Abstract
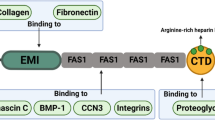
Integrity of the extracellular matrix (ECM) is essential for maintaining the normal structure and function of connective tissues. ECM is secreted locally by cells and organized into a complex meshwork providing physical support to cells, tissues, and organs. Initially thought to act only as a scaffold, the ECM is now known to provide a myriad of signals to cells regulating all aspects of their phenotype from morphology to differentiation. Matricellular proteins are a class of ECM related molecules defined through their ability to modulate cell–matrix interactions. Matricellular proteins are expressed at high levels during development, but typically only appear in postnatal tissue in wound repair or disease, where their levels increase substantially. Members of the CCN family, tenascin-C, osteopontin, secreted protein acidic rich in cysteine (SPARC), bone sialoprotein, thrombospondins, and galectins have all been classed as matricellular proteins. Periostin, a 90 kDa secreted homophilic cell adhesion protein, was recently added to matricellular class of proteins based on its expression pattern and function during development as well as in wound repair. Periostin is expressed in connective tissues including the periodontal ligament, tendons, skin and bone, and is also prominent in neoplastic tissues, cardiovascular disease, as well as in connective tissue wound repair. This review will focus on the functional role of periostin in tissue physiology. Fundamentally, it appears that periostin influences cell behaviour as well as collagen fibrillogenesis, and therefore exerts control over the structural and functional properties of connective tissues in both health and disease. Periostin is a novel matricellular protein with close homology to Drosophila fasciclin 1. In this review, the functional role of periostin is discussed in the context of connective tissue physiology, in development, disease, and wound repair.




Similar content being viewed by others
Abbreviations
- ECM:
-
Extracellular matrix
- CCN2:
-
connective tissue growth factor
- FA:
-
focal adhesion
- HGF:
-
human gingival fibroblast
- OSF-2:
-
osteoblast specific factor-2
- RCO:
-
rat calvarial osteoblast
- SPARC:
-
secreted protein acidic rich in cysteine
- TSP:
-
thrombospondin
References
Abraham PA, Perejda AJ, Carnes WH, Uitto J (1982) Marfan syndrome. Demonstration of abnormal elastin in aorta. J Clin Invest 70:1245–1252 doi:10.1172/JCI110723
Alford AI, Hankenson KD (2006) Matricellular proteins: extracellular modulators of bone development, remodeling, and regeneration. Bone 38:749–757 doi:10.1016/j.bone.2005.11.017
Baril P, Gangeswaran R, Mahon PC, Caulee K, Kocher HM, Harada T et al (2007) Periostin promotes invasiveness and resistance of pancreatic cancer cells to hypoxia-induced cell death: role of the beta4 integrin and the PI3k pathway. Oncogene 26:2082–2094 doi:10.1038/sj.onc.1210009
Berk BC, Fujiwara K, Lehoux S (2007) ECM remodeling in hypertensive heart disease. J Clin Invest 117:568–575 doi:10.1172/JCI31044
Berrier AL, Yamada KM (2007) Cell–matrix adhesion. J Cell Physiol 213:565–573 doi:10.1002/jcp.21237
Boileau C, Jondeau G, Mizuguchi T, Matsumoto N (2005) Molecular genetics of Marfan syndrome. Curr Opin Cardiol 20:194–200 doi:10.1097/01.hco.0000162398.21972.cd
Borg TK, Markwald R (2007) Periostin: more than just an adhesion molecule. Circ Res 101:230–231 doi:10.1161/CIRCRESAHA.107.159103
Bornstein P (2000) Matricellular proteins: an overview. Matrix Biol 19:555–556 doi:10.1016/S0945-053X(00)00103-7
Bornstein P, Sage EH (2002) Matricellular proteins: extracellular modulators of cell function. Curr Opin Cell Biol 14:608–616 doi:10.1016/S0955-0674(02)00361-7
