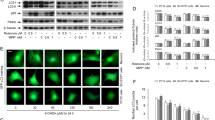
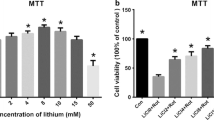
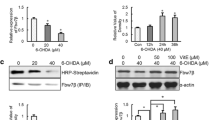
Abstract
Parkinson’s disease (PD) is a common movement disorder among neurodegenerative diseases, involving neuronal cell death in the substantia nigra of the midbrain. Although mechanisms of cell death in PD have been studied, the exact molecular pathogenesis is still unclear. Here, we explore the relationship between two types of cell death, autophagy and apoptosis, which have been studied separately in parkinsonian mimetic model of 6-hydroxydopamine (6-OHDA). 6-OHDA induced autophagy firstly and then later inhibition of autophagy flux occurred with apoptosis. The apoptosis was prevented by treatment of pan-caspase inhibitor, zVAD-fmk (benzyloxycarbonyl-VAD-fluoromethylketone (zVAD)), or early phase inhibitor of autophagy, 3-methyladenine (3-MA), indicating that autophagic induction was followed by the apoptosis. Interestingly, late step inhibitor of autophagy, bafilomycin A1 (BafA), aggravated 6-OHDA-induced apoptosis. This was associated with mitochondrial abnormality such as the inhibition of damaged mitochondrial clearance and aberrant increase of extracellular oxygen consumption. Furthermore, treatment of BafA did not inhibit 6-OHDA-mediated superoxide formation but strongly reduced the hydrogen peroxide production to below basal levels, indicating failure from superoxide to hydrogen peroxide. These results were accompanied by a lowered expression and activity of copper/zinc superoxide dismutase (Cu/Zn-SOD) but not of manganese SOD (MnSOD) and catalase. Thus, the present study suggests that crosstalk among apoptosis, autophagy, and oxidative stress is a causative factor of 6-OHDA-induced neuronal death and provides a mechanistic understanding of PD pathogenesis.







Similar content being viewed by others
Abbreviations
- 3-MA:
-
3-Methyladenine
- 6-OHDA:
-
6-Hydroxydopamine
- AnV:
-
Annexin V
- BafA:
-
Bafilomycin A1
- COXIV:
-
Cytochrome C oxidase subunit IV
- DCF-DA:
-
Dichlorodihydrofluorescein diacetate
- DHE:
-
Dihydroethidium
- DMSO:
-
Dimethylsulfoxide
- FACS:
-
Fluorescence-activated cell sorting
- GD:
-
Glucose deprivation
- LC3:
-
Microtubule-associated protein 1A/1B-light chain 3
- LDH:
-
Lactate dehydrogenase
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PD:
-
Parkinson’s disease
- PI:
-
Propidium iodide
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- tBID:
-
Truncated BH3-interacting domain death agonist
- TNFa:
-
Tumor necrosis factor alpha
- zVAD:
-
Benzyloxycarbonyl-VAD-fluoromethylketone
References
Shults CW (2003) Treatments of Parkinson disease: circa 2003. Arch Neurol 60:1680–1684. doi:10.1001/archneur.60.12.1680
Eriksen JL, Wszolek Z, Petrucelli L (2005) Molecular pathogenesis of Parkinson disease. Arch Neurol 62:353–357. doi:10.1001/archneur.62.3.353
Ross CA, Smith WW (2007) Gene-environment interactions in Parkinson’s disease. Parkinsonism Relat Disord 13:S309–S315. doi:10.1016/S1353-8020(08)70022-1
Henchcliffe C, Beal MF (2008) Mitochondrial biology and oxidative stress in Parkinson disease pathogenesis. Nat Clin Pract Neurol 4:600–609. doi:10.1038/ncpneuro0924
Zabel C, Nguyen HP, Hin SC, Hartl D, Mao L, Klose J (2010) Proteasome and oxidative phosphorylation changes may explain why aging is a risk factor for neurodegenerative disorders. J Proteomics 73:2230–2238. doi:10.1016/j.jprot.2010.08.008
Cheung ZH, Ip NY (2009) The emerging role of autophagy in Parkinson’s disease. Mol Brain 16:2–29. doi:10.1186/1756-6606-2-29
Blandini F, Armentero MT, Martignoni E (2008) The 6-hydroxydopamine model: news from the past. Parkinsonism Relat Disord 14:S124–S129. doi:10.1016/j.parkreldis.2008.04.015
Chandra J, Samali A, Orrenius S (2000) Triggering and modulation of apoptosis by oxidative stress. Free Radic Biol Med 29:323–333
Zhou C, Huang Y, Przedborski S (2008) Oxidative stress in Parkinson’s disease: a mechanism of pathogenic and therapeutic significance. Ann N Y Acad Sci 1147:93–104. doi:10.1196/annals.1427.023
