Abstract
Recurrent glioblastoma is characterized by resistance to radiotherapy or chemotherapy. In this study, we investigated the role of TRIM56 in radiosensitization and its potential underlying molecular mechanism. TRIM56 expression levels were measured in glioblastoma tissues and cell lines by immunohistochemical staining, western blot, and qRT-PCR. MTT assay, colony formation assay, and TUNEL assay were used to investigate the effect of TRIM56 on cell viability, cell proliferation, and cell apoptosis. Co-immunoprecipitation was used to clarify the interaction between TRIM56 and FOXM1. Finally, tumor xenograft experiments were performed to analyze the effect of TRIM56 on tumor growth in vivo. The expression of TRIM56 was significantly increased in glioblastoma tissues and cell lines and its expression was associated with poor prognosis of patients with glioblastoma. Moreover, TRIM56 reduced the radiosensitivity of glioblastoma cells and promoted DNA repairment. Mechanistically, TRIM56 promoted FOXM1 protein level, enhanced the stability of FOXM1 by de-ubiquitination, and promoted DNA damage repair through FOXM1 in glioblastoma cells. TRIM56 could reduce the radiosensitivity of glioblastoma in vivo. TRIM56 may suppress the radiosensitization of human glioblastoma by regulating FOXM1-mediated DNA repair. Targeting the TRIM56 may be an effective method to reverse radiotherapy-resistant in glioblastoma recurrent.
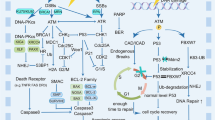
Graphical Abstract








Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- TRIM:
-
Tripartite-motif
- TRIM56:
-
Tripartite-motif-containing protein 56
- Ub:
-
Ubiquitin
- PCBP1:
-
Poly r(C) binding protein 1
References
Ostrom QT, Truitt G, Gittleman H, Brat DJ, Kruchko C, Wilson R, Barnholtz-Sloan JS (2020) Relative survival after diagnosis with a primary brain or other central nervous system tumor in the National Program of Cancer Registries, 2004 to 2014. Neurooncol Pract 7(3):306–312. https://doi.org/10.1093/nop/npz059
Nieder C, Astner ST, Grosu AL (2012) Glioblastoma research 2006–2010: pattern of citation and systematic review of highly cited articles. Clin Neurol Neurosurg 114(9):1207–1210. https://doi.org/10.1016/j.clineuro.2012.03.049
Yuan Y, Shi Q, Li M, Nagamuthu C, Andres E, Davis FG (2016) Canadian brain cancer survival rates by tumour type and region. Can J Public Health 107(1):e37–e42. https://doi.org/10.17269/cjph.107.5209
Batash R, Asna N, Schaffer P, Francis N, Schaffer M (2017) Glioblastoma multiforme, diagnosis and treatment; recent literature review. Curr Med Chem 24(27):3002–3009. https://doi.org/10.2174/0929867324666170516123206
Noch EK, Ramakrishna R, Magge R (2018) Challenges in the treatment of glioblastoma: multisystem mechanisms of therapeutic resistance. World Neurosurg 116:505–517. https://doi.org/10.1016/j.wneu.2018.04.022
Chuang DF, Lin X (2019) Targeted therapies for the treatment of glioblastoma in adults. Curr Oncol Rep 21(7):61. https://doi.org/10.1007/s11912-019-0807-1
Zhou W, Yao Y, Scott AJ, Wilder-Romans K, Dresser JJ, Werner CK, Sun H, Pratt D, Sajjakulnukit P, Zhao SG et al (2020) Purine metabolism regulates DNA repair and therapy resistance in glioblastoma. Nat Commun 11(1):3811. https://doi.org/10.1038/s41467-020-17512-x
Munir M (2010) TRIM proteins: another class of viral victims. Sci Signal 3(118):jc2. https://doi.org/10.1126/scisignal.3118jc2
Ozato K, Shin DM, Chang TH, Morse HC 3rd (2008) TRIM family proteins and their emerging roles in innate immunity. Nat Rev Immunol 8(11):849–860. https://doi.org/10.1038/nri2413
