Abstract
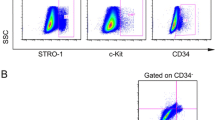
To find out differences and similarities in phenotypic, proliferative, and trans-differentiation properties of stem cells isolated from pulp of deciduous (SHEDs) and permanent (DPSCs) teeth with human bone marrow stem cells (BMSCs), we examined the expression of mesenchymal and embryonic stem cell markers in relation to the proliferation and osteogenic differentiation potentials of these cells. In this way, after isolating SHEDs, DPSCs, and BMSCs, cell proliferation was evaluated and population doubling time was calculated accordingly. Expression patterns of mesenchymal, hematopoietic, and embryonic stem cell markers were assessed followed by examining differentiation potential toward osseous tissue through alizarin red staining and qRT-PCR. Based on the results, the proliferation rates of SHEDs and DPSCs were significantly higher than that of BMSCs (P < 0.0001). High expression of mesenchymal stem cell markers and weak expression of hematopoietic markers were observed in all the three groups. The mean expression of OCT-4 was significantly higher in SHEDs and DPSCs (P = 0.028), while the expression of SSEA-4 was lower (P = 0.006) compared to BMSCs. Osteogenic differentiation potential of SHEDs was greater than DPSCs; however, it was lower than that of BMSCs. Conclusively, the distinctive immunophenotyping, proliferation rate, and differentiation pattern of SHEDs and DPSCs discriminate these cells from BMSCs. Furthermore, dissimilarity in differentiation potential is evidence implying that SHEDs might be more primitive stem cell population compared to DPSCs.







Similar content being viewed by others

References
Friedenstein, A. J., Gorskaja, J. F., & Kulagina, N. N. (1976). Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Experimental Hematology, 4, 267–274.
Bissels, U., Eckardt, D., & Bosio, A. (2013). In G. Steinhoff (Ed.), Regenerative medicine from protocol to patient (2nd ed., pp. 155–176). The Netherlands: Springer.
Estrela, C., Alencar, A. H., Kitten, G. T., Vencio, E. F., & Gava, E. (2011). Mesenchymal stem cells in the dental tissues: Perspectives for tissue regeneration. Brazilian Dental Journal, 22, 91–98.
Gronthos, S., Mankani, M., Brahim, J., Robey, P. G., & Shi, S. (2000). Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proceedings of the National Academy of Sciences of the United States of America, 97, 13625–13630.
d’Aquino, R., De Rosa, A., Laino, G., Caruso, F., Guida, L., Rullo, R., et al. (2009). Human dental pulp stem cells: from biology to clinical applications. Journal of Experimental Zoology Part B: Molecular and Developmental Evolution, 312B, 408–415.
Gronthos, S., Brahim, J., Li, W., Fisher, L. W., Cherman, N., Boyde, A., et al. (2002). Stem cell properties of human dental pulp stem cells. Journal of Dental Research, 81, 531–535.
Mezey, E., Chandross, K. J., Harta, G., Maki, R. A. & McKercher, S. R. (2000). Turning blood into brain: cells bearing neuronal antigens generated in vivo from bone marrow. Science (New York, NY), 290, 1779–1782.
Mezey, E., Key, S., Vogelsang, G., Szalayova, I., Lange, G. D., & Crain, B. (2003). Transplanted bone marrow generates new neurons in human brains. Proceedings of the National Academy of Sciences of the United States of America, 100, 1364–1369.
Santos, D. M. (2011). Genetic engineering: Recent developments in applications (1st edn). New York: CRC Press.
Kamota, T., Li, T. S., Morikage, N., Murakami, M., Ohshima, M., Kubo, M., et al. (2009). Ischemic pre-conditioning enhances the mobilization and recruitment of bone marrow stem cells to protect against ischemia/reperfusion injury in the late phase. Journal of the American College of Cardiology, 53, 1814–1822.
