript. All the authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that no competing financial interests exist.
Electronic supplementary material
ESM 1
(PDF 6532 kb)
ESM 2
(PDF 6473 kb)
Supplementary Figure 1
Schematic workflow employed to study the proteomic and phosphoproteomic alterations in normal oral keratinocytes chronically exposed to cigarette smoke (TIFF 1988 kb)
Rights and permissions
About this article
Cite this article
Rajagopalan, P., Nanjappa, V., Patel, K. et al. Role of protein kinase N2 (PKN2) in cigarette smoke-mediated oncogenic transformation of oral cells. J. Cell Commun. Signal. 12, 709–721 (2018). https://doi.org/10.1007/s12079-017-0442-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-017-0442-2