Blum D, Torch S, Lambeng N, Nissou M, Benabid AL, Sadoul R, Verna JM (2001) Molecular pathways involved in the neurotoxicity of 6-OHDA, dopamine and MPTP: contribution to the apoptotic theory in Parkinson’s disease. Prog Neurobiol 65:135–172
Venderova K, Park DS (2012) Programmed cell death in Parkinson’s disease. Cold Spring Harb Perspect Med 2:a009365. doi:10.1101/cshperspect.a009365
Yang Q, Mao Z (2010) Parkinson disease: a role for autophagy? Neuroscientist 16:335–341. doi:10.1177/1073858409357118
Dagda RK, Zhu J, Kulich SM, Chu CT (2008) Mitochondrially localized ERK2 regulates mitophagy and autophagic cell stress: implications for Parkinson’s disease. Autophagy 4:770–782
Jang BG, Choi BY, Kim JH, Kim MJ, Sohn M, Suh SW (2013) Impairment of autophagic flux promotes glucose reperfusion-induced neuro2A cell death after glucose deprivation. PLoS One 8:e76466. doi:10.1371/journal.pone.0076466
Liu Y, Wong TP, Aarts M, Rooyakkers A, Liu L, Lai TW, Wu DC, Lu J, Tymianski M, Craig AM, Wang YT (2007) NMDA receptor subunits have differential roles in mediating excitotoxic neuronal death both in vitro and in vivo. J Neurosci 27:2846–2857. doi:10.1523/JNEUROSCI. 0116-07.2007
Puissant A, Fenouille N, Auberger P (2012) When autophagy meets cancer through p62/SQSTM1. Am J Cancer Res 2:397–413
Klionsky DJ, Abdalla FC, Abeliovich H et al (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8:445–544
Zhao H, Kalivendi S, Zhang H, Joseph J, Nithipatikom K, Vasquez-Vivar J, Kalyanaraman B (2003) Superoxide reacts with hydroethidine but forms a fluorescent product that is distinctly different from ethidium: potential implications in intracellular fluorescence detection of superoxide. Free Radic Biol Med 34:1359–1368
Gomes A, Fernandes E, Lima JL (2005) Fluorescence probes used for detection of reactive oxygen species. J Biochem Biophys Methods 65:45–80. doi:10.1016/j.jbbm.2005.10.003
Arsikin K, Kravic-Stevovic T, Jovanovic M, Ristic B, Tovilovic G, Zogovic N, Bumbasirevic V, Trajkovic V, Harhaji-Trajkovic L (2012) Autophagy-dependent and -independent involvement of AMP-activated protein kinase in 6-hydroxydopamine toxicity to SH-SY5Y neuroblastoma cells. Biochim Biophys Acta 1822:1826–1836. doi:10.1016/j.bbadis.2012.08.006
Ossola B, Lantto TA, Puttonen KA, Tuominen RK, Raasmaja A, Mannisto PT (2012) Minocycline protects SH-SY5Y cells from 6-hydroxydopamine by inhibiting both caspase-dependent and -independent programmed cell death. J Neurosci Res 90:682–690. doi:10.1002/jnr.22791
Chu CT, Zhu J, Dagda R (2007) Beclin 1-independent pathway of damage-induced mitophagy and autophagic stress: implications for neurodegeneration and cell death. Autophagy 3:663–666
Dagda RK, Zhu J, Kulich SM, Chu CT (2008) Mitochondrially localized ERK2 regulates mitophagy and autophagic cell stress: implications for Parkinson’s disease. Autophagy 4:770–782
Van Humbeeck C, Cornelissen T, Hofkens H, Mandemakers W, Gevaert K, De Strooper B, Vandenberghe W (2011) Parkin interacts with Ambra1 to induce mitophagy. J Neurosci 31:10249–10261. doi:10.1523/JNEUROSCI. 1917-11.2011
Kim SJ, Syed GH, Siddiqui A (2013) Hepatitis C virus induces the mitochondrial translocation of Parkin and subsequent mitophagy. PLoS Pathog 9:e1003285. doi:10.1371/journal.ppat.1003285
Lemasters JJ (2014) Variants of mitochondrial autophagy: types 1 and 2 mitophagy and micromitophagy (type 3). Redox Biol 2:749–754. doi:10.1016/j.redox.2014.06.004
Hailey DW, Rambold AS, Satpute-Krishnan P, Mitra K, Sougrat R, Kim PK, Lippincott-Schwartz J (2010) Mitochondria supply membranes for autophagosome biogenesis during starvation. Cell 141:656–667. doi:10.1016/j.cell.2010.04.009
Parone PA, James DI, Da Cruz S, Mattenberger Y, Donzé O, Barja F, Martinou JC (2006) Inhibiting the mitochondrial fission machinery does not prevent Bax/Bak-dependent apoptosis. Mol Cell Biol 26:7397–7408. doi:10.1128/MCB. 02282-05
