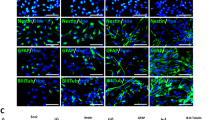
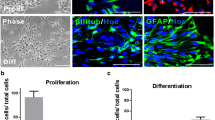
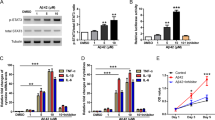
Abstract
Excessive accumulation of amyloid-β (Aβ) caused by cleavage of amyloid precursor protein (APP) is thought to be the primary cause of Alzheimer’s disease (AD). Two key enzymes ADAM10 and BACE1 are involved in the initial cleavage of APP, resulting in the onset of two pathways, the amyloidogenic pathway and the non-amyloidogenic pathway, respectively. Altering APP metabolism towards the non-amyloidogenic pathway is thought to reduce Aβ production. It has been reported that, in vivo, exogenous neurotrophic factors make APP apt to entering the non-amyloidogenic pathway. Since astrocytes secrete a battery of neurotrophic factors, we investigated the role of astrocyte-derived factors in the dynamics of Aβ generation in neural cells. Results show that C6 glioma cell-conditioned medium (GCM), obtained from cultured astrocyte-derived C6 glioma cells, inhibit Aβ1-42 production and shift APP processing towards the non-amyloidogenic pathway in APPswe-HEK293 cells. Such effect is attributed to two key APP cleavage enzymes, ADAM10 and BACE1. Two neurotrophic factors in the GCM, nerve growth factor and fibroblast growth factor 2, are responsible for the up-regulation of ADAM10 and down-regulation of BACE1, respectively. Our findings enhance our understanding of the relationship between astrocytes and Aβ generation, indicating that stimulation of astrocytic neurotrophic factors could slow AD progression.






Similar content being viewed by others
Abbreviations
- Aβ:
-
Amyloid β-protein
- AD:
-
Alzheimer’s disease
- ADAM10:
-
A disintegrin and metalloproteinase domain 10
- APP:
-
Amyloid precursor protein
- BACE1:
-
Beta-site amyloid precursor protein-cleaving enzyme 1
- BDNF:
-
Brain-derived neurotrophic factor
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- EGF:
-
Epidermal growth factor
- FGF2:
-
Fibroblast growth factor 2
- FGFR1:
-
Fibroblast growth factor receptor 1
- GCM:
-
Glioma cell-conditioned medium
- GDNF:
-
Glial cell line-derived neurotrophic factor
- NGF:
-
Nerve growth factor
- TrkA:
-
High affinity NGF tyrosine kinase receptor
References
Ardiles AO, Maripillán J, Lagos VL, Toro R, Mora IG, Villarroel L, Alés E, Borges R, Cárdenas AM (2006) A rapid exocytosis mode in chromaffin cells with a neuronal phenotype. J Neurochem 99:29–41
Avila-Munoz E, Arias C (2014) When astrocytes become harmful: functional and inflammatory responses that contribute to Alzheimer’s disease. Ageing Res Rev 18:29–40
Bonda DJ, Lee H-g, Camins A, Pallàs M, Casadesus G, Smith MA, Zhu X (2011) The sirtuin pathway in ageing and Alzheimer disease: mechanistic and therapeutic considerations. Lancet Neurol 10:275–279
Brown DR (1999) Neurons depend on astrocytes in a coculture system for protection from glutamate toxicity. Mol Cell Neurosci 13:379–389
Cai H, Wang Y, McCarthy D, Wen H, Borchelt DR, Price DL, Wong PC (2001) BACE1 is the major β-secretase for generation of Aβ peptides by neurons. Nat Neurosci 4:233–234
Campos FL, Cristovao AC, Rocha SM, Fonseca CP, Baltazar G (2012) GDNF contributes to oestrogen-mediated protection of midbrain dopaminergic neurones. J Neuroendocrinol 24:1386–1397
Colombo A, Wang H, Kuhn P-H, Page R, Kremmer E, Dempsey PJ, Crawford HC, Lichtenthaler SF (2013) Constitutive α- and β-secretase cleavages of the amyloid precursor protein are partially coupled in neurons, but not in frequently used cell lines. Neurobiol Dis 49:137–147
Dawbarn D, Allen SJ (2003) Neurotrophins and neurodegeneration. Neuropathol Appl Neurobiol 29:211–230
de Bernardo S, Canals S, Casarejos MJ, Rodriguez-Martin E, Mena MA (2003) Glia-conditioned medium induces de novo synthesis of tyrosine hydroxylase and increases dopamine cell survival by differential signaling pathways. J Neurosci Res 73:818–830
Dhandapani KM, Hadman M, De Sevilla L, Wade MF, Mahesh VB, Brann DW (2003) Astrocyte protection of neurons: role of transforming growth factor-beta signaling via a c-Jun-AP-1 protective pathway. J Biol Chem 278:43329–43339
