Abstract
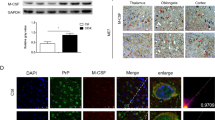
Microglial cells are resident mononuclear phagocytes of the central nervous system (CNS). Active proliferation of microglia in the brain has been identified in neurodegenerative disorders, including some kinds of prion disease. However, the detailed regional distribution between microglia and PrPSc deposition has not been presented, and investigation of fractalkine signaling which is involved in the regulation of activation of microglia in prion disease is not well documented. In this study, the disease phenomenon of microglial accumulation in the CNS was thoroughly analyzed using a scrapie-infected experimental model. Western blots of microglia-specific markers Iba1 and CD68, immunohistochemical and immunofluorescent assays demonstrated obviously activation of microglia in almost whole brain regions in the infected animals. Under the dynamic analysis on hallmarks of activation of microglia, a time-dependent increase of Iba1 and CD68 was detected, accompanied by accumulation of PrPSc and progression of neurodegenerative symptoms. With serial brain sections and double staining of Iba1 and PrPSc, we observed that the microglia distributed around PrPSc deposits in 263K-infected hamsters’ brains, proposing PrPSc phagocytosis. Flow cytometry assays with the single-cell suspensions prepared from the cortical region of the infected brains verified an activation of microglial population. ELISA assays of the cytokines in brain homogenates revealed significant upregulations of interleukin (IL)-1β, IL-6 and TNF-α when infected. Evaluation of fractalkine signaling in the infected hamsters’ brains showed progressively downregulation of CX3CL1 during the incubation. Prion peptide PrP106-126 also disrupted fractalkine and evoked microglial activation in rat primary neuron–glia mixed cultures. Our data here demonstrate an activated status of microglia in CNS tissues of infectious prion disease, possibly through fractalkine signaling deficiency.









Similar content being viewed by others
References
Akiyama H, McGeer PL (1990) Brain microglia constitutively express beta-2 integrins. J Neuroimmunol 30:81–93
Baker CA, Martin D, Manuelidis L (2002) Microglia from Creutzfeldt–Jakob disease-infected brains are infectious and show specific mRNA activation profiles. J Virol 76:10905–10913
Balu DT, Hodes GE, Hill TE et al (2009) Flow cytometric analysis of BrdU incorporation as a high-throughput method for measuring adult neurogenesis in the mouse. J Pharmacol Toxicol Methods 59:100–107
Campbell IL, Eddleston M, Kemper P, Oldstone MB, Hobbs MV (1994) Activation of cerebral cytokine gene expression and its correlation with onset of reactive astrocyte and acute-phase response gene expression in scrapie. J Virol 68:2383–2387
Cardona AE, Pioro EP, Sasse ME et al (2006) Control of microglial neurotoxicity by the fractalkine receptor. Nat Neurosci 9:917–924
Chang RC, Hudson PM, Wilson BC, Liu B, Abel H, Hong JS (2000) High concentrations of extracellular potassium enhance bacterial endotoxin lipopolysaccharide-induced neurotoxicity in glia-neuron mixed cultures. Neuroscience 97:757–764
Chapman GA, Moores K, Harrison D, Campbell CA, Stewart BR, Strijbos PJ (2000) Fractalkine cleavage from neuronal membranes represents an acute event in the inflammatory response to excitotoxic brain damage. J Neurosci 20:RC87
Cho SH, Sun B, Zhou Y et al (2011) CX3CR1 protein signaling modulates microglial activation and protects against plaque-independent cognitive deficits in a mouse model of Alzheimer disease. J Biol Chem 286:32713–32722
da Silva RP, Gordon S (1999) Phagocytosis stimulates alternative glycosylation of macrosialin (mouse CD68), a macrophage-specific endosomal protein. Biochem J 338(Pt 3):687–694
Dickson DW, Mattiace LA, Kure K, Hutchins K, Lyman WD, Brosnan CF (1991) Microglia in human disease, with an emphasis on acquired immune deficiency syndrome. Lab Invest 64:135–156
Fuhrmann M, Bittner T, Jung CK et al (2010) Microglial Cx3cr1 knockout prevents neuron loss in a mouse model of Alzheimer’s disease. Nat Neurosci 13:411–413
Gao JM, Gao C, Han J et al (2004) Dynamic analyses of PrP and PrP(Sc) in brain tissues of golden hamsters infected with scrapie strain 263 K revealed various PrP forms. Biomed Environ Sci 17:8–20
Graeber MB (2010) Changing face of microglia. Science 330:783–788
Guiroy DC, Wakayama I, Liberski PP, Gajdusek DC (1994) Relationship of microglia and scrapie amyloid-immunoreactive plaques in kuru, Creutzfeldt–Jakob disease and Gerstmann–Straussler syndrome. Acta Neuropathol 87:526–530
Harrison JK, Jiang Y, Chen S et al (1998) Role for neuronally derived fractalkine in mediating interactions between neurons and CX3CR1-expressing microglia. Proc Natl Acad Sci U S A 95:10896–10901