Bornstein P, Kyriakides TR, Yang Z, Armstrong LC, Birk DE (2000) Thrombospondin 2 modulates collagen fibrillogenesis and angiogenesis. J Investig Dermatol Symp Proc 5:61–66 doi:10.1046/j.1087-0024.2000.00005.x
Bornstein P, Agah A, Kyriakides TR (2004) The role of thrombospondins 1 and 2 in the regulation of cell–matrix interactions, collagen fibril formation, and the response to injury. Int J Biochem Cell Biol 36:1115–1125 doi:10.1016/j.biocel.2004.01.012
Butcher JT, Norris RA, Hoffman S, Mjaatvedt CH, Markwald RR (2007) Periostin promotes atrioventricular mesenchyme matrix invasion and remodeling mediated by integrin signaling through Rho/PI 3-kinase. Dev Biol 302:256–266 doi:10.1016/j.ydbio.2006.09.048
Byers PH, Siegel RC, Peterson KE, Rowe DW, Holbrook KA, Smith LT et al (1981) Marfan syndrome: abnormal alpha 2 chain in type I collagen. Proc Natl Acad Sci USA 78:7745–7749 doi:10.1073/pnas.78.12.7745
Callewaert B, Malfait F, Loeys B, De Paepe A (2008) Ehlers–Danlos syndromes and Marfan syndrome. Best Pract Res Clin Rheumatol 22:165–189 doi:10.1016/j.berh.2007.12.005
Canty EG, Kadler KE (2005) Procollagen trafficking, processing and fibrillogenesis. J Cell Sci 118:1341–1353 doi:10.1242/jcs.01731
Chen H, Herndon ME, Lawler J (2000) The cell biology of thrombospondin-1. Matrix Biol 19:597–614 doi:10.1016/S0945-053X(00)00107-4
Collod-Beroud G, Boileau C (2002) Marfan syndrome in the third millennium. Eur J Hum Genet 10:673–681 doi:10.1038/sj.ejhg.5200876
Culav EM, Clark CH, Merrilees MJ (1999) Connective tissues: matrix composition and its relevance to physical therapy. Phys Ther 79:308–319
Darby IA, Hewitson TD (2007) Fibroblast differentiation in wound healing and fibrosis. Int Rev Cytol 257:143–179 doi:10.1016/S0074-7696(07)57004-X
De Coster PJ, Martens LC, De Paepe A (2004) Orofacial manifestations of congenital fibrillin deficiency: pathogenesis and clinical diagnostics. Pediatr Dent 26:535–537
Dorn GW 2nd (2007) Periostin and myocardial repair, regeneration, and recovery. N Engl J Med 357:1552–1554 doi:10.1056/NEJMcibr074816
Elola MT, Wolfenstein-Todel C, Troncoso MF, Vasta GR, Rabinovich GA (2007) Galectins: matricellular glycan-binding proteins linking cell adhesion, migration, and survival. Cell Mol Life Sci 64:1679–1700 doi:10.1007/s00018-007-7044-8
Erkan M, Kleeff J, Gorbachevski A, Reiser C, Mitkus T, Esposito I et al (2007) Periostin creates a tumor-supportive microenvironment in the pancreas by sustaining fibrogenic stellate cell activity. Gastroenterology 132:1447–1464 doi:10.1053/j.gastro.2007.01.031
Francis G, Donnelly PV, Di Ferrante N (1976) Abnormally soluble collagen produced in fibroblasts cultures. Experientia 32:691–693 doi:10.1007/BF01919835
Gallagher GL, Jackson CJ, Hunyor SN (2007) Myocardial extracellular matrix remodeling in ischemic heart failure. Front Biosci 12:1410–1419 doi:10.2741/2157
Gillan L, Matei D, Fishman DA, Gerbin CS, Karlan BY, Chang DD (2002) Periostin secreted by epithelial ovarian carcinoma is a ligand for alpha(V)beta(3) and alpha(V)beta(5) integrins and promotes cell motility. Cancer Res 62:5358–5364
Goetsch SC, Hawke TJ, Gallardo TD, Richardson JA, Garry DJ (2003) Transcriptional profiling and regulation of the extracellular matrix during muscle regeneration. Physiol Genomics 14:261–271
Gruber HE, Sage EH, Norton HJ, Funk S, Ingram J, Hanley EN Jr (2005) Targeted deletion of the SPARC gene accelerates disc degeneration in the aging mouse. J Histochem Cytochem 53:1131–1138 doi:10.1369/jhc.5A6687.2005
Grzesik WJ, Narayanan AS (2002) Cementum and periodontal wound healing and regeneration. Crit Rev Oral Biol Med 13:474–484
Hamilton DW, Brunette DM (2007) The effect of substratum topography on osteoblast adhesion mediated signal transduction and phosphorylation. Biomaterials 28:1806–1819 doi:10.1016/j.biomaterials.2006.11.041
