Abstract
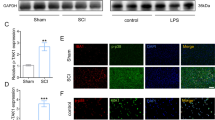
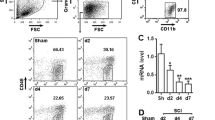
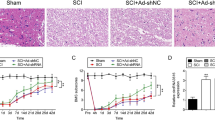
Mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MALT1) is involved in neural injury, neuroinflammation, microglia activation, and polarization, while its function in spinal cord injury (SCI) remains unclear. Thus, this study aimed to evaluate the role of MALT1 modification on SCI recovery and its underlying mechanism. SCI surgery or sham surgery was performed in Sprague–Dawley rats. Then, MALT1 knockdown or negative control lentivirus was injected into SCI rats. Subsequently, MALT1 expression, locomotor capability, neural injury, markers for microglia activation and polarization, inflammatory cytokine expressions, and nuclear factor (NF)-κB pathway were detected. SCI rats exhibited higher MALT1 expression, microglia activation and M1 polarization, neuroinflammation, and NF-κB pathway activation, while worse locomotor capacity compared to sham rats (all P < 0.05). In SCI rats, MALT1 knockdown alleviated Basso, Beattie, and Bresnahan score from 10 to 28 days and attenuated HE staining reflected neural injury (all P < 0.05). Besides, MALT1 knockdown declined the number of IBA1+ cells, IBA1+ iNOS+ cells, and IBA1+ CD86+ cells, while enhanced the number of IBA1+ Arg1+ cells and IBA1+ CD206+ cells in SCI rats (all P < 0.05). Meanwhile, MALT1 knockdown declined the expressions of IL-1β, IL-6, and TNF-α in SCI (all P < 0.05), but did not affect IL-10 expression (P > 0.05). Furthermore, MALT1 knockdown suppressed NF-κB pathway activation validated by immunofluorescence staining and western blot assays (all P < 0.05). MALT1 knockdown improves functional recovery, attenuates microglia activation, M1 polarization, and neuroinflammation via inhibiting NF-κB pathway in SCI.







Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Eckert MJ, Martin MJ (2017) Trauma: spinal cord injury. Surg Clin North Am 97(5):1031–1045. https://doi.org/10.1016/j.suc.2017.06.008
Witiw CD, Fehlings MG (2015) Acute spinal cord injury. J Spinal Disord Tech 28(6):202–210. https://doi.org/10.1097/BSD.0000000000000287
Fan B, Wei Z, Yao X, Shi G, Cheng X, Zhou X, Zhou H, Ning G et al (2018) Microenvironment imbalance of spinal cord injury. Cell Transplant 27(6):853–866. https://doi.org/10.1177/0963689718755778
Karsy M, Hawryluk G (2019) Modern medical management of spinal cord injury. Curr Neurol Neurosci Rep 19(9):65. https://doi.org/10.1007/s11910-019-0984-1
Cote MP, Murray M, Lemay MA (2017) Rehabilitation strategies after spinal cord injury: inquiry into the mechanisms of success and failure. J Neurotrauma 34(10):1841–1857. https://doi.org/10.1089/neu.2016.4577
Chay W, Kirshblum S (2020) Predicting outcomes after spinal cord injury. Phys Med Rehabil Clin N Am 31(3):331–343. https://doi.org/10.1016/j.pmr.2020.03.003
Kip E, Staal J, Tima HG, Verstrepen L, Romano M, Lemeire K, Suin V, Hamouda A et al (2018) Inhibition of MALT1 decreases neuroinflammation and pathogenicity of virulent rabies Virus in Mice. J Virol 92(22). https://doi.org/10.1128/JVI.00720-18
Zhang S, Yan Y, Wang Y, Sun Z, Han C, Qian X, Ren X, Feng Y et al (2021) Inhibition of MALT1 alleviates spinal ischemia/reperfusion injury-induced neuroinflammation by modulating glial endoplasmic reticulum stress in rats. J Inflamm Res 14:4329–4345. https://doi.org/10.2147/JIR.S319023