Hughes PM, Botham MS, Frentzel S, Mir A, Perry VH (2002) Expression of fractalkine (CX3CL1) and its receptor, CX3CR1, during acute and chronic inflammation in the rodent CNS. Glia 37:314–327
Jeffrey M, Halliday WG, Bell J et al (2000) Synapse loss associated with abnormal PrP precedes neuronal degeneration in the scrapie-infected murine hippocampus. Neuropathol Appl Neurobiol 26:41–54
Kordek R, Nerurkar VR, Liberski PP, Isaacson S, Yanagihara R, Gajdusek DC (1996) Heightened expression of tumor necrosis factor alpha, interleukin 1 alpha, and glial fibrillary acidic protein in experimental Creutzfeldt–Jakob disease in mice. Proc Natl Acad Sci U S A 93:9754–9758
Kreutzberg GW (1996) Microglia: A sensor for pathological events in the CNS. Trends Neurosci 19:312–318
Ladeby R, Wirenfeldt M, Dalmau I et al (2005a) Proliferating resident microglia express the stem cell antigen CD34 in response to acute neural injury. Glia 50:121–131
Ladeby R, Wirenfeldt M, Garcia-Ovejero D et al (2005b) Microglial cell population dynamics in the injured adult central nervous system. Brain Res Brain Res Rev 48:196–206
Lamb B (2012) The roles of fractalkine signaling in neurodegenerative disease. Mol Neurodegener 7(Suppl 1):L21
Lawson LJ, Perry VH, Gordon S (1992) Turnover of resident microglia in the normal adult mouse brain. Neuroscience 48:405–415
Mackay CR (1997) Chemokines: What chemokine is that? Curr Biol 7:R384–R386
Marella M, Chabry J (2004) Neurons and astrocytes respond to prion infection by inducing microglia recruitment. J Neurosci 24:620–627
McGeer PL, Kawamata T, Walker DG, Akiyama H, Tooyama I, McGeer EG (1993) Microglia in degenerative neurological disease. Glia 7:84–92
Meda L, Baron P, Scarlato G (2001) Glial activation in Alzheimer’s disease: The role of Abeta and its associated proteins. Neurobiol Aging 22:885–893
Mizuno T, Kawanokuchi J, Numata K, Suzumura A (2003) Production and neuroprotective functions of fractalkine in the central nervous system. Brain Res 979:65–70
Monif M, Burnstock G, Williams DA (2010) Microglia: Proliferation and activation driven by the P2X7 receptor. Int J Biochem Cell Biol 42:1753–1756
Nimmerjahn A, Kirchhoff F, Helmchen F (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308:1314–1318
Pan Y, Lloyd C, Zhou H et al (1997) Neurotactin, a membrane-anchored chemokine upregulated in brain inflammation. Nature 387:611–617
Perry VH, Gordon S (1988) Macrophages and microglia in the nervous system. Trends Neurosci 11:273–277
Sasaki A, Hirato J, Nakazato Y (1993) Immunohistochemical study of microglia in the Creutzfeldt–Jakob diseased brain. Acta Neuropathol 86:337–344
Shi XH, Han J, Zhang J et al (2010) Clinical, histopathological and genetic studies in a family with fatal familial insomnia. Infect Genet Evol 10:292–297
Solito E, Sastre M (2012) Microglia function in Alzheimer’s disease. Front Pharmacol 3:14
Streit WJ, Walter SA, Pennell NA (1999) Reactive microgliosis. Prog Neurobiol 57:563–581
Tooyama I, Kimura H, Akiyama H, McGeer PL (1990) Reactive microglia express class I and class II major histocompatibility complex antigens in Alzheimer’s disease. Brain Res 523:273–280
Williams A, Lucassen PJ, Ritchie D, Bruce M (1997a) PrP deposition, microglial activation, and neuronal apoptosis in murine scrapie. Exp Neurol 144:433–438
Williams A, Van Dam AM, Ritchie D, Eikelenboom P, Fraser H (1997b) Immunocytochemical appearance of cytokines, prostaglandin E2 and lipocortin-1 in the CNS during the incubation period of murine scrapie correlates with progressive PrP accumulations. Brain Res 754:171–180
Woodroofe MN, Sarna GS, Wadhwa M et al (1991) Detection of interleukin-1 and interleukin-6 in adult rat brain, following mechanical injury, by in vivo microdialysis: Evidence of a role for microglia in cytokine production. J Neuroimmunol 33:227–236
Zhang J, Chen L, Zhang BY et al (2004) Comparison study on clinical and neuropathological characteristics of hamsters inoculated with scrapie strain 263 K in different challenging pathways. Biomed Environ Sci 17:65–78
Zujovic V, Benavides J, Vige X, Carter C, Taupin V (2000) Fractalkine modulates TNF-alpha secretion and neurotoxicity induced by microglial activation. Glia 29:305–315
Acknowledgments
This work was supported by Chinese National Natural Science Foundation Grants (81100980), China Mega-Project for Infectious Disease (2011X10004-101), the SKLID Development Grant (2008SKLID102, 2011SKLID204, and 2011SKLID211), and the Young Scholar Scientific Research Foundation of China CDC (2012A102).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 4519 kb)
Rights and permissions
About this article
Cite this article
Xie, WL., Shi, Q., Zhang, J. et al. Abnormal Activation of Microglia Accompanied with Disrupted CX3CR1/CX3CL1 Pathway in the Brains of the Hamsters Infected with Scrapie Agent 263K. J Mol Neurosci 51, 919–932 (2013). https://doi.org/10.1007/s12031-013-0002-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-013-0002-z