Hamilton DW, Wong KS, Brunette DM (2006) Microfabricated discontinuous-edge surface topographies influence osteoblast adhesion, migration, cytoskeletal organization, and proliferation and enhance matrix and mineral deposition in vitro. Calcif Tissue Int 78:314–325 doi:10.1007/s00223-005-0238-x
Hamilton DW, Chehroudi B, Brunette DM (2007) Comparative response of epithelial cells and osteoblasts to microfabricated tapered pit topographies in vitro and in vivo. Biomaterials 28:2281–2293 doi:10.1016/j.biomaterials.2007.01.026
He J, Baum LG (2006) Galectin interactions with extracellular matrix and effects on cellular function. Methods Enzymol 417:247–256 doi:10.1016/S0076-6879(06)17017-2
Hinz B, Gabbiani G (2003) Cell–matrix and cell–cell contacts of myofibroblasts: role in connective tissue remodeling. Thromb Haemost 90:993–1002
Horiuchi K, Amizuka N, Takeshita S, Takamatsu H, Katsuura M, Ozawa H et al (1999) Identification and characterization of a novel protein, periostin, with restricted expression to periosteum and periodontal ligament and increased expression by transforming growth factor beta. J Bone Miner Res 14:1239–1249 doi:10.1359/jbmr.1999.14.7.1239
Humphries MJ, Travis MA, Clark K, Mould AP (2004) Mechanisms of integration of cells and extracellular matrices by integrins. Biochem Soc Trans 32:822–825 doi:10.1042/BST0320407
Iekushi K, Taniyama Y, Azuma J, Katsuragi N, Dosaka N, Sanada F et al (2007) Novel mechanisms of valsartan on the treatment of acute myocardial infarction through inhibition of the antiadhesion molecule periostin. Hypertension 49:1409–1414 doi:10.1161/HYPERTENSIONAHA.106.080994
Kii I, Kudo A (2007) Periostin function in the periodontal ligament and the periosteum. Clin Calcium 17:202–208
Kii I, Amizuka N, Minqi L, Kitajima S, Saga Y, Kudo A (2006) Periostin is an extracellular matrix protein required for eruption of incisors in mice. Biochem Biophys Res Commun 342:766–772 doi:10.1016/j.bbrc.2006.02.016
Kojima T, Freitas PH, Ubaidus S, Suzuki A, Li M, Yoshizawa M et al (2007) Histochemical examinations on cortical bone regeneration induced by thermoplastic bioresorbable plates applied to bone defects of rat calvariae. Biomed Res 28:219–229 doi:10.2220/biomedres.28.219
Kruzynska-Frejtag A, Wang J, Maeda M, Rogers R, Krug E, Hoffman S et al (2004) Periostin is expressed within the developing teeth at the sites of epithelial–mesenchymal interaction. Dev Dyn 229:857–868 doi:10.1002/dvdy.10453
Kudo H, Amizuka N, Araki K, Inohaya K, Kudo A (2004) Zebrafish periostin is required for the adhesion of muscle fiber bundles to the myoseptum and for the differentiation of muscle fibers. Dev Biol 267:473–487 doi:10.1016/j.ydbio.2003.12.007
Kudo Y, Siriwardena BS, Hatano H, Ogawa I, Takata T (2007) Periostin: novel diagnostic and therapeutic target for cancer. Histol Histopathol 22:1167–1174
Kuhn B, del Monte F, Hajjar RJ, Chang YS, Lebeche D, Arab S et al (2007) Periostin induces proliferation of differentiated cardiomyocytes and promotes cardiac repair. Nat Med 13:962–969 doi:10.1038/nm1619
Kuiper EJ, Roestenberg P, Ehlken C, Lambert V, van Treslong-de Groot HB, Lyons KM et al (2007) Angiogenesis is not impaired in connective tissue growth factor (CTGF) knock-out mice. J Histochem Cytochem 55:1139–1147 doi:10.1369/jhc.7A7258.2007
Kyriakides TR, Bornstein P (2003) Matricellular proteins as modulators of wound healing and the foreign body response. Thromb Haemost 90:986–992
Lallier TE, Spencer A (2007) Use of microarrays to find novel regulators of periodontal ligament fibroblast differentiation. Cell Tissue Res 327:93–109 doi:10.1007/s00441-006-0282-5