Pituch-Noworolska, A., Majka, M., Janowska-Wieczorek, A., Baj-Krzyworzeka, M., Urbanowicz, B., Malec, E., & Ratajczak, M. Z. (2003). Circulating CXCR4-positive stem/progenitor cells compete for SDF-1-positive niches in bone marrow, muscle and neural tissues: An alternative hypothesis to stem cell plasticity. Folia Histochemica et Cytobiologica, 41, 13–21.
Zhou, S. B., Wang, J., Chiang, C. A., Sheng, L. L., & Li, Q. F. (2013). Mechanical stretch upregulates SDF-1alpha in skin tissue and induces migration of circulating bone marrow-derived stem cells into the expanded skin. Stem Cells, 31, 2703–2713.
Deschaseaux, F., Sensébé, L., & Heymann, D. (2009). Mechanisms of bone repair and regeneration. Trends in Molecular Medicine, 15, 417–429.
Takahashi, K. (1992). Pulpal vascular changes in inflammation. Proceedings of the Finnish Dental Society, 88(Suppl 1), 381–385.
Karaoz, E., Demircan, P. C., Saglam, O., Aksoy, A., Kaymaz, F., & Duruksu, G. (2011). Human dental pulp stem cells demonstrate better neural and epithelial stem cell properties than bone marrow-derived mesenchymal stem cells. Histochemistry and Cell Biology, 136, 455–473.
Tamaki, Y., Nakahara, T., Ishikawa, H., & Sato, S. (2013). In vitro analysis of mesenchymal stem cells derived from human teeth and bone marrow. Odontology, 101, 121–132.
Mead, B., Berry, M., Logan, A., Scott, R. A., Leadbeater, W., & Scheven, B. A. (2015). Stem cell treatment of degenerative eye disease. Stem Cell Research, 14, 243–257. doi:10.1016/j.scr.2015.02.003. Epub 2015 Feb 24.
Jussila, M., & Thesleff, I. (2012). Signaling networks regulating tooth organogenesis and regeneration, and the specification of dental mesenchymal and epithelial cell lineages. Cold Spring Harbor Perspectives in Biology, 4, a008425.
Miura, M., Gronthos, S., Zhao, M., Lu, B., Fisher, L. W., Robey, P. G., & Shi, S. (2003). SHED: Stem cells from human exfoliated deciduous teeth. Proceedings of the National Academy of Sciences of the United States of America, 100, 5807–5812.
Majumdar, M. K., Thiede, M. A., Mosca, J. D., Moorman, M., & Gerson, S. L. (1998). Phenotypic and functional comparison of cultures of marrow-derived mesenchymal stem cells (MSCs) and stromal cells. Journal of Cellular Physiology, 176, 57–66.
Kazemnejad, S., Allameh, A., Soleimani, M., Gharehbaghian, A., Mohammadi, Y., Amirizadeh, N., & Jazayery, M. (2009). Biochemical and molecular characterization of hepatocyte-like cells derived from human bone marrow mesenchymal stem cells on a novel three-dimensional biocompatible nanofibrous scaffold. Journal of Gastroenterology and Hepatology, 24, 278–287.
Marycz, K., Smieszek, A., Grzesiak, J., Siudzinska, A., Maredziak, M., Donesz-Sikorska, A., & Krzak, J. (2015). The osteogenic properties of multipotent mesenchymal stromal cells in cultures on TiO2 sol–gel-derived biomaterial. BioMed Research International, 2015, 651097.
Lotfy, A., Salama, M., Zahran, F., Jones, E., Badawy, A., & Sobh, M. (2014). Characterization of mesenchymal stem cells derived from rat bone marrow and adipose tissue: A comparative study. International Journal of Stem Cells, 7, 135–142.
Ruth, V. (2006). http://www.doubling-time.com/compute.php. Accessed 10 Feb 2015.
Ruijter, J. M., Ramakers, C., Hoogaars, W. M., Karlen, Y., Bakker, O., van den Hoff, M. J., & Moorman, A. F. (2009). Amplification efficiency: Linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Research, 37, e45.
Pfaffl, M. W., Horgan, G. W., & Dempfle, L. (2002). Relative expression software tool (REST©) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Research, 30, e36.
