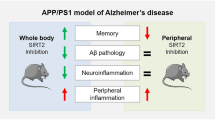
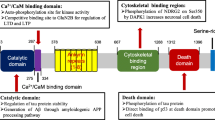
Abstract
Alzheimer’s disease (AD) is a neurodegenerative disease that is imposing an increasing burden on society. Currently, AD is the leading cause of senile dementia worldwide. Despite the long existence of AD, there is lack of therapies for AD, suggesting that new and effective treatment strategy must be explored. At present, sirtuin pathway has attracted attention from the researchers due to its promising results in laboratory models of aging. In addition, our understanding in the roles of sirtuin 6 in AD has expanded. It has been identified to be involved in telomere maintenance, DNA repair, genome integrity, energy metabolism, and inflammation, which ultimately regulate life span. Recent findings also demonstrate that sirtuin 6 is lacking in AD patients, proposing that it can be a new potential therapeutic target in AD. Therefore, exploring on how sirtuin 6 is related in AD manifestation may accelerate the research of AD further and benefits future AD patients. Keeping that in mind, this review aims to highlight the possible roles of sirtuin 6 in AD manifestation.

Similar content being viewed by others
References
Anand R, Gill KD, Mahdi AA (2014) Therapeutics of Alzheimer’s disease: past, present and future. Neuropharmacology 76:27–50. https://doi.org/10.1016/j.neuropharm.2013.07.004
Anekonda TS, Reddy PH (2006) Neuronal protection by sirtuins in Alzheimer’s disease. J Neurochem 96(2):305–313. https://doi.org/10.1111/j.1471-4159.2005.03492.x
Alzheimer’s Association (2011) Alzheimer’s disease facts and figures. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association 7:208
Alzheimer’s Association (2013) Alzheimer’s disease facts and figures. Alzheimers Dement 9:208–245
Alzheimer’s Association (2016) What is Alzheimer’s? http://wwwalzorg/alzheimers_disease_what_is_alzheimersasp Accessed 1 March 2016 2016
Baur JA, Ungvari Z, Minor RK, Le Couteur DG, De Cabo R (2012) Are sirtuins viable targets for improving healthspan and lifespan? Nat Rev Drug Discov 11(6):443–461. https://doi.org/10.1038/nrd3738
Bonda DJ, Lee HG, Camins A, Pallàs M, Casadesus G, Smith MA, Zhu X (2011) The sirtuin pathway in ageing and Alzheimer disease: mechanistic and therapeutic considerations. The Lancet Neurology 10(3):275–279. https://doi.org/10.1016/S1474-4422(11)70013-8
Castellani RJ, Rolston RK, Smith MA (2010) Alzheimer disease. Dis Mon 56(9):484–546. https://doi.org/10.1016/j.disamonth.2010.06.001
Chen J, Zhou Y, Mueller-Steiner S, Chen LF, Kwon H, Yi S, Mucke L, Gan L (2005) SIRT1 protects against microglia-dependent amyloid-β toxicity through inhibiting NF-κB signaling. J Biol Chem 280(48):40364–40374. https://doi.org/10.1074/jbc.M509329200
Chen L, Huang S, Lee L, Davalos A, Schiestl RH, Campisi J, Oshima J (2003) WRN, the protein deficient in Werner syndrome, plays a critical structural role in optimizing DNA repair. Aging Cell 2(4):191–199. https://doi.org/10.1046/j.1474-9728.2003.00052.x
Chiang K, Koo EH (2014) Emerging therapeutics for Alzheimer’s disease. Annu Rev Pharmacol Toxicol 54(1):381–405. https://doi.org/10.1146/annurev-pharmtox-011613-135932
Colombo A, Repici M, Pesaresi M, Santambrogio S, Forloni G, Borsello T (2007) The TAT-JNK inhibitor peptide interferes with beta amyloid protein stability. Cell Death & Differentiation 14(10):1845–1848. https://doi.org/10.1038/sj.cdd.4402202
Dali-Youcef N, Lagouge M, Froelich S, Koehl C, Schoonjans K, Auwerx J (2007) Sirtuins: the ‘magnificent seven’, function, metabolism and longevity. Ann Med 39(5):335–345. https://doi.org/10.1080/07853890701408194
Dias-Santagata D, Fulga TA, Duttaroy A, Feany MB (2007) Oxidative stress mediates tau-induced neurodegeneration in Drosophila. J Clin Investig 117(1):236–245. https://doi.org/10.1172/JCI28769
Do Carmo S, Cuello AC (2013) Modeling Alzheimer’s disease in transgenic rats. Mol Neurodegener 8(1):37. https://doi.org/10.1186/1750-1326-8-37