Mandell MA, Jain A, Arko-Mensah J, Chauhan S, Kimura T, Dinkins C, Silvestri G, Münch J, Kirchhoff F, Simonsen A et al (2014) TRIM proteins regulate autophagy and can target autophagic substrates by direct recognition. Dev Cell 30(4):394–409. https://doi.org/10.1016/j.devcel.2014.06.013
Nenasheva VV, Stepanenko EA, Makarova IV, Khaidarova NV, Antonov SA, Kozikova LV, Polteva EA, Kovaleva GV, Ayed Z, Vovk AD et al (2018) Expression of the human TRIM14 and its mutant form (P207L) promotes apoptosis in transgenic loaches. Mol Biol Rep 45(6):2087–2093. https://doi.org/10.1007/s11033-018-4365-7
Hatakeyama S (2017) TRIM family proteins: roles in autophagy, immunity, and carcinogenesis. Trends Biochem Sci 42(4):297–311. https://doi.org/10.1016/j.tibs.2017.01.002
Jaworska AM, Wlodarczyk NA, Mackiewicz A, Czerwinska P (2020) The role of TRIM family proteins in the regulation of cancer stem cell self-renewal. Stem Cells 38(2):165–173. https://doi.org/10.1002/stem.3109
Fiskin E, Bhogaraju S, Herhaus L, Kalayil S, Hahn M, Dikic I (2017) Structural basis for the recognition and degradation of host TRIM proteins by Salmonella effector SopA. Nat Commun 8:14004. https://doi.org/10.1038/ncomms14004
Zhao L, Wang ZG, Zhang P, Yu XF, Su XJ (2019) Poly r(C) binding protein 1 regulates posttranscriptional expression of the ubiquitin ligase TRIM56 in ovarian cancer. IUBMB Life 71(2):177–182. https://doi.org/10.1002/iub.1948
Zhao L, Zhang P, Su XJ, Zhang B (2018) The ubiquitin ligase TRIM56 inhibits ovarian cancer progression by targeting vimentin. J Cell Physiol 233(3):2420–2425. https://doi.org/10.1002/jcp.26114
Zhang PB, Huang ZL, Xu YH, Huang J, Huang XY, Huang XY (2019) Systematic analysis of gene expression profiles reveals prognostic stratification and underlying mechanisms for muscle-invasive bladder cancer. Cancer Cell Int 19:337. https://doi.org/10.1186/s12935-019-1056-y
Zhang X, Lv QL, Huang YT, Zhang LH, Zhou HH (2017) Akt/FoxM1 signaling pathway-mediated upregulation of MYBL2 promotes progression of human glioma. J Exp Clin Cancer Res 36(1):105. https://doi.org/10.1186/s13046-017-0573-6
Tao W, Zhang A, Zhai K, Huang Z, Huang H, Zhou W, Huang Q, Fang X, Prager BC, Wang X et al (2020) SATB2 drives glioblastoma growth by recruiting CBP to promote FOXM1 expression in glioma stem cells. EMBO Mol Med 12(12):e12291. https://doi.org/10.15252/emmm.202012291
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K et al (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10(5):459–466. https://doi.org/10.1016/s1470-2045(09)70025-7
Lesueur P, Lequesne J, Grellard JM, Dugué A, Coquan E, Brachet PE, Geffrelot J, Kao W, Emery E, Berro DH et al (2019) Phase I/IIa study of concomitant radiotherapy with olaparib and temozolomide in unresectable or partially resectable glioblastoma: OLA-TMZ-RTE-01 trial protocol. BMC Cancer 19(1):198. https://doi.org/10.1186/s12885-019-5413-y
Fabbro-Peray P, Zouaoui S, Darlix A, Fabbro M, Pallud J, Rigau V, Mathieu-Daude H, Bessaoud F, Bauchet F, Riondel A et al (2019) Association of patterns of care, prognostic factors, and use of radiotherapy-temozolomide therapy with survival in patients with newly diagnosed glioblastoma: a French national population-based study. J Neurooncol 142(1):91–101. https://doi.org/10.1007/s11060-018-03065-z
Rajendra J, Datta KK, Ud Din Farooqee SB, Thorat R, Kumar K, Gardi N, Kaur E, Nair J, Salunkhe S, Patkar K et al (2018) Enhanced proteasomal activity is essential for long term survival and recurrence of innately radiation resistant residual glioblastoma cells. Oncotarget 9(45):27667–27681. https://doi.org/10.18632/oncotarget.25351