Monajemi M, Pang YCF, Bjornson S, Menzies SC, van Rooijen N, Sly LM (2018) Malt1 blocks IL-1beta production by macrophages in vitro and limits dextran sodium sulfate-induced intestinal inflammation in vivo. J Leukoc Biol 104(3):557–572. https://doi.org/10.1002/JLB.3VMA0118-019R
Liu W, Guo W, Hang N, Yang Y, Wu X, Shen Y, Cao J, Sun Y et al (2016) MALT1 inhibitors prevent the development of DSS-induced experimental colitis in mice via inhibiting NF-kappaB and NLRP3 inflammasome activation. Oncotarget 7(21):30536–30549. https://doi.org/10.18632/oncotarget.8867
Luo Y, Xu T, Liu W, Rong Y, Wang J, Fan J, Yin G, Cai W (2021) Exosomes derived from GIT1-overexpressing bone marrow mesenchymal stem cells promote traumatic spinal cord injury recovery in a rat model. Int J Neurosci 131(2):170–182. https://doi.org/10.1080/00207454.2020.1734598
Du Y, Zhao Y, Li C, Zheng Q, Tian J, Li Z, Huang TY, Zhang W et al (2018) Inhibition of PKCdelta reduces amyloid-beta levels and reverses Alzheimer disease phenotypes. J Exp Med 215(6):1665–1677. https://doi.org/10.1084/jem.20171193
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21. https://doi.org/10.1089/neu.1995.12.1
Staal J, Driege Y, Haegman M, Kreike M, Iliaki S, Vanneste D, Lork M, Afonina IS et al (2021) Defining the combinatorial space of PKC::CARD-CC signal transduction nodes. FEBS J 288(5):1630–1647. https://doi.org/10.1111/febs.15522
Jaworski M, Thome M (2016) The paracaspase MALT1: biological function and potential for therapeutic inhibition. Cell Mol Life Sci 73(3):459–473. https://doi.org/10.1007/s00018-015-2059-z
Hachmann J, Salvesen GS (2016) The paracaspase MALT1. Biochimie 122:324–338. https://doi.org/10.1016/j.biochi.2015.09.018
O’Shea TM, Burda JE, Sofroniew MV (2017) Cell biology of spinal cord injury and repair. J Clin Invest 127(9):3259–3270. https://doi.org/10.1172/JCI90608
Gensel JC, Zhang B (2015) Macrophage activation and its role in repair and pathology after spinal cord injury. Brain Res 1619:1–11. https://doi.org/10.1016/j.brainres.2014.12.045
Lyu J, Xie D, Bhatia TN, Leak RK, Hu X, Jiang X (2021) Microglial/macrophage polarization and function in brain injury and repair after stroke. CNS Neurosci Ther 27(5):515–527. https://doi.org/10.1111/cns.13620
Perego C, Fumagalli S, De Simoni MG (2011) Temporal pattern of expression and colocalization of microglia/macrophage phenotype markers following brain ischemic injury in mice. J Neuroinflammation 8:174. https://doi.org/10.1186/1742-2094-8-174
Kroner A, Greenhalgh AD, Zarruk JG, Passos Dos Santos R, Gaestel M, David S (2014) TNF and increased intracellular iron alter macrophage polarization to a detrimental M1 phenotype in the injured spinal cord. Neuron 83(5):1098–1116. https://doi.org/10.1016/j.neuron.2014.07.027
Zhao R, Ying M, Gu S, Yin W, Li Y, Yuan H, Fang S, Li M (2019) Cysteinyl leukotriene receptor 2 is involved in inflammation and neuronal damage by mediating microglia M1/M2 polarization through NF-kappaB pathway. Neuroscience 422:99–118. https://doi.org/10.1016/j.neuroscience.2019.10.048
Tian X, Liu H, Xiang F, Xu L, Dong Z (2019) beta-Caryophyllene protects against ischemic stroke by promoting polarization of microglia toward M2 phenotype via the TLR4 pathway. Life Sci 237:116915. https://doi.org/10.1016/j.lfs.2019.116915