Tirino, V., Paino, F., De Rosa, A., & Papaccio, G. (2012). Identification, isolation, characterization, and banking of human dental pulp stem cells. Methods in Molecular Biology (Clifton, N.J.), 879, 443–463.
Shi, S., Robey, P. G., & Gronthos, S. (2001). Comparison of human dental pulp and bone marrow stromal stem cells by cDNA microarray analysis. Bone, 29, 532–539.
Nanci, A. (2012). Ten cate’s oral histology: Development, structure, and function (8th edn.). Missouri: Mosby.
Dominici, M., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F., Krause, D., et al. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy, 8, 315–317.
Scuteri, A., Donzelli, E., Foudah, D., Caldara, C., Redondo, J., D’Amico, G., et al. (2014). Mesengenic differentiation: Comparison of human and rat bone marrow mesenchymal stem cells. International Journal of Stem Cells, 7, 127–134.
Jones, E. A., Kinsey, S. E., English, A., Jones, R. A., Straszynski, L., Meredith, D. M., et al. (2002). Isolation and characterization of bone marrow multipotential mesenchymal progenitor cells. Arthritis and Rheumatism, 46, 3349–3360.
Cheng, P. H., Snyder, B., Fillos, D., Ibegbu, C. C., Huang, A. H., & Chan, A. W. (2008). Postnatal stem/progenitor cells derived from the dental pulp of adult chimpanzee. BMC Cell Biology, 9, 20.
Dissanayaka, W. L., Zhu, X., Zhang, C., & Jin, L. (2011). Characterization of dental pulp stem cells isolated from canine premolars. Journal of Endodontics, 37, 1074–1080.
Ferro, F., Spelat, R., D’Aurizio, F., Puppato, E., Pandolfi, M., Beltrami, A. P., et al. (2012). Dental pulp stem cells differentiation reveals new insights in Oct4A dynamics. PLoS ONE, 7, e41774.
Ma, D., Gao, J., Yue, J., Yan, W., Fang, F., & Wu, B. (2012). Changes in proliferation and osteogenic differentiation of stem cells from deep caries in vitro. Journal of Endodontics, 38, 796–802.
Huang, A. H., Chen, Y. K., Chan, A. W., Shieh, T. Y., & Lin, L. M. (2009). Isolation and characterization of human dental pulp stem/stromal cells from nonextracted crown-fractured teeth requiring root canal therapy. Journal of Endodontics, 35, 673–681.
Bakopoulou, A., Leyhausen, G., Volk, J., Tsiftsoglou, A., Garefis, P., Koidis, P., & Geurtsen, W. (2011). Assessment of the impact of two different isolation methods on the osteo/odontogenic differentiation potential of human dental stem cells derived from deciduous teeth. Calcified Tissue International, 88, 130–141.
Bakopoulou, A., Leyhausen, G., Volk, J., Tsiftsoglou, A., Garefis, P., Koidis, P., & Geurtsen, W. (2011). Comparative analysis of in vitro osteo/odontogenic differentiation potential of human dental pulp stem cells (DPSCs) and stem cells from the apical papilla (SCAP). Archives of Oral Biology, 56, 709–721.
Bernardi, L., Luisi, S. B., Fernandes, R., Dalberto, T. P., Valentim, L., Bogo Chies, J. A., et al. (2011). The isolation of stem cells from human deciduous teeth pulp is related to the physiological process of resorption. Journal of endodontics, 37, 973–979.
Gang, E. J., Bosnakovski, D., Figueiredo, C. A., Visser, J. W., & Perlingeiro, R. C. (2007). SSEA-4 identifies mesenchymal stem cells from bone marrow. Blood, 109, 1743–1751.
Riekstina, U., Cakstina, I., Parfejevs, V., Hoogduijn, M., Jankovskis, G., Muiznieks, I., et al. (2009). Embryonic stem cell marker expression pattern in human mesenchymal stem cells derived from bone marrow, adipose tissue, heart and dermis. Stem Cell Reviews, 5, 378–386.