Seo YS, Ko IO, Park H, Jeong YJ, Park JA, Kim KS, Park MJ, Lee HJ (2019) Radiation-induced changes in tumor vessels and microenvironment contribute to therapeutic resistance in glioblastoma. Front Oncol 9:1259. https://doi.org/10.3389/fonc.2019.01259
Lord CJ, Garrett MD, Ashworth A (2006) Targeting the double-strand DNA break repair pathway as a therapeutic strategy. Clin Cancer Res 12(15):4463–4468. https://doi.org/10.1158/1078-0432.CCR-06-1269
Sun J, Zhu Z, Li W, Shen M, Cao C, Sun Q, Guo Z, Liu L, Wu D (2020) UBE2T-regulated H2AX monoubiquitination induces hepatocellular carcinoma radioresistance by facilitating CHK1 activation. J Exp Clin Cancer Res 39(1):222. https://doi.org/10.1186/s13046-020-01734-4
Short SC, Giampieri S, Worku M, Alcaide-German M, Sioftanos G, Bourne S, Lio KI, Shaked-Rabi M, Martindale C (2011) Rad51 inhibition is an effective means of targeting DNA repair in glioma models and CD133+ tumor-derived cells. Neuro Oncol 13(5):487–499. https://doi.org/10.1093/neuonc/nor010
Laoukili J, Stahl M, Medema RH (2007) FoxM1: at the crossroads of ageing and cancer. Biochim Biophys Acta 1775(1):92–102. https://doi.org/10.1016/j.bbcan.2006.08.006
Nandi D, Cheema PS, Jaiswal N, Nag A (2018) FoxM1: repurposing an oncogene as a biomarker. Semin Cancer Biol 52(Pt 1):74–84. https://doi.org/10.1016/j.semcancer.2017.08.009
Song X, Fiati Kenston SS, Zhao J, Yang D, Gu Y (2017) Roles of FoxM1 in cell regulation and breast cancer targeting therapy. Med Oncol 34(3):41. https://doi.org/10.1007/s12032-017-0888-3
Zhang X, Zhang L, Du Y, Zheng H, Zhang P, Sun Y, Wang Y, Chen J, Ding P, Wang N et al (2017) A novel FOXM1 isoform, FOXM1D, promotes epithelial-mesenchymal transition and metastasis through ROCKs activation in colorectal cancer. Oncogene 36(6):807–819. https://doi.org/10.1038/onc.2016.249
Kelleher FC, O’Sullivan H (2016) FOXM1 in sarcoma: role in cell cycle, pluripotency genes and stem cell pathways. Oncotarget. 7(27):42792–42804. https://doi.org/10.18632/oncotarget.8669
Nicolau-Neto P, Palumbo A, De Martino M, Esposito F, de AlmeidaSimão T, Fusco A, Nasciutti LE, Da MeirelesCosta N, RibeiroPinto LF (2018) UBE2C is a transcriptional target of the cell cycle regulator FOXM1. Genes (Basel) 9:4. https://doi.org/10.3390/genes9040188
Wang K, Zhu X, Yao Y, Yang M, Zhou F, Zhu L (2018) Corosolic acid induces cell cycle arrest and cell apoptosis in human retinoblastoma Y-79 cells via disruption of MELK-FoxM1 signaling. Oncol Rep 39(6):2777–2786. https://doi.org/10.3892/or.2018.6339
Rajamanickam S, Panneerdoss S, Gorthi A, Timilsina S, Onyeagucha B, Kovalskyy D, Ivanov D, Hanes MA, Vadlamudi RK, Chen Y et al (2016) Inhibition of FoxM1-mediated DNA repair by imipramine blue suppresses breast cancer growth and metastasis. Clin Cancer Res 22(14):3524–3536. https://doi.org/10.1158/1078-0432.Ccr-15-2535
Gartel AL (2017) FOXM1 in cancer: interactions and vulnerabilities. Cancer Res 77(12):3135–3139. https://doi.org/10.1158/0008-5472.Can-16-3566
Liao GB, Li XZ, Zeng S, Liu C, Yang SM, Yang L, Hu CJ, Bai JY (2018) Regulation of the master regulator FOXM1 in cancer. Cell Commun Signal 16(1):57. https://doi.org/10.1186/s12964-018-0266-6
Bowman A, Nusse R (2011) Location, location, location: FoxM1 mediates β-catenin nuclear translocation and promotes glioma tumorigenesis. Cancer Cell 20(4):415–416. https://doi.org/10.1016/j.ccr.2011.10.003