Zhu J, Cao D, Guo C, Liu M, Tao Y, Zhou J, Wang F, Zhao Y et al (2019) Berberine facilitates angiogenesis against ischemic stroke through modulating microglial polarization via AMPK signaling. Cell Mol Neurobiol 39(6):751–768. https://doi.org/10.1007/s10571-019-00675-7
Fusco R, Siracusa R, D’Amico R, Cordaro M, Genovese T, Gugliandolo E, Peritore AF, Crupi R et al (2020) Mucosa-associated lymphoid tissue lymphoma translocation 1 inhibitor as a novel therapeutic tool for lung injury. Int J Mol Sci 21(20). https://doi.org/10.3390/ijms21207761
Wu TM, Nan FH, Chen KC, Wu YS (2019) Sarcodia suieae acetyl-xylogalactan regulate RAW 264.7 macrophage NF-kappa B activation and IL-1 beta cytokine production in macrophage polarization. Sci Rep 9(1):19627. https://doi.org/10.1038/s41598-019-56246-9
Shenderov K, Barber DL, Mayer-Barber KD, Gurcha SS, Jankovic D, Feng CG, Oland S, Hieny S et al (2013) Cord factor and peptidoglycan recapitulate the Th17-promoting adjuvant activity of mycobacteria through mincle/CARD9 signaling and the inflammasome. J Immunol 190(11):5722–5730. https://doi.org/10.4049/jimmunol.1203343
Van Opdenbosch N, Lamkanfi M (2019) Caspases in cell death, inflammation, and disease. Immunity 50(6):1352–1364. https://doi.org/10.1016/j.immuni.2019.05.020
Qureshi N, Morrison DC (1823) Reis J (2012) Proteasome protease mediated regulation of cytokine induction and inflammation. Biochim Biophys Acta 11:2087–2093. https://doi.org/10.1016/j.bbamcr.2012.06.016
Zhang J, Zheng Y, Luo Y, Du Y, Zhang X, Fu J (2019) Curcumin inhibits LPS-induced neuroinflammation by promoting microglial M2 polarization via TREM2/ TLR4/ NF-kappaB pathways in BV2 cells. Mol Immunol 116:29–37. https://doi.org/10.1016/j.molimm.2019.09.020
Yang X, Xu S, Qian Y, Xiao Q (2017) Resveratrol regulates microglia M1/M2 polarization via PGC-1alpha in conditions of neuroinflammatory injury. Brain Behav Immun 64:162–172. https://doi.org/10.1016/j.bbi.2017.03.003
Xu L, Botchway BOA, Zhang S, Zhou J, Liu X (2018) Inhibition of NF-kappaB signaling pathway by resveratrol improves spinal cord injury. Front Neurosci 12:690. https://doi.org/10.3389/fnins.2018.00690
Liu C, Shi Z, Fan L, Zhang C, Wang K, Wang B (2011) Resveratrol improves neuron protection and functional recovery in rat model of spinal cord injury. Brain Res 1374:100–109. https://doi.org/10.1016/j.brainres.2010.11.061
Gao S, Xu T, Guo H, Deng Q, Xun C, Liang W, Sheng W (2019) Ameliorative effects of echinacoside against spinal cord injury via inhibiting NLRP3 inflammasome signaling pathway. Life Sci 237:116978. https://doi.org/10.1016/j.lfs.2019.116978
Afonina IS, Elton L, Carpentier I, Beyaert R (2015) MALT1—a universal soldier: multiple strategies to ensure NF-kappaB activation and target gene expression. FEBS J 282(17):3286–3297. https://doi.org/10.1111/febs.13325
Unterreiner A, Touil R, Anastasi D, Dubois N, Niwa S, Calzascia T, Bornancin F (2019) Myeloid innate signaling pathway regulation by MALT1 paracaspase activity. J Vis Exp (143). https://doi.org/10.3791/58439
DeVore SB, Khurana Hershey GK (2022) The role of the CBM complex in allergic inflammation and disease. J Allergy Clin Immunol. https://doi.org/10.1016/j.jaci.2022.06.023
Kjell J, Olson L (2016) Rat models of spinal cord injury: from pathology to potential therapies. Dis Model Mech 9(10):1125–1137. https://doi.org/10.1242/dmm.025833
Funding
This study was supported by the 2021 Nanshan District Health Technology Plan Project (NS094).