Esposito E, Iacono A, Muià C, Crisafulli C, Mattace Raso G, Bramanti P, Meli R, Cuzzocrea S (2008) Signal transduction pathways involved in protective effects of melatonin in C6 glioma cells. J Pineal Res 44:78–87
Feng Z, Zhang JT (2004) Protective effect of melatonin on beta-amyloid-induced apoptosis in rat astroglioma C6 cells and its mechanism. Free Radic Biol Med 37:1790–1801
Fisk L, Nalivaeva NN, Boyle JP, Peers CS, Turner AJ (2007) Effects of hypoxia and oxidative stress on expression of neprilysin in human neuroblastoma cells and rat cortical neurones and astrocytes. Neurochem Res 32:1741–1748
Furman JL, Sama DM, Gant JC, Beckett TL, Murphy MP, Bachstetter AD, Van Eldik LJ, Norris CM (2012) Targeting astrocytes ameliorates neurologic changes in a mouse model of Alzheimer’s disease. J Neurosci 32:16129–16140
Gandhi S, Refolo LM, Sambamurti K (2004) Amyloid precursor protein compartmentalization restricts beta-amyloid production: therapeutic targets based on BACE compartmentalization. J Mol Neurosci 24:137–143
Gu X, Sun J, Li S, Wu X, Li L (2013) Oxidative stress induces DNA demethylation and histone acetylation in SH-SY5Y cells: potential epigenetic mechanisms in gene transcription in Aβ production. Neurobiol Aging 34:1069–1079
Hailer NP, Wirjatijasa F, Roser N, Hischebeth GT, Korf HW, Dehghani F (2001) Astrocytic factors protect neuronal integrity and reduce microglial activation in an in vitro model of N-methyl-D-aspartate-induced excitotoxic injury in organotypic hippocampal slice cultures. Eur J Neurosci 14:315–326
Heneka MT, Sastre M, Dumitrescu-Ozimek L, Dewachter I, Walter J, Klockgether T, Van Leuven F (2005) Focal glial activation coincides with increased BACE1 activation and precedes amyloid plaque deposition in APP[V717I] transgenic mice. J Neuroinflammation 2:22
Jiang Q, Yan Z, Feng J (2006) Neurotrophic factors stabilize microtubules and protect against rotenone toxicity on dopaminergic neurons. J Biol Chem 281:29391–29400
Katsouri L, Ashraf A, Birch AM, Lee KK, Mirzaei N, Sastre M (2014) Systemic administration of fibroblast growth factor-2 (FGF2) reduces BACE1 expression and amyloid pathology in APP23 mice. Neurobiol Aging
Kells AP, Fong DM, Dragunow M, During MJ, Young D, Connor B (2004) AAV-mediated gene delivery of BDNF or GDNF is neuroprotective in a model of Huntington disease. Mol Ther 9:682–688
Kraft AW, Hu X, Yoon H, Yan P, Xiao Q, Wang Y, Gil SC, Brown J, Wilhelmsson U, Restivo JL (2013) Attenuating astrocyte activation accelerates plaque pathogenesis in APP/PS1 mice. FASEB J 27:187–198
Kuhn PH, Wang H, Dislich B, Colombo A, Zeitschel U, Ellwart JW, Kremmer E, Rossner S, Lichtenthaler SF (2010) ADAM10 is the physiologically relevant, constitutive alpha-secretase of the amyloid precursor protein in primary neurons. EMBO J 29:3020–3032
Lammich S, Kojro E, Postina R, Gilbert S, Pfeiffer R, Jasionowski M, Haass C, Fahrenholz F (1999) Constitutive and regulated alpha-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc Natl Acad Sci U S A 96:3922–3927
Lammich S, Buell D, Zilow S, Ludwig A-K, Nuscher B, Lichtenthaler SF, Prinzen C, Fahrenholz F, Haass C (2010) Expression of the anti-amyloidogenic secretase ADAM10 is suppressed by its 5′-untranslated region. J Biol Chem 285:15753–15760
Martinez R, Gomes FC (2002) Neuritogenesis induced by thyroid hormone-treated astrocytes is mediated by epidermal growth factor/mitogen-activated protein kinase-phosphatidylinositol 3-kinase pathways and involves modulation of extracellular matrix proteins. J Biol Chem 277:49311–49318
Matrone C, Ciotti MT, Mercanti D, Marolda R, Calissano P (2008a) NGF and BDNF signaling control amyloidogenic route and Abeta production in hippocampal neurons. Proc Natl Acad Sci U S A 105:13139–13144
Matrone C, Di Luzio A, Meli G, D’Aguanno S, Severini C, Ciotti MT, Cattaneo A, Calissano P (2008b) Activation of the amyloidogenic route by NGF deprivation induces apoptotic death in PC12 cells. J Alzheimers Dis 13:81–96