Larsen M, Artym VV, Green JA, Yamada KM (2006) The matrix reorganized: extracellular matrix remodeling and integrin signaling. Curr Opin Cell Biol 18:463–471 doi:10.1016/j.ceb.2006.08.009
Leask A, Abraham DJ (2006) All in the CCN family: essential matricellular signaling modulators emerge from the bunker. J Cell Sci 119:4803–4810 doi:10.1242/jcs.03270
Leask A, Denton CP, Abraham DJ (2004) Insights into the molecular mechanism of chronic fibrosis: the role of connective tissue growth factor in scleroderma. J Invest Dermatol 122:1–6 doi:10.1046/j.0022-202X.2003.22133.x
Li P, Oparil S, Feng W, Chen YF (2004) Hypoxia-responsive growth factors upregulate periostin and osteopontin expression via distinct signaling pathways in rat pulmonary arterial smooth muscle cells. J Appl Physiol 97:1550–1558 discussion 1549 doi:10.1152/japplphysiol.01311.2003
Li G, Oparil S, Sanders JM, Zhang L, Dai M, Chen LB et al (2006) Phosphatidylinositol-3-kinase signaling mediates vascular smooth muscle cell expression of periostin in vivo and in vitro. Atherosclerosis 188:292–300 doi:10.1016/j.atherosclerosis.2005.11.002
Lindner V, Wang Q, Conley BA, Friesel RE, Vary CP (2005) Vascular injury induces expression of periostin: implications for vascular cell differentiation and migration. Arterioscler Thromb Vasc Biol 25:77–83
Litvin J, Selim AH, Montgomery MO, Lehmann K, Rico MC, Devlin H et al (2004) Expression and function of periostin-isoforms in bone. J Cell Biochem 92:1044–1061 doi:10.1002/jcb.20115
Litvin J, Chen X, Keleman S, Zhu S, Autieri M (2007) Expression and function of periostin-like factor in vascular smooth muscle cells. Am J Physiol Cell Physiol 292:C1672–C1680 doi:10.1152/ajpcell.00153.2006
Lock JG, Wehrle-Haller B, Stromblad S (2008) Cell–matrix adhesion complexes: master control machinery of cell migration. Semin Cancer Biol 18:65–76 doi:10.1016/j.semcancer.2007.10.001
Lukashev ME, Werb Z (1998) ECM signalling: orchestrating cell behaviour and misbehaviour. Trends Cell Biol 8:437–441 doi:10.1016/S0962-8924(98)01362-2
Majesky MW (1994) Neointima formation after acute vascular injury. Role of counteradhesive extracellular matrix proteins. Tex Heart Inst J 21:78–85
Martinek N, Shahab J, Sodek J, Ringuette M (2007) Is SPARC an evolutionarily conserved collagen chaperone? J Dent Res 86:296–305
Midwood KS, Williams LV, Schwarzbauer JE (2004) Tissue repair and the dynamics of the extracellular matrix. Int J Biochem Cell Biol 36:1031–1037 doi:10.1016/j.biocel.2003.12.003
Muller U (1999) Ten years of gene targeting: targeted mouse mutants, from vector design to phenotype analysis. Mech Dev 82:3–21 doi:10.1016/S0925-4773(99)00021-0
Murphy-Ullrich JE (2001) The de-adhesive activity of matricellular proteins: is intermediate cell adhesion an adaptive state? J Clin Invest 107:785–790 doi:10.1172/JCI12609
Nakazawa T, Nakajima A, Seki N, Okawa A, Kato M, Moriya H et al (2004) Gene expression of periostin in the early stage of fracture healing detected by cDNA microarray analysis. J Orthop Res 22:520–525 doi:10.1016/j.orthres.2003.10.007
Nasuti JF, Zhang PJ, Feldman MD, Pasha T, Khurana JS, Gorman JH 3rd et al (2004) Fibrillin and other matrix proteins in mitral valve prolapse syndrome. Ann Thorac Surg 77:532–536 doi:10.1016/S0003-4975(03)01584-4
Norris RA, Damon B, Mironov V, Kasyanov V, Ramamurthi A, Moreno-Rodriguez R et al (2007) Periostin regulates collagen fibrillogenesis and the biomechanical properties of connective tissues. J Cell Biochem 101:695–711 doi:10.1002/jcb.21224
Norris RA, Borg TK, Butcher JT, Baudino TA, Banerjee I, Markwald RR (2008a) Neonatal and adult cardiovascular pathophysiological remodeling and repair: developmental role of periostin. Ann N Y Acad Sci 1123:30–40 doi:10.1196/annals.1420.005