Etchegaray J-P, Zhong L, Mostoslavsky R (2013) The histone deacetylase SIRT6: at the crossroads between epigenetics, metabolism and disease. Curr Top Med Chem 13(23):2991–3000. https://doi.org/10.2174/15680266113136660213
Franco S, Blasco MA, Siedlak SL, Harris PL, Moreira PI, Perry G, Smith MA (2006) Telomeres and telomerase in Alzheimer’s disease: Epiphenomena or a new focus for therapeutic strategy? Alzheimers Dement 2(3):164–168
Gella A, Durany N (2009) Oxidative stress in Alzheimer disease. Cell Adhes Migr 3(1):88–93. https://doi.org/10.4161/cam.3.1.7402
Harley CB, Futcher AB, Greider CW (1990) Telomeres shorten during ageing of human fibroblasts. Nature 345(6274):458–460
Hashimoto Y, Tsuji O, Niikura T, Yamagishi Y, Ishizaka M, Kawasumi M, Chiba T, Kanekura K, Yamada M, Tsukamoto E, Kouyama K, Terashita K, Aiso S, Lin A, Nishimoto I (2003) Involvement of c-Jun N-terminal kinase in amyloid precursor protein-mediated neuronal cell death. J Neurochem 84(4):864–877. https://doi.org/10.1046/j.1471-4159.2003.01585.x
Hippius H, Neundorfer G (2003) The discovery of Alzheimer’s disease. Dialogues Clin Neurosci 5(1):101–108
Holmquist GP (1998) Endogenous lesions, S-phase-independent spontaneous mutations, and evolutionary strategies for base excision repair. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 400(1-2):59–68. https://doi.org/10.1016/S0027-5107(98)00051-7
Jung ES, Choi H, Song H, Hwang YJ, Kim A, Ryu H, Mook-Jung I (2016) p53-dependent SIRT6 expression protects Aβ42-induced DNA damage. Sci Rep 6
Kaidi A, Weinert BT, Choudhary C, Jackson SP (2010) Human SIRT6 promotes DNA end resection through CtIP deacetylation. Science 329(5997):1348–1353. https://doi.org/10.1126/science.1192049
Kaluski S, Portillo M, Besnard A, Stein D, Einav M, Zhong L, Ueberham U, Arendt T, Mostoslavsky R, Sahay A, Toiber D (2017) Neuroprotective functions for the histone deacetylase SIRT6. Cell Rep 18(13):3052–3062. https://doi.org/10.1016/j.celrep.2017.03.008
Kantarci K, Senjem ML, Lowe VJ, Wiste HJ, Weigand SD, Kemp BJ, Frank AR, Shiung MM, Boeve BF, Knopman DS, Petersen RC, Jack CR (2010) Effects of age on the glucose metabolic changes in mild cognitive impairment. Am J Neuroradiol 31(7):1247–1253. https://doi.org/10.3174/ajnr.A2070
Kawanishi S, Oikawa S (2004) Mechanism of telomere shortening by oxidative stress. Ann N Y Acad Sci 1019(1):278–284. https://doi.org/10.1196/annals.1297.047
Kugel S, Mostoslavsky R (2014) Chromatin and beyond: the multitasking roles for SIRT6. Trends Biochem Sci 39(2):72–81. https://doi.org/10.1016/j.tibs.2013.12.002
Lagalwar S, Guillozet-Bongaarts AL, Berry RW, Binder LI (2006) Formation of phospho-SAPK/JNK granules in the hippocampus is an early event in Alzheimer disease. J Neuropathol Exp Neurol 65(5):455–464. https://doi.org/10.1097/01.jnen.0000229236.98124.d8
Landry J, Sutton A, Tafrov ST, Heller RC, Stebbins J, Pillus L, Sternglanz R (2000) The silencing protein SIR2 and its homologs are NAD-dependent protein deacetylases. Proc Natl Acad Sci 97(11):5807–5811. https://doi.org/10.1073/pnas.110148297
Lindahl T, Barnes D (2000) Repair of endogenous DNA damage. Cold Spring Harbor symposia on quantitative biology Cold Spring Harbor Laboratory Press, In, pp 127–134
Liszt G, Ford E, Kurtev M, Guarente L (2005) Mouse Sir2 homolog SIRT6 is a nuclear ADP-ribosyltransferase. J Biol Chem 280(22):21313–21320. https://doi.org/10.1074/jbc.M413296200
Liu M, Huo YR, Wang J, Wang C, Liu S, Liu S, Wang J, Ji Y (2016) Telomere shortening in Alzheimer’s disease patients. Ann Clin Lab Sci 46(3):260–265
Mao Z, Hine C, Tian X, van Meter M, Au M, Vaidya A, Seluanov A, Gorbunova V (2011) SIRT6 promotes DNA repair under stress by activating PARP1. Science 332(6036):1443–1446. https://doi.org/10.1126/science.1202723
Michan S, Sinclair D (2007) Sirtuins in mammals: insights into their biological function. Biochem J 404(1):1–13. https://doi.org/10.1042/BJ20070140