Misonou H, Morishima-Kawashima M, Ihara Y (2000) Oxidative stress induces intracellular accumulation of amyloid β-protein (Aβ) in human neuroblastoma cells. Biochemistry 39:6951–6959
Mori T, Koyama N, Arendash GW, Horikoshi-Sakuraba Y, Tan J, Town T (2010) Overexpression of human S100B exacerbates cerebral amyloidosis and gliosis in the Tg2576 mouse model of Alzheimer’s disease. Glia 58:300–314
O’Brien RJ, Wong PC (2011) Amyloid precursor protein processing and Alzheimer’s disease. Annu Rev Neurosci 34:185–204
Postina R, Schroeder A, Dewachter I, Bohl J, Schmitt U, Kojro E, Prinzen C, Endres K, Hiemke C, Blessing M (2004) A disintegrin-metalloproteinase prevents amyloid plaque formation and hippocampal defects in an Alzheimer disease mouse model. J Clin Investig 113:1456
Prince M, Bryce R, Albanese E, Wimo A, Ribeiro W, Ferri CP (2013) The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement 9:63–75.e62
Quincozes‐Santos A, Gottfried C (2011) Resveratrol modulates astroglial functions: neuroprotective hypothesis. Ann N Y Acad Sci 1215:72–78
Ramirez G, Toro R, Dobeli H, von Bernhardi R (2005) Protection of rat primary hippocampal cultures from A beta cytotoxicity by pro-inflammatory molecules is mediated by astrocytes. Neurobiol Dis 19:243–254
Saragovi HU, Gehring K (2000) Development of pharmacological agents for targeting neurotrophins and their receptors. Trends Pharmacol Sci 21:93–98
Selkoe DJ, Schenk D (2003) Alzheimer’s disease: molecular understanding predicts amyloid-based therapeutics. Annu Rev Pharmacol Toxicol 43:545–584
Tanaka J, Toku K, Zhang B, Ishihara K, Sakanaka M, Maeda N (1999) Astrocytes prevent neuronal death induced by reactive oxygen and nitrogen species. Glia 28:85–96
Thinakaran G, Koo EH (2008) Amyloid precursor protein trafficking, processing, and function. J Biol Chem 283:29615–29619
Tian L, Guo R, Yue X, Lv Q, Ye X, Wang Z, Chen Z, Wu B, Xu G, Liu X (2012) Intranasal administration of nerve growth factor ameliorate beta-amyloid deposition after traumatic brain injury in rats. Brain Res 1440:47–55
Wahlberg LU, Lind G, Almqvist PM, Kusk P, Tornoe J, Juliusson B, Soderman M, Sellden E, Seiger A, Eriksdotter-Jonhagen M, Linderoth B (2012) Targeted delivery of nerve growth factor via encapsulated cell biodelivery in Alzheimer disease: a technology platform for restorative neurosurgery. J Neurosurg 117:340–347
Williams BJ, Eriksdotter-Jonhagen M, Granholm AC (2006) Nerve growth factor in treatment and pathogenesis of Alzheimer’s disease. Prog Neurobiol 80:114–128
Woodbury ME, Ikezu T (2014) Fibroblast growth factor-2 signaling in neurogenesis and neurodegeneration. J NeuroImmune Pharmacol : Off J Soc NeuroImmune Pharmacol 9:92–101
Xue B, Jiao J, Zhang L, Li KR, Gong YT, Xie JX, Wang XM (2007) Triptolide upregulates NGF synthesis in rat astrocyte cultures. Neurochem Res 32:1113–1119
Yan R, Vassar R (2014) Targeting the β secretase BACE1 for Alzheimer’s disease therapy. Lancet Neurol 13:319–329
Yang C, Liu Y, Ni X, Li N, Zhang B, Fang X (2014) Enhancement of the nonamyloidogenic pathway by exogenous NGF in an Alzheimer transgenic mice model. Neuropeptides
Yi P, Schrott L, Castor T, Alexander J (2012) Bryostatin-1 vs. TPPB: dose-dependent APP processing and PKC-α,-δ, and-ε isoform activation in SH-SY5Y neuronal cells. J Mol Neurosci 48:234–244
Zhang Y, Tatsuno T, Carney JM, Mattson MP (1993) Basic FGF, NGF, and IGFs protect hippocampal and cortical neurons against iron-induced degeneration. J Cereb Blood Flow Metab 13:378–388
Acknowledgments
We are grateful to Professor Hui Fu (Wuhan University, School of Medicine, China) for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Funding
This work was supported by the National Natural Science Foundation of China (31371331).
Rights and permissions
About this article
Cite this article
Xie, H., Xiao, Z. & Huang, J. C6 Glioma-Secreted NGF and FGF2 Regulate Neuronal APP Processing Through Up-Regulation of ADAM10 and Down-Regulation of BACE1, Respectively. J Mol Neurosci 59, 334–342 (2016). https://doi.org/10.1007/s12031-015-0690-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0690-7