Davies, O. G., Cooper, P. R., Shelton, R. M., Smith, A. J., & Scheven, B. A. (2015). A comparison of the in vitro mineralisation and dentinogenic potential of mesenchymal stem cells derived from adipose tissue, bone marrow and dental pulp. Journal of Bone and Mineral Metabolism, 33, 371–382. doi:10.1007/s00774-014-0601-y. Epub 2014 Jul 6.
Kern, S., Eichler, H., Stoeve, J., Kluter, H., & Bieback, K. (2006). Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells, 24, 1294–1301.
Martland, M., & Robison, R. (1924). The possible significance of hexosephosphoric esters in ossification: Part V. The enzyme in the early stages of bone development. Biochemical Journal, 18, 1354–1357.
Robison, R., Macleod, M., & Rosenheim, A. H. (1930). The possible significance of hexosephosphoric esters in ossification: Calcification in vitro. Biochemical Journal, 24, 1927–1941.
Bellows, C. G., Aubin, J. E., & Heersche, J. N. M. (1991). Initiation and progression of mineralization of bone nodules formed in vitro: The role of alkaline phosphatase and organic phosphate. Bone and Mineral, 14, 27–40.
Park, S. J., Bae, H. S., & Park, J. C. (2015). Osteogenic differentiation and gene expression profile of human dental follicle cells induced by human dental pulp cells. Journal of Molecular Histology, 46, 93–106.
Ducy, P. (2000). Cbfa1: A molecular switch in osteoblast biology. Developmental Dynamics, 219, 461–471.
Viereck, V., Siggelkow, H., Tauber, S., Raddatz, D., Schutze, N., & Hüfner, M. (2002). Differential regulation of Cbfa1/Runx2 and osteocalcin gene expression by vitamin-D3, dexamethasone, and local growth factors in primary human osteoblasts. Journal of Cellular Biochemistry, 86, 348–356.
Calvi, L. M., Sims, N. A., Hunzelman, J. L., Knight, M. C., Giovannetti, A., Saxton, J. M., et al. (2001). Activated parathyroid hormone/parathyroid hormone-related protein receptor in osteoblastic cells differentially affects cortical and trabecular bone. Journal of Clinical Investigation, 107, 277–286.
Koyama, N., Okubo, Y., Nakao, K., & Bessho, K. (2009). Evaluation of pluripotency in human dental pulp cells. Journal of Oral and Maxillofacial Surgery, 67, 501–506.
Ponnaiyan, D., & Jegadeesan, V. (2014). Comparison of phenotype and differentiation marker gene expression profiles in human dental pulp and bone marrow mesenchymal stem cells. European Journal of Dentistry, 8, 307–313.
Acknowledgments
The authors would like to thank Ms Zahra Ghaempanah for her invaluable assistance.
Source of Financial Support
This research was funded and supported by a grant from Tehran University of Medical Sciences (TUMS); Grant No. 92-02-69-19971 and Avicenna Research Institute. No external funding, apart from the support of the authors’ institutions, was available for this study. TUMS and Avicenna Research Institute had no involvement in the study design, data collection, analysis and interpretation, writing of the report, manuscript preparation, or the decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest and they do not have any financial or personal relationships with other people or organizations that could inappropriately influence (bias) the work.
Informed Consent
All samples were collected under a protocol approved by the Medical Ethics Committee of Tehran University of Medical Sciences (TUMS) and Avicenna Research Institute. Informed consent was obtained from the donors and/or parents of the donors according to guidelines of the Medical Ethics Committee, Ministry of Health, I.R. of Iran. The extraction of teeth and bone marrow aspiration was in the treatment plan of donors. People involved in diagnosis and treatment planning had no participation in the present study.
Additional information
Manijeh Khanmohammadi and Sayeh Khanjani have contributed equally.
Rights and permissions
About this article
Cite this article
Aghajani, F., Hooshmand, T., Khanmohammadi, M. et al. Comparative Immunophenotypic Characteristics, Proliferative Features, and Osteogenic Differentiation of Stem Cells Isolated from Human Permanent and Deciduous Teeth with Bone Marrow. Mol Biotechnol 58, 415–427 (2016). https://doi.org/10.1007/s12033-016-9941-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-016-9941-2