Norris RA, Moreno-Rodriguez RA, Sugi Y, Hoffman S, Amos J, Hart MM et al (2008b) Periostin regulates atrioventricular valve maturation. Dev Biol 316:200–213 doi:10.1016/j.ydbio.2008.01.003
Oka T, Xu J, Kaiser RA, Melendez J, Hambleton M, Sargent MA et al (2007) Genetic manipulation of periostin expression reveals a role in cardiac hypertrophy and ventricular remodeling. Circ Res 101:313–321 doi:10.1161/CIRCRESAHA.107.149047
Oku E, Kanaji T, Takata Y, Oshima K, Seki R, Morishige S, Imamura R, Ohtsubo K, Hashiguchi M, Osaki K et al. (2008) Periostin and bone marrow fibrosis. Int J Hematol 88(1):57–63 doi:10.1007/s12185-008-0095-2
Oshima A, Tanabe H, Yan T, Lowe GN, Glackin CA, Kudo A (2002) A novel mechanism for the regulation of osteoblast differentiation: transcription of periostin, a member of the fasciclin I family, is regulated by the bHLH transcription factor, twist. J Cell Biochem 86:792–804 doi:10.1002/jcb.10272
Park YW, Kang YM, Butterfield J, Detmar M, Goronzy JJ, Weyand CM (2004) Thrombospondin 2 functions as an endogenous regulator of angiogenesis and inflammation in rheumatoid arthritis. Am J Pathol 165:2087–2098
Pirinen S (1998) Genetic craniofacial aberrations. Acta Odontol Scand 56:356–359 doi:10.1080/000163598428310
Priest RE, Moinuddin JF, Priest JH (1973) Letter: collagen of Marfan syndrome is abnormally soluble. Nature 245:264–266 doi:10.1038/245264a0
Raines EW (2000) The extracellular matrix can regulate vascular cell migration, proliferation, and survival: relationships to vascular disease. Int J Exp Pathol 81:173–182 doi:10.1046/j.1365-2613.2000.00155.x
Rios H, Koushik SV, Wang H, Wang J, Zhou HM, Lindsley A et al (2005) Periostin null mice exhibit dwarfism, incisor enamel defects, and an early-onset periodontal disease-like phenotype. Mol Cell Biol 25:11131–11144 doi:10.1128/MCB.25.24.11131-11144.2005
Robinson PN, Booms P (2001) The molecular pathogenesis of the Marfan syndrome. Cell Mol Life Sci 58:1698–1707 doi:10.1007/PL00000807
Roy S, Patel D, Khanna S, Gordillo GM, Biswas S, Friedman A et al (2007) Transcriptome-wide analysis of blood vessels laser captured from human skin and chronic wound-edge tissue. Proc Natl Acad Sci U S A 104:14472–14477 doi:10.1073/pnas.0706793104
Schuler M, Owen GR, Hamilton DW, de Wild M, Textor M, Brunette DM et al (2006) Biomimetic modification of titanium dental implant model surfaces using the RGDSP-peptide sequence: a cell morphology study. Biomaterials 27:4003–4015 doi:10.1016/j.biomaterials.2006.03.009
Shimazaki M, Nakamura K, Kii I, Kashima T, Amizuka N, Li M et al (2008) Periostin is essential for cardiac healing after acute myocardial infarction. J Exp Med 205:295–303 doi:10.1084/jem.20071297
Snider P, Hinton RB, Moreno-Rodriguez RA, Wang J, Rogers R, Lindsley A et al (2008) Periostin is required for maturation and extracellular matrix stabilization of noncardiomyocyte lineages of the heart. Circ Res 102:752–760 doi:10.1161/CIRCRESAHA.107.159517
Stamenkovic I (2003) Extracellular matrix remodelling: the role of matrix metalloproteinases. J Pathol 200:448–464 doi:10.1002/path.1400
Suzuki H, Amizuka N, Kii I, Kawano Y, Nozawa-Inoue K, Suzuki A et al (2004) Immunohistochemical localization of periostin in tooth and its surrounding tissues in mouse mandibles during development. Anat Rec A Discov Mol Cell Evol Biol 281:1264–1275 doi:10.1002/ar.a.20080
Tai IT, Dai M, Chen LB (2005) Periostin induction in tumor cell line explants and inhibition of in vitro cell growth by anti-periostin antibodies. Carcinogenesis 26:908–915 doi:10.1093/carcin/bgi034
Takayama G, Arima K, Kanaji T, Toda S, Tanaka H, Shoji S et al (2006) Periostin: a novel component of subepithelial fibrosis of bronchial asthma downstream of IL-4 and IL-13 signals. J Allergy Clin Immunol 118:98–104 doi:10.1016/j.jaci.2006.02.046