Michishita E, McCord RA, Berber E, Kioi M, Padilla-Nash H, Damian M, Cheung P, Kusumoto R, Kawahara TLA, Barrett JC, Chang HY, Bohr VA, Ried T, Gozani O, Chua KF (2008) SIRT6 is a histone H3 lysine 9 deacetylase that modulates telomeric chromatin. Nature 452(7186):492–496. https://doi.org/10.1038/nature06736
Mostoslavsky R, Chua KF, Lombard DB, Pang WW, Fischer MR, Gellon L, Liu P, Mostoslavsky G, Franco S, Murphy MM, Mills KD, Patel P, Hsu JT, Hong a, Ford E, Cheng HL, Kennedy C, Nunez N, Bronson R, Frendewey D, Auerbach W, Valenzuela D, Karow M, Hottiger MO, Hursting S, Barrett JC, Guarente L, Mulligan R, Demple B, Yancopoulos GD, Alt FW (2006) Genomic instability and aging-like phenotype in the absence of mammalian SIRT6. Cell 124(2):315–329. https://doi.org/10.1016/j.cell.2005.11.044
Multani AS, Chang S (2007) WRN at telomeres: implications for aging and cancer. J Cell Sci 120(5):713–721. https://doi.org/10.1242/jcs.03397
Outeiro TF, Marques O, Kazantsev A (2008) Therapeutic role of sirtuins in neurodegenerative disease. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1782:363–369
Prince M, Wimo A, Guerchet M, Ali G-C, Wu Y-T, Prina M (2015) World Alzheimer report 2015: the global impact of dementia Alzheimer’s Disease International (ADI)
Proctor CJ, Kirkwood TB (2002) Modelling telomere shortening and the role of oxidative stress. Mech Ageing Dev 123(4):351–363. https://doi.org/10.1016/S0047-6374(01)00380-3
Sancar A, Lindsey-Boltz LA, Ünsal-Kaçmaz K, Linn S (2004) Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu Rev Biochem 73(1):39–85. https://doi.org/10.1146/annurev.biochem.73.011303.073723
Santana I, Farinha F, Freitas S, Rodrigues V, Carvalho Á (2015) The epidemiology of dementia and Alzheimer disease in Portugal: estimations of prevalence and treatment-costs. Acta Medica Portuguesa 28(2):182–188. 10.20344/amp.6025
Simpson JE, Ince PG, Matthews FE, Shaw PJ, Heath PR, Brayne C, Garwood C, Higginbottom A, Wharton SB, MRC Cognitive Function and Ageing Neuropathology Study Group (2015) A neuronal DNA damage response is detected at the earliest stages of Alzheimer’s neuropathology and correlates with cognitive impairment in the Medical Research Council’s Cognitive Function and Ageing Study ageing brain cohort. Neuropathol Appl Neurobiol 41(4):483–496. https://doi.org/10.1111/nan.12202
Smith MA, Sayre LM, Monnier VM, Perry G (1995) Radical AGEing in Alzheimer’s disease. Trends Neurosci 18(4):172–176. https://doi.org/10.1016/0166-2236(95)93897-7
Tennen RI, Bua DJ, Wright WE, Chua KF (2011) SIRT6 is required for maintenance of telomere position effect in human cells. Nat Commun 2:433. https://doi.org/10.1038/ncomms1443
Wątroba M, Szukiewicz D (2016) The role of sirtuins in aging and age-related diseases. Adv Med Sci 61(1):52–62. https://doi.org/10.1016/j.advms.2015.09.003
Xiao C, Kim HS, Lahusen T, Wang RH, Xu X, Gavrilova O, Jou W, Gius D, Deng CX (2010) SIRT6 deficiency results in severe hypoglycemia by enhancing both basal and insulin-stimulated glucose uptake in mice. J Biol Chem 285(47):36776–36784. https://doi.org/10.1074/jbc.M110.168039
Xiao C, Wang RH, Lahusen TJ, Park O, Bertola A, Maruyama T, Reynolds D, Chen Q, Xu X, Young HA, Chen WJ, Gao B, Deng CX (2012) Progression of chronic liver inflammation and fibrosis driven by activation of c-JUN signaling in Sirt6 mutant mice. J Biol Chem 287(50):41903–41913. https://doi.org/10.1074/jbc.M112.415182
Yoshida H, Hastie CJ, McLauchlan H, Cohen P, Goedert M (2004) Phosphorylation of microtubule-associated protein tau by isoforms of c-Jun N-terminal kinase (JNK). J Neurochem 90(2):352–358. https://doi.org/10.1111/j.1471-4159.2004.02479.x
Acknowledgements
We would like to acknowledge Research University Top Down Grant (1001/PPSK/870035) and the USM-RIKEN Centre for Ageing Science (URICAS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declares that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mohamad Nasir, N.F., Zainuddin, A. & Shamsuddin, S. Emerging Roles of Sirtuin 6 in Alzheimer’s Disease. J Mol Neurosci 64, 157–161 (2018). https://doi.org/10.1007/s12031-017-1005-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-017-1005-y