Schotte P, Declercq W, Van Huffel S, Vandenabeele P, Beyaert R (1999) Non-specific effects of methyl ketone peptide inhibitors of caspases. FEBS Lett 442:117–121
Nylandsted J, Gyrd-Hansen M, Danielewicz A, Fehrenbacher N, Lademann U, Høyer-Hansen M, Weber E, Multhoff G, Rohde M, Jäättelä M (2004) Heat shock protein 70 promotes cell survival by inhibiting lysosomal membrane permeabilization. J Exp Med 200:425–435. doi:10.1084/jem.20040531
Uchiyama A, Kim JS, Kon K, Jaeschke H, Ikejima K, Watanabe S, Lemasters JJ (2008) Translocation of iron from lysosomes into mitochondria is a key event during oxidative stress-induced hepatocellular injury. Hepatology 48:1644–1654. doi:10.1002/hep.22498
Liu B, Yang P, Ye Y, Zhou Y, Li L, Tashiro S, Onodera S, Ikejima T (2011) Role of ROS in the protective effect of silibinin on sodium nitroprusside-induced apoptosis in rat pheochromocytoma PC12 cells. Free Radic Res 45:835–847. doi:10.3109/10715762.2011.580343
Ramírez C, Pham K, Franco MF, Chwa M, Limb A, Kuppermann BD, Kenney MC (2013) Hydroquinone induces oxidative and mitochondrial damage to human retinal Müller cells (MIO-M1). Neurotoxicology 39:102–108. doi:10.1016/j.neuro.2013.08.004
Ricci JE, Gottlieb RA, Green DR (2003) Caspase-mediated loss of mitochondrial function and generation of reactive oxygen species during apoptosis. J Cell Biol 160:65–75. doi:10.1083/jcb.200208089
Cowburn AS, White JF, Deighton J, Walmsley SR, Chilvers ER (2005) z-VAD-fmk augmentation of TNF alpha-stimulated neutrophil apoptosis is compound specific and does not involve the generation of reactive oxygen species. Blood 105:2970–2972. doi:10.1182/blood-2004-07-2870
Rikiishi H (2012) Novel insights into the interplay between apoptosis and autophagy. Int J Cell Biol 2012:317645. doi:10.1155/2012/317645
Li H, Horke S, Förstermann U (2013) Oxidative stress in vascular disease and its pharmacological prevention. Trends Pharmacol Sci 34:313–319. doi:10.1016/j.tips.2013.03.007
Miao L, St Clair DK (2009) Regulation of superoxide dismutase genes: implications in disease. Free Radic Biol Med 47:344–356. doi:10.1016/j.freeradbiomed.2009.05.018
Furukawa Y, Torres AS, O’Halloran TV (2004) Oxygen-induced maturation of SOD1: a key role for disulfide formation by the copper chaperone CCS. EMBO J 23:2872–2881. doi:10.1038/sj.emboj.7600276
Banci L, Bertini I, Cantini F, Kozyreva T, Massagni C, Palumaa P, Rubino JT, Zovo K (2012) Human superoxide dismutase 1 (hSOD1) maturation through interaction with human copper chaperone for SOD1 (hCCS). Proc Natl Acad Sci U S A 109:13555–13660. doi:10.1073/pnas.1207493109
Di Noto L, Whitson LJ, Cao X, Hart PJ, Levine RL (2005) Proteasomal degradation of mutant superoxide dismutases linked to amyotrophic lateral sclerosis. J Biol Chem 280:39907–39913. doi:10.1074/jbc.M506247200
Fei E, Jia N, Yan M, Ying Z, Sun Q, Wang H, Zhang T, Ma X, Ding H, Yao X, Shi Y, Wang G (2006) SUMO-1 modification increases human SOD1 stability and aggregation. Biochem Biophys Res Commun 347:406–412. doi:10.1016/j.bbrc.2006.06.092
Ezzi SA, Urushitani M, Julien JP (2007) Wild-type superoxide dismutase acquires binding and toxic properties of ALS-linked mutant forms through oxidation. J Neurochem 102:170–178. doi:10.1111/j.1471-4159.2007.04531.x
Acknowledgments
We thank Dr. Daniel Principe for editing the language of the manuscript. This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2014R1A1A1002109). This research was also supported by the Hallym University Specialization Fund (HRF-S-51).
Conflict of Interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Sua In and Chang-Won Hong contributed equally to this work.
Rights and permissions
About this article
Cite this article
In, S., Hong, CW., Choi, B. et al. Inhibition of Mitochondrial Clearance and Cu/Zn-SOD Activity Enhance 6-Hydroxydopamine-Induced Neuronal Apoptosis. Mol Neurobiol 53, 777–791 (2016). https://doi.org/10.1007/s12035-014-9087-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9087-9