Takeshita S, Kikuno R, Tezuka K, Amann E (1993) Osteoblast-specific factor 2: cloning of a putative bone adhesion protein with homology with the insect protein fasciclin I. Biochem J 294(Pt 1):271–278
Tayebjee MH, MacFadyen RJ, Lip GY (2003) Extracellular matrix biology: a new frontier in linking the pathology and therapy of hypertension? J Hypertens 21:2211–2218 doi:10.1097/00004872-200312000-00002
Tilman G, Mattiussi M, Brasseur F, van Baren N, Decottignies A (2007) Human periostin gene expression in normal tissues, tumors and melanoma: evidences for periostin production by both stromal and melanoma cells. Mol Cancer 6:80 doi:10.1186/1476-4598-6-80
Tomokiyo A, Maeda H, Fujii S, Wada N, Shima K, Akamine A (2008) Development of a multipotent clonal human periodontal ligament cell line. Differentiation 76:337–347 doi:10.1111/j.1432-0436.2007.00233.x
Uitto J (1979) Biochemistry of the elastic fibers in normal connective tissues and its alterations in diseases. J Invest Dermatol 72:1–10 doi:10.1111/1523-1747.ep12530093
Uitto J, Lichtenstein JR (1976) Defects in the biochemistry of collagen in diseases of connective tissue. J Invest Dermatol 66:59–79 doi:10.1111/1523-1747.ep12481404
Wallner K, Li C, Fishbein MC, Shah PK, Sharifi BG (1999) Arterialization of human vein grafts is associated with tenascin-C expression. J Am Coll Cardiol 34:871–875 doi:10.1016/S0735-1097(99)00272-7
Wallner K, Shah PK, Sharifi BG (2002) Balloon catheterization induces arterial expression of new tenascin-C isoform. Atherosclerosis 161:75–83 doi:10.1016/S0021-9150(01)00627-X
Westling L, Mohlin B, Bresin A (1998) Craniofacial manifestations in the Marfan syndrome: palatal dimensions and a comparative cephalometric analysis. J Craniofac Genet Dev Biol 18:211–218
Whiteman P, Hutchinson S, Handford PA (2006) Fibrillin-1 misfolding and disease. Antioxid Redox Signal 8:338–346 doi:10.1089/ars.2006.8.338
Wilde J, Yokozeki M, Terai K, Kudo A, Moriyama K (2003) The divergent expression of periostin mRNA in the periodontal ligament during experimental tooth movement. Cell Tissue Res 312:345–351 doi:10.1007/s00441-002-0664-2
Woodruff PG, Boushey HA, Dolganov GM, Barker CS, Yang YH, Donnelly S et al (2007) Genome-wide profiling identifies epithelial cell genes associated with asthma and with treatment response to corticosteroids. Proc Natl Acad Sci U S A 104:15858–15863 doi:10.1073/pnas.0707413104
Yan Q, Sage EH (1999) SPARC, a matricellular glycoprotein with important biological functions. J Histochem Cytochem 47:1495–1506
Yang Z, Kyriakides TR, Bornstein P (2000) Matricellular proteins as modulators of cell–matrix interactions: adhesive defect in thrombospondin 2-null fibroblasts is a consequence of increased levels of matrix metalloproteinase-2. Mol Biol Cell 11:3353–3364
Zelenka PS (2004) Regulation of cell adhesion and migration in lens development. Int J Dev Biol 48:857–865 doi:10.1387/ijdb.041871pz
Acknowledgements
Dr Hamilton would like to thank Dr Simon Conway for graciously providing periostin knockout mice. He would also like to thanks Dr David Holdsworth for performing the microCT analysis, and Pastor Salano for his help in analyzing the data.
Author information
Authors and Affiliations
Corresponding author
Additional information
This review also appears on line under CCNExpress section of the Newsletters page on ICCNS Website at http://ccnsociety.com.
Rights and permissions
About this article
Cite this article
Hamilton, D.W. Functional role of periostin in development and wound repair: implications for connective tissue disease. J. Cell Commun. Signal. 2, 9–17 (2008). https://doi.org/10.1007/s12079-008-0023-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-008-0023-5