Zhang C, Han X, Xu X, Zhou Z, Chen X, Tang Y, Cheng J, Moazzam NF, Liu F, Xu J et al (2018) FoxM1 drives ADAM17/EGFR activation loop to promote mesenchymal transition in glioblastoma. Cell Death Dis 9(5):469. https://doi.org/10.1038/s41419-018-0482-4
Ma Q, Liu Y, Shang L, Yu J, Qu Q (2017) The FOXM1/BUB1B signaling pathway is essential for the tumorigenicity and radioresistance of glioblastoma. Oncol Rep 38(6):3367–3375. https://doi.org/10.3892/or.2017.6032
Zhang N, Wu X, Yang L, Xiao F, Zhang H, Zhou A, Huang Z, Huang S (2012) FoxM1 inhibition sensitizes resistant glioblastoma cells to temozolomide by downregulating the expression of DNA-repair gene Rad51. Clin Cancer Res 18(21):5961–5971. https://doi.org/10.1158/1078-0432.CCR-12-0039
Im J, Lawrence J, Seelig D, Nho RS (2018) FoxM1-dependent RAD51 and BRCA2 signaling protects idiopathic pulmonary fibrosis fibroblasts from radiation-induced cell death. Cell Death Dis 9(6):584. https://doi.org/10.1038/s41419-018-0652-4
Komander D (2009) The emerging complexity of protein ubiquitination. Biochem Soc Trans 37(Pt 5):937–953. https://doi.org/10.1042/bst0370937
Komander D, Rape M (2012) The ubiquitin code. Annu Rev Biochem 81:203–229. https://doi.org/10.1146/annurev-biochem-060310-170328
Faktor J, Pjechová M, Hernychová L, Vojtěšek B (2019) Protein ubiquitination research in oncology. Klin Onkol 32(Supplementum 3):56–64. https://doi.org/10.14735/amko20193S
Karunarathna U, Kongsema M, Zona S, Gong C, Cabrera E, Gomes AR, Man EP, Khongkow P, Tsang JW, Khoo US et al (2016) OTUB1 inhibits the ubiquitination and degradation of FOXM1 in breast cancer and epirubicin resistance. Oncogene 35(11):1433–1444. https://doi.org/10.1038/onc.2015.208
Kongsema M, Zona S, Karunarathna U, Cabrera E, Man EP, Yao S, Shibakawa A, Khoo US, Medema RH, Freire R et al (2016) RNF168 cooperates with RNF8 to mediate FOXM1 ubiquitination and degradation in breast cancer epirubicin treatment. Oncogenesis 5(8):e252. https://doi.org/10.1038/oncsis.2016.57
Acknowledgements
Not applicable
Funding
This work was supported by the National Natural Science Foundation of China (31700932), the National Natural Science Foundation of China (81973531), the National Natural Science Foundation of China (82073937), Shenzhen Science and Technology Project (JCYJ20180228175059744), and Shenzhen Key Medical Discipline Construction Fund (SZXK059). Shenzhen Science and Technology Innovation Committee for Basic Research (Grant No. JCYJ20170818144044876).
Author information
Authors and Affiliations
Contributions
Yun Dong and Yiping Xiong designed the study and supervised the data collection, Duanyang Zhou analyzed the data and interpreted the data, and Min Yao, Xiao Wang, Wenchuan Bi, and Jian Zhang prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the standards upheld by the Ethics Committee of Shenzhen University and with those of the 1964 Helsinki Declaration and its later amendments for ethical research involving human subjects. All animal experiments were approved by the Ethics Committee of Shenzhen University for the use of animals and conducted in accordance with the National Institutes of Health Laboratory Animal Care and Use Guidelines.
Consent to Participate
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dong, Y., Xiong, Y., Zhou, D. et al. TRIM56 Reduces Radiosensitization of Human Glioblastoma by Regulating FOXM1-Mediated DNA Repair. Mol Neurobiol 59, 5312–5325 (2022). https://doi.org/10.1007/s12035-022-02898-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-02898-0