Author information
Authors and Affiliations
Contributions
SG and QPZ substantially contributed to the conception and the design of the study. SZ and HC were responsible for the acquisition of the data. GC and CC were responsible for the analysis of the data. JZ and WW contributed to the interpretation of the data. QHZ contributed to manuscript drafting and edited English language. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
The protocols of this study were approved by Animal Care and Use Committee, and the experiments were carried out by following the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Supplementary Figure 1.
Co-localization of MALT1 and IBA1 in SCI rats. The presentation (A) and further comparison (B) of MALT1+ cells in different groups. The presentation of MALT1+ IBA1+ cells (C), co-localization analysis between MALT1 and IBA1 from the Pearson coefficient (D), and the comparison of IBA1+ cells MATL1 fluorescence intensity (E) in different groups. *** represented P<0.001, ** represented P<0.01, * represented P<0.05, NS: non-significant. (PNG 1714 kb)

Supplementary Figure 2.
Detection of upstream molecules of MALT1 in SCI rats. The presentation (A) and further comparison (B) of IBA1+ cells CARD9 fluorescent intensity in different groups. The presentation (C) and further comparison (D) of IBA1+ cells BCL10 fluorescent intensity in different groups. NS: non-significant. (PNG 1322 kb)

Supplementary Figure 3.
Effect of MI-2 and Rottlerin on locomotor capability and neural injury in SCI. Comparison of BBB score among different groups (A). Presentation of neural injury in different groups using HE staining (B). In (A): *** represented P<0.001 between SCI group and sham group, # represented P<0.05 between SCI group and SCI+MI-2 group, $ represented P<0.05 between SCI group and SCI+ Rottlerin group. (PNG 1099 kb)

Supplementary Figure 4.
Effect of MI-2 and Rottlerin on microglia M1 polarization in SCI. The presentation (A) and further comparison (B) of IBA1+ iNOS+ cells in different groups. The presentation (C) and further comparison (D) of IBA1+ CD86+ cells in different groups. *** represented P<0.001, ** represented P<0.01, * represented P<0.05, NS: non-significant. (PNG 987 kb)

Supplementary Figure 5.
Effect of MI-2 and Rottlerin on microglia M2 polarization in SCI. The presentation (A) and further comparison (B) of IBA1+ Arg1+ cells in different groups. The presentation (C) and further comparison (D) of IBA1+ CD206+ cells in different groups. ** represented P<0.01, * represented P<0.05, NS: non-significant. (PNG 892 kb)

Supplementary Figure 6.
Effect of MI-2 and Rottlerin on inflammatory cytokine expressions in SCI. Comparison of the levels of IL-1β (A), IL-6 (B), TNF-α (C) and IL-10 (D) in different groups. *** represented P<0.001, ** represented P<0.01, * represented P<0.05, NS: non-significant. (PNG 1234 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Q., Zhang, S., Chen, H. et al. Targeting of MALT1 May Improve Functional Recovery and Attenuate Microglia M1 Polarization-Mediated Neuroinflammation During Spinal Cord Injury. Mol Neurobiol 60, 2632–2643 (2023). https://doi.org/10.1007/s12035-023-03208-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03208-y