Abstract
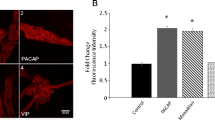
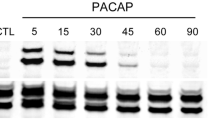
Calcium influx is required for the pituitary adenylyl cyclase activating polypeptide (PACAP)-induced increase in guinea pig cardiac neuron excitability, noted as a change from a phasic to multiple action potential firing pattern. Intracellular recordings indicated that pretreatment with the nonselective cationic channel inhibitors, 2-aminoethoxydiphenylborate (2-APB), 1-[β-[3-(4-methoxyphenyl)propoxy]-4-methoxyphenethyl]-1H-imidazole HCl (SKF 96365), and flufenamic acid (FFA) reduced the 20-nM PACAP-induced excitability increase. Additional experiments tested whether 2-APB, FFA, and SKF 96365 could suppress the increase in excitability by PACAP once it had developed. The increased action potential firing remained following application of 2-APB but was diminished by FFA. SKF 96365 transiently depressed the PACAP-induced excitability increase. A decrease and recovery of action potential amplitude paralleled the excitability shift. Since semiquantitative PCR indicated that cardiac neurons express TRPC subunit transcripts, we hypothesize that PACAP activates calcium-permeable, nonselective cationic channels, which possibly are members of the TRPC family. Our results are consistent with calcium influx being required for the initiation of the PACAP-induced increase in excitability, but suggest that it may not be required to sustain the peptide effect. The present results also demonstrate that nonselective cationic channel inhibitors could have other actions, which might contribute to the inhibition of the PACAP-induced excitability increase.





Similar content being viewed by others
References
Adams DJ, Cuevas J (2004) Electrophysiological properties of intrinsic cardiac neurons. In: Armour JA, Ardell JL (eds) Basic and clinical neurocardiology. Oxford University Press, Oxford, pp 1–60
Beaudet MM, Parsons RL, Braas KM, May V (2000) Mechanisms mediating pituitary adenylate cyclase-activating polypeptide depolarization of rat sympathetic neurons. J Neurosci 20:7353–7361
Braas KM, May V (1999) Pituitary adenylate cyclase-activating polypeptides directly stimulate sympathetic neuron neuropeptide Y release through PAC1 receptor activation of specific intracellular signaling pathways. J Biol Chem 39:27702–27710
Braas KM, May V, Harakall SA, Hardwick JC, Parsons RL (1998) Pituitary adenylate cyclase-activating polypeptide expression and modulation of neuronal excitability in guinea pig cardiac ganglia. J Neurosci 18:9766–9779
Braas KM, Rossignal TM, Girard BM, May V, Parsons RL (2004) Pituitary adenylyl cyclase activating polypeptide (PACAP) decreases neuronal somatostatin immunoreactivity in cultured guinea-pig parasympathetic cardiac ganglia. Neurosci 126:335–346
Calupca MA, Vizzard MA, Parsons RL (2000) Origin of pituitary adenylate cyclase-activating polypeptide (PACAP)-immunoreactive fibers innervating guinea pig parasympathetic cardiac ganglia. J Comp Neurol 423:26–39
Calupca MA, Locknar SA, Parsons RL (2002) TRPC6 immunoreactivity is colocalized with neuronal nitric oxide synthase in extrinsic fibers innervating guinea pig intrinsic cardiac ganglia. J Comp Neurol 450:283–291
Chatterjee TK, Sharmi RV, Fisher RA (1996) Molecular cloning of a novel variant of the pituitary adenylate cyclase-activating polypeptide (PACAP) receptor that stimulates calcium influx by activation of L-type calcium channels. J Biol Chem 271:32226–32232
Clapham DE, Runnels LW, Strubing C (2001) The TRP ion channel family. Nat Rev Neurosci 2:387–396
Edwards FR, Hirst GD, Klemm MF, Steele PA (1995) Different types of ganglion cell in the cardiac plexus of guinea-pigs. J Physiol 486:453–471
Hoover DB, Tompkins JD, Parsons RL (2009) Differential activation of guinea pig intrinsic cardiac neurons by the PAC1 agonists maxadilan and pituitary adenylate cyclase-activating polypeptide 27 (PACAP27). J Pharmacol Exp Ther 331:197–203
Jan CR, Ho CM, Wu SN, Tseng CJ (1999) Multiple effects of 1-[β-[3-(4-methoxyphenyl) propoxy]-4-methoxy-phenyl]-1H-imidazole hydrochloride (SKF 96365) on Ca2+ signaling in MDCK cells: depletion of thapsigargin-sensitive Ca2+ store followed by capacitative Ca2+ entry, activation of direct Ca2+ entry, and inhibition of thapsigargin-induced capacitative Ca2+ entry. Naunyn-Schmiedeberg’s Arch Pharmacol 359:92–101
Liu S, Qu MH, Ren W et al (2008) Differential expression of canonical (classical) transient receptor potential channels in guinea pig enteric nervous system. J Comp Neurol 511:847–862
Merriam LA, Barstow KL, Parsons RL (2004) Pituitary adenylate cyclase-activating polypeptide enhances the hyperpolarization-activated nonselective cationic conductance, Ih, in dissociated guinea pig intracardiac neurons. Regul Pept 123:123–133
Merriam LA, Locknar SA, Girard BM, Parsons RL (2010) Somatic ATP release from guinea pig sympathetic neurons does not require calcium-induced calcium release from internal stores. Am J Physiol Cell Physiol 299:836–843
Parsons RL, Locknar SA, Young BA, Hoard JL, Hoover DB (2006) Presence and co-localization of vasoactive intestinal polypeptide (VIP) with neuronal nitric oxide synthase (nNOS) in cells and nerve fibers within guinea pig intrinsic cardiac ganglia and cardiac tissue. Cell Tiss Res 323:197–209
Strubing C, Krapivinsky G, Krapivinsky L, Clapham DE (2001) TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron 29:645–655
Tompkins JD, Parsons RL (2008) Identification of intracellular signaling cascades mediating the PACAP-induced increase in guinea pig cardiac neuron excitability. J Mol Neurosci 36:292–298
Tompkins JD, Hardwick JC, Locknar SA, Merriam LA, Parsons RL (2006) Ca2+ influx, but not Ca2+ release from internal stores, is required for the PACAP-induced increase in excitability in guinea pig intracardiac neurons. J Neurophysiol 95:2134–2142
Tompkins JD, Ardell JL, Hoover DB, Parsons RL (2007) Neurally released pituitary adenylate cyclase-activating polypeptide enhances guinea pig intrinsic cardiac neuron excitability. J Physiol 582:87–93
Tompkins JD, Lawrence YT, Parsons RL (2009) Enhancement of I h, but not inhibition of I M, is a key mechanism underlying the PACAP-induced increase in excitability of guinea pig intrinsic cardiac neurons. Am J Physiol Regul Integr Comp Physiol 297:R52–R59
Vaudry D, Falluel-Morel A, Bourgault S et al (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61:283–357
Yan HD, Villalobos C, Andrade R (2009) TRPC channels mediate a muscarinic receptor-induced after depolarization in cerebral cortex. J Neurosci 29:10038–10046
Yau HJ, Baranauskas G, Martina M (2010) Flufenamic acid decreases neuronal excitability through modulation of voltage-gated sodium channel gating. J Physiol 588:3869–3882
Acknowledgments
This work was supported in part by NIH grants P20 RR16435, P30 RR032135, and P30 GM103498 to RLP.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Merriam, L.A., Roman, C.W., Baran, C.N. et al. Pretreatment with Nonselective Cationic Channel Inhibitors Blunts the PACAP-Induced Increase in Guinea Pig Cardiac Neuron Excitability. J Mol Neurosci 48, 721–729 (2012). https://doi.org/10.1007/s12031-012-9763-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9763-z